Abstract
BACKGROUND:
High body mass index (BMI) is regarded as a poor prognostic factor in breast cancer (BC). However, its association with pathological complete response (pCR) and survival after neoadjuvant chemotherapy (NAC) remains controversial.
OBJECTIVE:
We wanted to assess the prognostic impact of BMI in this setting.
METHODS:
Single-center, retrospective review of 314 BC patients undergoing NAC from 2010 to 2018. Patients were categorized as underweight/normal weight (UW/NW) (BMI < 25) or overweight/obese (OW/OB) (BMI ≥ 25). The relationship between BMI and other traditional clinical-pathological prognostic variables with the pCR rate was investigated using logistic regression analysis. The effect on event-free survival (EFS) and overall survival (OS) was estimated by the Cox proportional hazards regression analysis.
RESULTS:
One hundred and twenty-two patients were UW/NW while 192 were OW/OB. Multivariate analysis revealed that hormonal receptors negative, HER2 positive, and clinical tumor stage (cT) 1–2 were independent predictor factors for pCR. Multivariate analysis confirmed tumor grade G3 and lack of pCR as independent adverse prognostic factors for EFS, while factors associated with worse OS were cT3-4, hormone receptors negative, and lack of pCR. Non-significant differences in pCR, EFS, or OS were observed between the two baseline BMI categories.
CONCLUSIONS:
In our experience, BMI is not associated with pCR, EFS, or OS in BC patients receiving NAC. Achieving pCR is the most consistent factor associated with EFS and OS. Prospective and well-designed studies taking into account other important biological and anthropometric factors are needed to determine the exact role of BMI in this setting.
Keywords
Introduction
Preoperative or neoadjuvant chemotherapy (NAC) is commonly administered in both, locally advanced and early-stage patients, allowing for breast conservation in patients with large tumors who would otherwise require mastectomy and improving aesthetic results in some candidates for breast conservation [1,2]. Achievement of a pathological complete response (pCR) has been proposed as one of the most relevant endpoints in NAC studies [3,4]. pCR is an independent prognostic factor predicting better long-term outcomes in terms of disease-free survival (DFS), event-free survival (EFS), and overall survival (OS) in randomized trials comparing the same chemotherapy regimen administered either preoperatively or postoperatively [5–8]. The pCR rate and its influence on EFS and OS vary considerably among the different BC molecular subtypes, being more relevant for triple-negative (TN) and human epidermal growth factor 2 (HER2) positive tumors [9–12].
Obesity has been demonstrated to be an important factor related to risk and prognosis in many types of cancer, including BC [13–16]. The processes linked to obesity and poor prognosis in BC are multiple and include the presence of higher levels of estrogen, insulin and insulin-like factors, oxidative stress, and inflammatory markers [17,18]. Obese patients tend to have reduced physical activity, which leads to an increased risk of type 2 diabetes, metabolic syndrome, and cardiovascular disease, which also contributes to this obesity-related poor prognosis in BC patients [19,20].
Overweight and obese BC patients, defined as a body mass index (BMI) of 25.0 to 29.9 kg/m2 and of 30 kg/m2 or more respectively [21], are at a higher risk of relapse after surgery and show worse results in terms of response and survival [22–24]. However, conclusions about the association of BMI with pCR or survival in the setting of NAC are not conclusive enough, with several studies presenting contradictory results [25–37].
The objective of our work was to study the influence of BMI on the pCR and clinical outcomes of patients treated with NAC at our center.
Patients and methods
Study population
We performed a retrospective review of the medical records of all BC patients who received NAC from 2010 to 2018 at the Department of Medical Oncology, Hospital Universitario de Gran Canaria Dr. Negrin (Spain).
Patients with histology-confirmed, non-metastatic, unilateral BC were included in this study provided they were 18 years or older and received a minimum of two NAC cycles. Figure 1 depicts a flowchart of the steps involved in the patient selection process. The tumor-node-metastasis classification was re-coded according to the 8th edition of the American Joint Committee on Cancer staging manual of 2017. The positive or negative status of ER, PR, and HER2 was evaluated following the American Society of Clinical Oncology (ASCO)/College of American Pathologists’ clinical practice guidelines [38,39]. BMI was calculated by dividing the weight in kilograms by the square of the height in meters before starting NAC [21].
