Abstract
BACKGROUND:
Approximately 70–80% of breast cancer express ER-alpha and hormonal therapies, given significant improvements in patient survival. About 50% of ER-positive breast cancer patients with advanced disease insensitive to tamoxifen treatment when diagnosed. Recent studies have shown that ERα-36 is a crucial factor in the resistance of tamoxifen.
OBJECTIVE:
This study aims to determine the association between ERα-36 expression and de novo resistance of tamoxifen in patients with ER-positive breast cancer.
METHODS:
This study was an observational study using a cross-sectional method and was conducted at Wahidin Sudirohusodo Hospital and Unhas Hospital. ERα-36 protein expression was assessed using an immunohistochemistry assay. The association of ERα-36 expression and resistance of tamoxifen was tested with the Chi-square test.
RESULTS:
A total of 50 locally advanced breast cancer cases were included in this study, 22 cases (44%) had overexpression of ERα-36, and 28 cases (56%) had not, 24 cases (48%) had experience resistance to tamoxifen and 26 cases (52%) had not. There was a significant association between ERα-36 expressions and resistance of tamoxifen (p = 0.000).
CONCLUSIONS:
There was an association between the expression of ER-α36 with de novo resistance of tamoxifen in ER-positive breast cancer. ER-α36 could act as a worth considering biomarker for de novo resistance of tamoxifen in therapeutic strategies.
Introduction
Breast cancer is the most common cancer in women and is the second leading cause of cancer death in women around the world [1]. Brand et al. reported that recurrence/metastasis in patients with breast cancer still occurred despite optimal therapy. The failure rate for breast cancer therapy ranges from 20–55%. One of the causes of therapy failure is resistance to systemic therapy [2].
Hormonal therapy is given to breast cancer with positive estrogen and progesterone hormone receptors. Breast cancer has estrogen receptor (ERα/ERα66) positive in about 70–80% of cases at diagnosis. Estrogen receptors have a major role in the development of breast cancer [3]. Impaired ER function is the main therapeutic strategy in targeting cancer with hormone receptor-positive. Selective estrogen receptor modulator (SERMs), such as tamoxifen, can bind to estrogen receptors and block interactions between estrogen and estrogen receptors, thereby inhibiting estrogen receptor target genes and reducing tumor growth [4].
Tamoxifen has been the primary endocrine therapy for early and advanced breast cancer for nearly three decades [5]. Clinical research has shown that continuous therapy with tamoxifen for more than 10 years can effectively increase patient survival and reduce the recurrence of ER-positive breast cancer but found that about 40–50% of patients develop de novo resistance of tamoxifen (primary) and acquired resistance of tamoxifen (acquired/secondary) in 30–40% [5,6]. Resistance of tamoxifen occurs during breast cancer treatment in 1–2 years [7,8]. The recommended course of tamoxifen for prevention is 5–10 years; however, many adverse effects from hot flushes, the resistance of tamoxifen until endometrial cancer [8–10].
The alpha estrogen receptor has several isoforms. One of the ERα isoforms is ERα36. ERα36 is abundant in cell membranes and cytoplasm, which regulates the signaling pathway of non-genomic estrogens, resistant to antiestrogens [10–12].
These findings increase the possibility that ER-α36 may be involved in de novo resistance of tamoxifen in breast cancer [13,14]. Here, we determine the association between ER-α36 expression and de novo resistance of tamoxifen in patients with ER-positive breast cancer.
Methods
Approximately 50 consecutive patients were treated at Wahidin Sudirohusodo Hospital and Unhas Hospital, Makassar, Indonesia, from January 2014 to June 2020. All patients were ER-positive primary breast cancer with invasive ductal carcinoma based on histopathological results, locally advanced stage, and received neoadjuvant chemotherapy with TAC regimen (docetaxel, doxorubicin, and cyclophosphamide), modified radical mastectomy, and adjuvant tamoxifen treatment (20 mg/day). The follow-up data were available for all patients, with a length follow-up of 24 months.
