Abstract
BACKGROUND:
Breast cancer (BC) is the most common cancer among women worldwide and a leading cause of death in Indonesia. The primary treatment of locally advanced BC is neoadjuvant chemotherapy (NAC). The rapid proliferation of tumor cells in a neoplastic microenvironment is largely due to hypoxia, which also encourages the development of chemoresistant BC. The master regulator of the hypoxia response is hypoxia-inducible factor-1α (HIF-1α). The response evaluation criteria in solid tumors (RECIST) is an objective response metric that demonstrates the efficacy of a NAC based mostly on the size of the tumor. Ca15-3 is the protein product of the MUC1 gene and is the most widely used serum marker in BC. The purpose of this study is to investigate the relationship between HIF-1α and RECIST and between Ca15-3 and RECIST and to assess the relationship among all of them in BC.
METHODS:
This observational study used the prospective cohort method included 11 patients with histopathologically confirmed BC, specifically invasive ductal carcinoma. We evaluated the changes in HIF-1α and Ca15-3 serum levels using ELISA and measured tumor lesions with RECIST. The procedure was carried out twice. Serum levels were measured at baseline, and after receiving two cycles of NAC (5 weeks).
RESULTS:
Among the 11 patients included in this study, HIF-1α, Ca15-3, and RECIST decreased significantly after NAC. The changes in RECIST correlated with Ca15-3: each unit decrease in RECIST score was associated with a 0.3-unit decrease in Ca15-3 levels (
CONCLUSIONS:
There was a decrease in HIF-1α, followed by a decrease in Ca15-3 and RECIST in response to chemotherapy. There was a statistically significant correlation between Ca15-3 and response to chemotherapy. This study evidences the relationship between factors that shape the local tumor microenvironment.
Background
According to the Global Cancer Observatory (GLOBOCAN 2018), breast cancer (BC) is the most common cancer among Indonesian women [1]. In Indonesia, it registered around 65,858 (16.6%) and accounted for 22,430 (9.6%) deaths of all cancer. The majority of patients with BC are diagnosed at stages III and IV [2]. In recent years, the main treatment choice for locally advanced BC (LABC) is neoadjuvant chemotherapy (NAC), a primary systemic preoperative therapy before surgery for LABC [3]. Neoadjuvant chemotherapy in the treatment of BC benefit for decrease the tumor size, eradicating micrometastases, and assessing the efficacy of chemotherapy [4]. The rapid proliferation of tumor cells in the neoplastic microenvironment is often due to its hypoxic environment. This condition contributes to the development of resistance to chemotherapy and radiation in addition to progressivity, proliferation, poor prognosis, and the possibility of metastasis [5]. In a hypoxic state, the oxygen concentration in cancer cells is less than 10 mmHg, while in healthy cells, the oxygen concentration ranges from 40 to 60 mmHg [6]. The consequences of hypoxia in BC include genomic changes that provide tumor cells with adaptive responses to reduced oxygen availability [7].
Hypoxia contributes to chemoresistance in BC by altering gene expression regulation in cancer cells. The master regulator of the hypoxia response is the hypoxia-inducible factor-1α (HIF-1α). Overexpression of the HIF-1α protein has been found in many types of neoplastic tumor microenvironments including BC, where high levels of HIF-1α greatly affect the growth rate and metastatic potential of BC [8].
Expression of the HIF-1α protein is an important predictive biomarker for pathological complete response (pCR) in LABC initially treated with NAC. Loss of the HIF-1α protein may indicate a favorable response to NAC and provides a good prognosis for patients with BC [9]. Currently, Ca15-3, the protein product of the MUC1 gene, is the most widely used serum marker in BC often used to assess therapeutic response and prognosis [9]. Determination of Ca15-3 levels in postoperative patients with BC is useful for the early detection of recurrence or metastasis [10]. Furthermore, the response evaluation criteria in solid tumors (RECIST) is an objective response parameter that can demonstrate the efficacy of NAC [11]. This study aimed to establish the relationship between HIF-1α and RECIST, compare Ca15-3 levels to RECIST values, as well as assess the relationship among these three variable factors in LABC.
Methods
Study design and setting
The observational study used the prospective cohort method was conducted at Dr. R.D. Kandou Hospital Manado. This study was approved by the ethics committee for research at Dr. R.D. Kandou Hospital Manado (license no. 048/EC/KEPK KANDOU/VI/2020) and has been registered with the ISRCTN registry no. ISRCTN63220723. This study is reported according to the Strengthening the Reporting of Cohort Studies in Surgery (STROCSS) guidelines [12].
