Abstract
Increased utilization of chemotherapy in breast cancer patients has led to improved survival outcomes but it has also resulted in rising incidence of adverse effects. Occurrence of new/unreported side effect poses challenge in front of clinicians. We report the case of a 53-year lady with locally advanced, hormone receptor-positive, and human epidermal growth factor-2 (HER-2) negative right breast carcinoma. She was started on neoadjuvant chemotherapy (NACT) (doxorubicin and cyclophosphamide), to facilitate breast-conserving surgery. She developed an inflammatory reaction involving the affected breast after each of three cycles of NACT (2 cycles of doxorubicin & cyclophosphamide, and 1 cycle of docetaxel). Infectious causes and disease progression were ruled out. She was then prescribed hormone therapy but the disease progressed after three months of therapy and the patient had to be subjected to modified radical mastectomy (MRM). She then received adjuvant radiotherapy and is currently doing well on second-line hormone therapy.
Keywords
Introduction
Chemotherapeutic drugs act on rapidly dividing malignant cells with a high turnover rate by disrupting specific phases of the cell cycle [1]. The non-specific nature of these drugs affects the vital metabolic reactions in normal tissues as well [1]. Many adverse effects of chemotherapeutic drugs have been well described and strategies to manage these adverse effect have been devised [2–5]. Inflammatory reactions to chemotherapeutic drugs are rare and have been reported only in a handful of case reports [6–9]. Sometimes it is difficult to delineate the exact etiology of such presentations.
Doxorubicin along with cyclophosphamide (AC regimen), followed by docetaxel has been used for the neo/adjuvant therapy of breast cancer as first line agents for many years [10] and their side effect profile is well known [10,11]. We report a case of locally advanced breast cancer with the development of acute inflammation of the breast after neoadjuvant chemotherapy with two most widely recommended regimens for breast carcinoma [12].
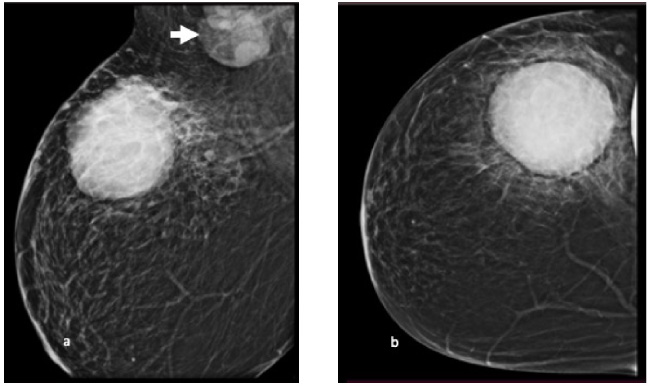
Mammogram at diagnosis- Mediolateral oblique (a) and cranio-caudal view (b) of right breast shows a large, well circumscribed, round shaped high density mass (in upper outer quadrant). Multiple enlarged lymph nodes are seen in the axilla (white arrow).
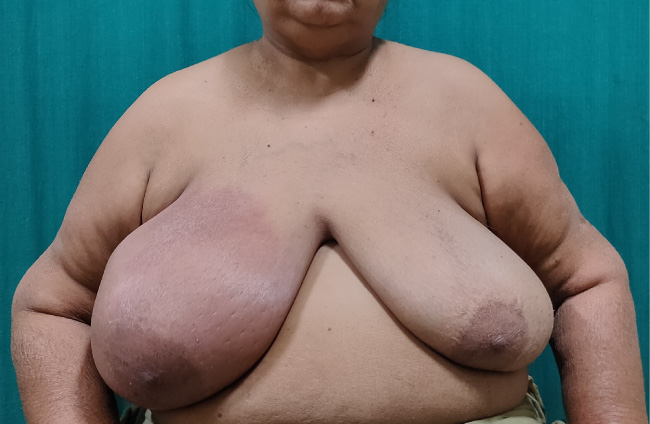
Edema, and erythema of the right breast after first cycle of chemotherapy (AC regimen).
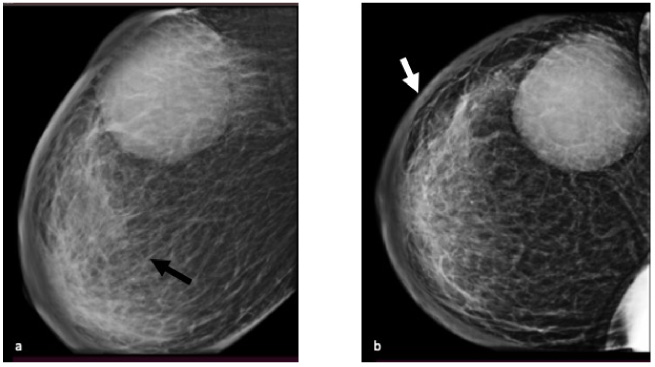
Mammogram after first cycle of chemotherapy (AC regimen)—Mediolateral oblique (a) and cranio-caudal view (b) of right breast shows diffuse skin thickening (white arrow) and diffusely increased parenchymal density with coarse trabeculations (black arrow) suggestive of edematous breast parenchyma, in addition to mass in upper outer quadrant.