Immunohistochemistry assay
Tissue samples were deparaffinized by immersing them in xylol 3 times each for 5 minutes. Soak tissue samples in ethanol for 5 minutes. Soak in 90%, 80%, and 70% alcohol for 5 minutes, then wash the tissue samples with running water. Put the tissue samples in H2O2 solution in cold methanol and leave it for 15 minutes. Soak with distilled water for 5 minutes. Soak in the target retrieval solution and heat it in the microwave for 30 minutes. After that, leave it for 30 minutes, then wash with distilled water. Wash in phosphate-buffered saline (PBS). Give a barrier around the preparation with a pap pen—drip with peroxide block for 15 minutes. Wash with PBS for 5 minutes 2 times. Drip the superblock for 5 minutes. Wash with PBS for 5 minutes 2 times. Drips the primary antibody (a polyclonal anti-ER-α36 antibody by Bioryt, USA; dilution 1:50). Keep overnight at 4 °C in a humidified chamber. The sections were incubated with the second antibody (horseradish peroxidase-conjugated goat antirabbit immunoglobin; 1:100) for 45 minutes. Wash with PBS for 5 minutes 2 times. Drop the DAB substrate mixed with chromogen diaminobenzidine (DAB). Wash in distilled water. Soak with haematoxylin for 2 minutes. Rinse with running water until it is clear. Perform dehydration process by soaking tissue sample in 70% alcohol, 80%, 90% for 5 minutes, dry, cover, give ethylene. Examine with a microscope.
Characteristics of patients (n = 50)
Characteristics of patients (n = 50)
Abbreviations: ER-α36: estrogen receptor-α36, PR: progesterone receptor, HER-2: human epidermal growth factor receptor 2.
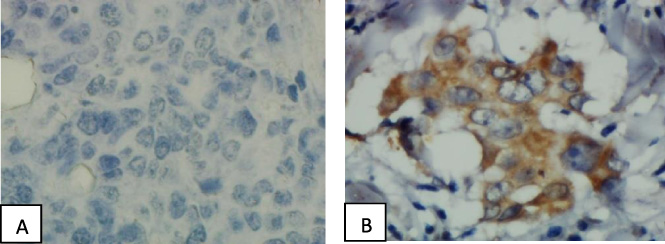
Immunohistochemical staining for ER-α36 showed (A) negative and (B) positive.
The staining intensity in the cytoplasm and the membrane cell was evaluated. Scoring for ER-α36 staining was graded as follows: no staining or staining viewed in less than 10% of tumor cells was rendered a score 0; weak/scantily visible staining detected in ≥10% of tumor cells was given a score 1+; an intermediate or plenary solid staining observed in ≥10% of tumor cells was marked a score 2+ or 3+, respectively. Scores of 0 and 1+ were deemed negative, whilst 2+ and 3+ were deemed positive [5]. Human epidermal growth factor receptor 2 (HER-2), estrogen receptor, and progesterone receptor expression were determined using an immunohistochemistry assay using each specific antibody. The immunostained slides were evaluated by a pathologist who independently checked all slide in a blinded way.
The association between expression of ER-α36, expression of HER2, and adjuvant tamoxifen treatment was prescribed using the Chi-square test. Statistical tests were two-sided, and p values < 0.05 were deemed as statistically significant. The statistic was analyzed using SPSS 22.0 software.
Association between HER2 status with ERα-36 status
Association between HER2 status with ERα-36 status
The proportion of patients with ERα-36 status was found to be lower in positive HER-2 expression than in negative HER-2, which is 45.5% compared to 54.5%. Based on the results of statistical tests, the difference is not significant (p > 0.05).
Association HER2 status with resistance of tamoxifen
This research has been approved by the Health Study Ethical Committee, Medical Faculty of Hasanuddin University, with the registry number 204/UN4.6.4.5.31/PP36/2020.
Association between ERα-36 status with resistance of tamoxifen
Association between ERα-36 status with resistance of tamoxifen
During the study period from January 2014 to June 2020, breast cancer patients data were obtained at Wahidin Sudirohusodo Hospital, and Unhas Hospital Makassar, which met the criteria of the study were 50 patients. The median age was 47 years with a range of 31–79 years. Age group less than 50 years was 31 cases (62%) and more than equal to 50 years was 19 cases (38%). Tamoxifen duration was 24 months with a range of 4–62 months. Tamoxifen duration group less than 24 months was 24 cases (48%) and more than equal to 24 months was 26 cases (52%). Based on the results of the histopathological examination, found the highest proportion of grading was grade II (76%). We measured the expression of PR, HER2, and ERα36 in 50 human breast cancer tissues by IHC staining. This study showed 86% of patients denoted high levels of PR expression in their tumors, while the remaining 14% of tumors denoted a low level of expression. The number of patients with HER2 overexpression was 22 (44%), and 28 cases (56%) showed negative. Twenty-two cases (22/50, 44%) showed positive expression of ERα36 positive (ERα36+), and 28 cases (28/50, 56%) showed negative (Table 1).