Eligibility criteria
The inclusion criteria for this study were LABC with a histopathologically confirmed diagnosis of invasive ductal carcinoma; NAC regimen of cisplatin (80 mg/BSA) and gemcitabine (1000 mg/BSA) in 2 cycles; Karnofsky Performance Status Scale >70
Response criteria
Lesion measurement
All measurements are recorded in matrix notation using a clinical caliper. All baseline measurements were taken at the start of therapy or within 4 weeks of treatment. Target lesions were evaluated according to the RECIST criteria [11]: Complete Response (CR): Disappearance of all target lesions; Partial Response (PR): At least a 30% decrease in the sum of the longest diameter (LD) of the target lesion, with the baseline sum LD as the reference; Progressive Disease (PD): At least a 20% increase in the sum of the LD of the target lesion, with the smallest sum LD recorded since the treatment started or the appearance of one or more new lesions as the reference; Stable Disease (SD): Neither sufficient shrinkage to qualify for PR nor sufficient increase to qualify for PD, taking as a reference the smallest sum LD since the treatment started.
HIF-1α and Ca15-3 quantification
Blood samples (4–5 mL) were collected from a peripheral vein. The blood serum was separated in a plain vacuum tube, aliquoted, and stored at −20 °C until used in subsequent assays for HIF-1α and Ca15-3 in the Biomolecular and Immunology Laboratory at our institution. Ca15-3 levels were determined using an ELISA Kit Elecsys CA 15-3 II (Roche Diagnostics, cat. no. 07027001190, Indianapolis, USA) and HIF-1α activity was measured spectrophotometrically using the Human HIF-1α ELISA Kit (Merck Millipore, cat. no. RAB1057, Burlington, MA, USA) [13]. HIF-1α and Ca15-3 levels were measured before NAC and after receiving NAC according to the manufacturer’s instructions.
Statistical analysis
All statistical analyses were performed using R statistical software version 3.6.2 [14] Descriptive tabulations were performed according to the type of variable. Numeric scale variables are presented as means and standard deviations, or as medians and interquartile ranges (IQRs) if the Shapiro-Wilk’s test showed an abnormal distribution. Categorical variables are displayed in totals and proportions.
Results
A total of 11 patients with LABC who received NAC participated in this study (Table 1). Their mean age was 60 years old with a standard deviation of 10 years. The median body mass index (BMI) was 22, which showed that all subjects were within the normal range. Their mean hemoglobin concentration indicated anemia (11.6 ± 2.4 g/dL). Monocyte levels were average, but the median lymphocyte and quartile values (25%; IQR 21%–31%) were somewhat suggestive of lymphocytopenia.
Patient characteristics
Note: BMI, body mass index; SD, standard deviation; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Patient characteristics
Note: BMI, body mass index; SD, standard deviation; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Table 2 summarizes the RECIST, Ca15-3, and HIF-1α levels for the patients pre- and post-chemotherapy. Overall, these results indicated a decrease in both HIF-1α scores, Ca15-3 levels, and RECIST after NAC. For HIF-1α, the mean was reduced from 269.8 pg/mL before chemotherapy to 231.8 pg/mL with a mean reduction of 38.0 pg/mL (Fig. 1) per patient. Ca15-3 levels also fell by about 2.1 U/mL from the initial pre-chemotherapy 21.8 U/mL (Fig. 2). The patients’ target lesion diameter decreased by 30.3 mm on average according to the RECIST scores (Fig. 3).
RECIST, Ca15-3, and HIF-1α levels before and after chemotherapy
Note: RECIST: response evaluation criteria in solid tumors; HIF-1α, hypoxia-inducible factor-1α; SD, standard deviation.
Table 3 shows the relationship between the changes in HIF-1α, Ca15-3, and RECIST according to the existing data. Among them, only changes in RECIST and Ca15-3 correlated even though the coefficient of the relationship was very small: each unit change in RECIST was associated with a 0.3-unit reduction in Ca15-3 levels (95% CI −0.05; −0.01 and
Linear regression model of the change in RECIST, Ca15-3, and HIF-1α levels
Note: RECIST: response evaluation criteria in solid tumors; CI, confidence interval; HIF-1α, hypoxia-inducible factor-1α.
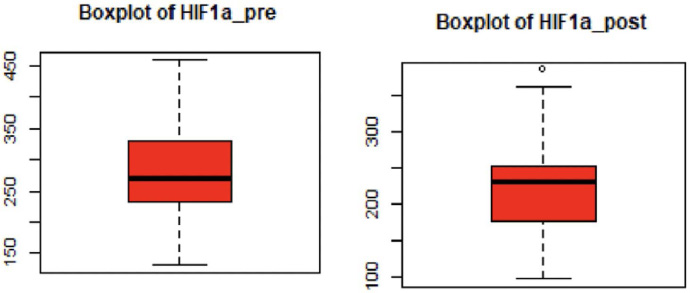
Changes in HIF-1α before and after the second cycle of chemotherapy.
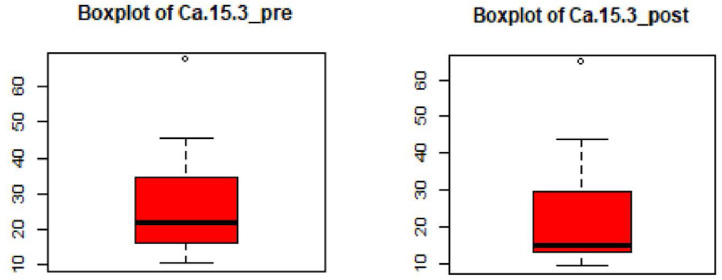
Changes in Ca15-3 before and after the second cycle of chemotherapy.