Flow-chart showing the steps of patient selection.
All cases were discussed at a weekly multidisciplinary tumor board. Staging and treatment procedures were performed following local and international guidelines and recommendations across time. Following completion of NAC two categories of pathologic response were identified: pCR and non-pCR. The absence of invasive cancer on the resected breast specimen and regional lymph nodes was characterized as a pCR (i.e., ypT0/Tis ypN0) [40].
The study was approved by the Provincial Ethical Committee for Research with Medicines of Las Palmas and was carried out following the 1964 Declaration of Helsinki and its later amendments as well as Good Clinical Practice Guidelines. Waiver of informed consent was authorized.
Statistical methods
Patients were categorized into two groups according to the baseline BMI: underweight/normal weight (UW/NW) (BMI < 25 kg/m2) and overweight/obese (OW/OB) (BMI ≥ 25 kg/m2) [21]. Differences in patient characteristics between both groups were calculated using the Pearson’s Chi-square test for categorical variables, the Cochrane–Armitage trend test for ordinal variables, and the Mann-Whitney U test for continuous variables. Median follow-up was calculated using the reverse Kaplan–Meier method. EFS and OS curves were plotted using the Kaplan–Meier method, and comparisons between both BMI groups and pCR groups were made by the log-rank test. OS was calculated from the time of start NAC to the time of death or last follow-up visit. EFS was calculated from the time of the start of NAC to the time of disease progression, relapse (local or metastatic), or death of any cause; otherwise, to the time of the last follow-up. Logistic regression analysis was performed to evaluate the influence of the clinical and pathological prognostic factors in achieving a pCR. The associations of EFS and OS with the prognostic factors were estimated using the Cox proportional hazards regression analysis. Variables with p < 0.1 in the univariate analysis were entered into multivariate analysis. A two-tailed p < 0.05 was considered statistically significant. Statistical analyses were performed using MedCalc Statistical Software version 18.2.1 (MedCalc Software bvba, Ostend, Belgium).
Results
Patient characteristics
The main characteristics of the 314 patients included in the study are summarized in Table 1. The median BMI was 26.0 kg/cm2 (interquartile range (IQR), 23.5–29.3), range 18.3–52.0. There were 122 patients (38.1%) in the BMI < 25 group (UW/NW), and 192 patients (61.1%) in the BMI ≥ 25 group (OW/OB). Baseline characteristics of the patients were well-balanced except for a greater proportion of postmenopausal patients, older than 40 years, and with lobular histology in the OW/OB group.
Patient characteristics
Patient characteristics
BMI = body mass index; cN = clinical nodal; cT = clinical tumor; ER = estrogen receptor; HER2 = human epidermal growth factor receptor 2; Post = postmenopausal; PR = progesterone receptor; Pre = premenopausal.
Sequential anthracycline-based chemotherapy followed by a taxane was the most commonly used scheme in 259 patients (82.5%), being the FEC-P regimen (4 cycles of fluorouracil, epirubicin, and cyclophosphamide, followed by 8 weekly doses of paclitaxel) [41] administered to 222 patients (70.7%). Regarding anti-HER2 therapy, 90 out of 95 patients (94.7%) with HER2 positive tumors received trastuzumab alone or trastuzumab plus pertuzumab in addition to NAC. A combination of docetaxel and carboplatin was given to 51 patients (16.2%). Forty TN patients (12.7%) were enrolled in a phase II study receiving 6 cycles of docetaxel plus carboplatin [42]. This later regimen plus trastuzumab was also administered to 11 HER2-positive patients (3.5%), including 4 elderly and 2 patients with basal left ventricular dysfunction.
No reduction or capped doses were initially given to obese patients. An analysis of the final number of chemotherapy cycles administered over the initially planned number showed no differences between UW/NW and OW/OB patients (p = 0.296). One hundred fourteen UW/NW patients (93.4%) received ≥80% of the planned number of cycles, compared to 177 OW/OB patients (92.18%) (p = 0.677).