The proportion of patients with resistance of tamoxifen was found to be similar either in positive HER-2 expression or in negative HER-2. Based on the results of statistical tests, the difference is not significant (p > 0.05).
The proportion of patients with the resistance of tamoxifen was found to be higher in positive ERα-36 expression than in negative ERα-36, which is 77.3% compared to 22.7%. Based on the results of statistical tests, the difference is significant (p = 0.000).
Discussion
Wang et al. have identified and cloned a 36 kDa ERα66 variant, ERα36. This variant is the product of a transcript initiated from an unidentified promoter located in the first intron of the ERα66 gene, suggesting that this expression exhibits a different transcription regulation than ERα66 [14]. ERα36 is widely present in cell membranes and cytoplasm, regulating non-nongenomic estrogen signaling pathways that are resistant to antiestrogens [16].
In this study, 22 patients (44%) were found with overexpression of ERα-36 and negative expression of 28 patients (56%). This is in line with a study conducted by Shi et al., which reported the percentage of ERα-36 overexpression of 39–46% [14]. Similarly, the study of Sun et al. reported a percentage of overexpression of ERα-36 of 39.5% and revealed that ERα-36 was an independent prognostic factor for predicting poor outcomes in patients [12].
Shi et al. reported that about 40% of breast cancers with ERα-66 positive also expressed ERα36 in their tumors, and this group of patients may benefit slightly from tamoxifen treatment compared with the group of patients with ER-a66-positive/ER-a36-negative tumors. In addition, overexpression of ERα36 is associated with poor prognosis and resistance to tamoxifen treatment [14].
In this study, there were 24 cases of resistance to tamoxifen (48%) and no resistance to tamoxifen in 26 cases (52%). The results of this study are in line with the study by Clarke et al. reported that about 40–50% of breast cancer with ER-positive had resistance to tamoxifen at the time of diagnosis. The mechanism of de novo resistance in ER-positive breast cancer is not known with certainty. Several studies have shown that 55–60% of women with positive estrogen receptors respond well to hormonal therapy [17]. On the other hand, 40% of breast cancer with positive estrogen receptor expression, which initially responded to tamoxifen therapy, became resistant to tamoxifen [18].
A retrospective study of 896 breast cancer cases revealed that about 40% of the total breast cancer cases were ERα-36; approximately 40% of breast cancer cases are ER-positive for ERα-66 and ERα-36 coexpression [14]. Breast cancer overexpressing ERα-36 is less beneficial with tamoxifen therapy than those with low ERα-36 expression [14]. Several studies concluded that the increased level of ERα-36 expression in breast cancer cells is one of the mechanisms underlying the de novo resistance of tamoxifen found in ER-positive breast cancer. ERα-36-mediated estrogen signaling is insensitive to antiestrogens such as tamoxifen [15].
The above studies are in line with this study which showed a significant relationship (p = 0.000) between ERα-36 expression and the incidence of resistance of tamoxifen in breast cancer patients with positive estrogen receptors, where the percentage of positive ERα-36 expression against the resistance of tamoxifen was 77.3% higher than those without resistance of tamoxifen at 22.7%. In comparison, negative ERα-36 expression on resistance of tamoxifen was 25%, lower than those without resistance of tamoxifen by 75%.
This is because ERα-36 mediates non-genomic signaling pathways, including the activation of PI3K/AKT and MAPK/ERK. Activation of ERα-36 increases the proliferation and increased invasion of breast cancer cells. Increased levels of ERα-36 in breast cancer tissue were associated with decreased responsiveness to tamoxifen in ERα-66 positive breast cancer patients [19,20].
Hence, ER-α36 is a worth considering biomarker for tumor behavior and predictive factor, being a novel indicator to select optimal candidates for therapeutic strategies [21].
Conclusion
Early recognition of patients with ER-positive breast cancer who may be experienced de novo resistance to tamoxifen is significant in a clinical setting. There is a strong relationship between ER-α36 expression with de novo resistance of tamoxifen in ER-positive breast cancer. ER-α36 could consider as a biomarker for the diagnostic, prognosis, and treatment of breast cancer.