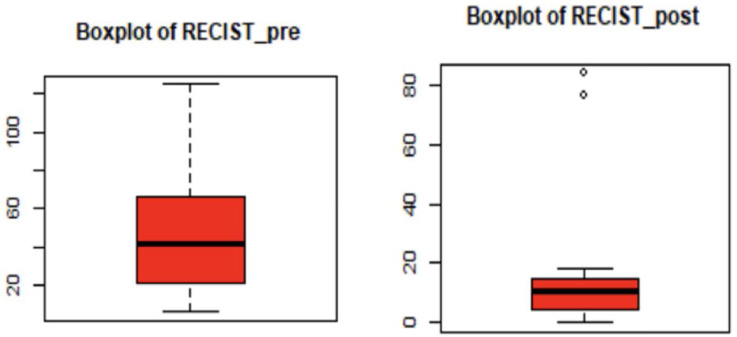
Change in tumor size according to RECIST before and after the second cycle of chemotherapy.
The results indicated a decrease in both HIF 1α, Ca15-3, and RECIST values after chemotherapy among all 11 women (Table 2). Hypoxia is common in most malignant diseases and is associated with tumor biology and the development of chemotherapy-resistant tumors [9]. Low-oxygen conditions affect the function of mitochondria and cells; as such, in order to survive, mitochondria must functionally adapt to low oxygen pressure to maintain cellular bioenergetics [15]. Therefore, hypoxic conditions can cause genome changes that allow tumor cells to survive despite the poor nutrition in the tumor microenvironment [16]. Hypoxia occurs due to the presence of necrotic areas and the blockage and compression of the blood vessels that innervate the tumor [6]. Tumor cells in these hypoxic regions begin to adjust to low oxygen pressure conditions by activating multiple survival pathways.
Most invasive BCs overexpress HIF-1α, which is associated with a malignant prognosis. HIF-1α in patients with LABC treated with NAC is significantly higher compared with operable primary BC [9]. HIF-1α expression in patients with BC decreases significantly after NAC [15]. In our study, the mean value of HIF-1α decreased after chemotherapy (
Serum Ca15-3 (mucin 1) is overexpressed in patients with BC > 90%. Ca15-3 promotes tumor invasion and metastasis by activating protein kinase signaling pathways stimulated by mitogens and downregulating E-cadherin. Ca15-3 levels are generally elevated in advanced-stage BC, especially stage III and IV [17]. Ca15-3 as a tumor marker has low sensitivity and specificity in the early stages of the disease, which increases as the disease progresses. Elevated levels of Ca15-3 predict a poor prognosis with an increased risk of metastasis [18]. In this study, there was a decrease in the level of Ca15-3 after NAC (
Determining NAC response could predict patient survival rates and monitor chemotherapy response. The reduction in tumor size (objective response) according to RECIST is an important target in evaluating chemotherapy response [20]. The data obtained in this study indicated that a significant reduction in tumor mass determined chemotherapy response. There was a mean decrease in the target lesion of 30.3 mm (
Table 3 shows the relationship between changes in HIF-1α, Ca15-3 levels, and RECIST. There was a weak correlation between RECIST and Ca15-3: Each unit of change in RECIST was associated with a decrease in Ca15-3 levels by 0.03 units (95% CI −0.05; −0.01 and
A study by Chaika et al. described an interesting relationship between Ca15-3 and HIF-1α cross-regulation modulated by MUC1 in cancer cells. They demonstrated that overexpression of MUC1 maintains high levels of the protein HIF-1α in a hypoxic state [21]. In our study, the results showed a decrease in both HIF-1 α and Ca15-3 after NAC, but these were not statistically significant (
Overall, the study results indicated a decrease in HIF-1α, Ca15-3, and RECIST values, however, these were not statistically significant. This is likely due to the small sample size, which should be addressed in future studies.
Conclusions
There was a decrease in HIF-1α, followed by a decrease in Ca15-3 and RECIST in response to chemotherapy. There was a correlation between Ca15-3 and RECIST values; however, we found no statistically significant correlation between HIF-1 α and Ca15-3 and chemotherapy response. Further research is needed, including studies with larger samples sizes.
Footnotes
6.
BC: Breast cancer; NAC: neoadjuvant chemotherapy; HIF-1α: hypoxia-inducible factor-1α; RECIST: response evaluation criteria in solid tumors; LABC: locally advanced breast cancer; pCR: pathological complete response; CR: Complete Response; PR: Partial Response; PD: Progressive Disease; SD: Stable Disease; STROCSS: Strengthening the Reporting of Cohort Studies in Surgery; mg: milligram; BSA: Body surface area; mL: milliliter; IQRs: interquartile ranges; BMI: body mass index; g: gram; dL: deciliter; pg: picogram; U: units; CI: confidence interval.