A 53-year-old postmenopausal lady, a known diabetic, and hypertensive with no known history of allergy, presented to us with a history of a lump in her right breast for the last six months. She had been on long-term steroids (dexamethasone 0.5 mg daily) for pain in her knees. Physical examination revealed a hard lump of 7 × 6 cm size in the upper outer quadrant of the right breast, free from skin and chest wall, with multiple hard mobile matted lymph nodes in the ipsilateral axilla, largest being 4 × 4 cm in size. The remaining examination was unremarkable. Mammography revealed a large well-circumscribed, irregular, heterogeneous, high-density lesion (BIRADS 4C) (Fig. 1). Core biopsy of the lesion from the right breast confirmed invasive breast carcinoma. It was estrogen receptor (ER) negative, progesterone receptors (PR) positive, HER-2 negative by immunohistochemistry (IHC), and ki-67 was 60–65%. There were no distant metastasis observed on CECT of chest, abdomen, and bone scan. She was staged having cT3N2aM0 disease. After discussion in the multidisciplinary tumor board (MDT) neoadjuvant chemotherapy was planned to facilitate breast conserving surgery. Three days following the first cycle of AC regimen [Doxorubicin (60 mg/
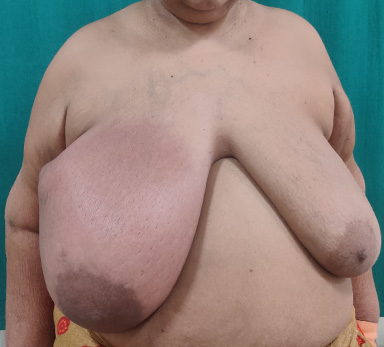
Edema, and erythema of the right breast after first cycle of docetaxel.
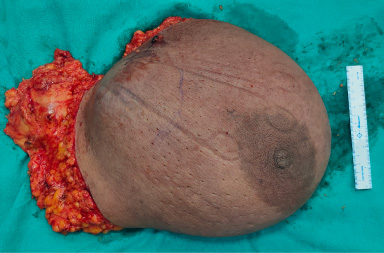
MRM specimen.
This case of locally advanced breast cancer is noteworthy due to the development of an acute inflammatory reaction in the affected breast in response to the two most commonly employed regimens of chemotherapy for breast carcinoma [1]. As a result, neoadjuvant chemotherapy had to be stopped and alternative treatment modalities were utilized.
The patient was prescribed neoadjuvant hormone therapy (tab letrozole 2.5 mg daily), since downsizing the tumor to facilitate BCS was the primary goal. Although not a standard approach, there is some evidence that postmenopausal, hormone receptor positive tumors, neoadjuvant hormone therapy can downstage tumors in majority of the patients within 3–4 months [13,14]. In addition to all the advantages that preoperative chemotherapy offers, hormonal agents also have a much favourable toxicity profile. The only downside seems to be a supposedly longer duration of treatment required to achieve response. Aromatase inhibitors have been shown to provide better response rates, pathological complete response rates, and breast conservation rates than tamoxifen and are the preferred agent [14]. Unfortunately, our patient did not respond to the hormone therapy and after three months of therapy the disease showed signs of progression and had to be stopped.
Such inflammatory reactions to cancer chemotherapy have rarely been reported and may represent tumor cell death or response to therapy. Feng et al. described a similar reaction to docetaxel therapy in a patient with breast carcinoma which raised concerns for tumor necrosis, mastitis, or disease progression [2]. They speculated that the heightened immune response can be attributed to tumor necrosis. But, even in their case, the patient had tolerated four cycles of the AC regimen uneventfully. Ruling out infectious etiologies and disease progression seems to be a reasonable outlook in such cases. Even though such reactions are not well reported, they can present in unexpected settings without any previous indications or history. The inflammatory reaction could not be attributed to any particular chemotherapeutic drugs and the cause remained unclear.
Footnotes
Funding
No funding was received for this work.
Conflicts of interest
None of the authors disclose any conflict of interest, financial or otherwise.