Factors associated with pCR
The pCR rate was 22.4%, and did not differ between patients with BMI < 25 and those with BMI ≥ 25 (19% versus 24.5%, p = 0.259). Nine variables were included in a logistic regression model to test their association with the achievement of a pCR (Table 2). In the univariate analysis, 5 variables showed a significant influence on pCR: clinical tumor stage (cT) 1–2, nuclear grade G3, hormone receptors (HR) negative, HER2 positive, and the TN subtype. BMI failed to reach the predefined cut-off of p < 0.1 at this setting, although we did enter it into the multivariate analysis locking for a possible significance as an independent prognostic factor. A secondary analysis was performed taking the BMI as a continuous variable showing again no significant relation with the pCR (p = 0.458). Multivariate analysis revealed that only HR negative (p < 0.0001), HER2 positive (p < 0.0001), and cT1-2 (p = 0.007) were independent predictor factors for pCR.
Univariate and multivariate logistic regression analysis for pathological complete response
Univariate and multivariate logistic regression analysis for pathological complete response
BMI = body mass index; CI = confidence interval; cT = clinical tumor; ER = estrogen receptor; HER2 = human epidermal growth factor receptor 2; OR = odds ratio; Post = postmenopausal; PR = progesterone receptor; Pre = premenopausal.
At a median follow-up of 59 months (IQR, 29.3–78.9), 66 EFS events (21%) have occurred and 34 patients (10.8%) have died. The median EFS was 123.2 months (95% confidence interval (CI), 101.1–123.2) and the median OS has not been reached.
Univariate Cox proportional hazards regression analysis revealed 5 variables associated with a worse EFS: cT3-4, nuclear grade G3, HER2 negative, TN subtype, and non-pCR after NAC. Multivariate analysis confirmed only tumor grade G3 (p = 0.012) and absence of pCR (p = 0.012) as independent prognostic factors associated with a worse EFS (Table 3). For OS, univariate analysis showed 6 factors associated with a worse outcome: postmenopausal status, cT3-4, nuclear grade G3, HR negative, TN subtype, and non-pCR. Of these, only cT3-4 (p = 0.003), HR negative (p = 0.003), and no-pCR (p = 0.037) were significant independent unfavorable prognostic factors for OS in the multivariate analysis (Table 4). Non-significant differences in EFS or OS were observed between UW/NW and OW/OB patients (Fig. 2).
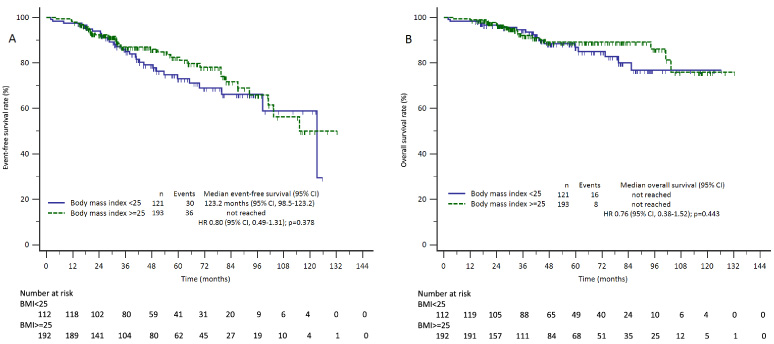
Kaplan–Meier curves of event-free survival (A) and overall survival (B) according to the body mass index (BMI) group.
Univariate and multivariate Cox proportional hazards regression analysis for event-free survival
BMI = body mass index; CI = confidence interval; cT = clinical tumor; ER = estrogen receptor; HER2 = human epidermal growth factor receptor 2; HR = hazard ratio; pCR = pathological complete response; Post = postmenopausal; PR = progesterone receptor; Pre = premenopausal.
Univariate and multivariate Cox proportional hazards regression analysis for overall survival
BMI = body mass index; CI = confidence interval; cT = clinical tumor; ER = estrogen receptor; HER2 = human epidermal growth factor receptor 2; HR = hazard ratio; pCR = pathological complete response; Post = postmenopausal; PR = progesterone receptor; Pre = premenopausal.
As shown in Figure 3, the pCR was associated with longer EFS and OS. Median EFS for patients without or with pCR was 115.2 months (95% CI, 98.5–123.2) versus not reached, HR 0.38 (95% CI, 0.21–0.70), p = 0.021. Kaplan–Meier estimates for OS also favored patients achieving pCR, even though no statistical significance was reached. Median OS was not reached in both groups, HR 0.26 (95% CI, 0.11–0.63), p = 0.051.
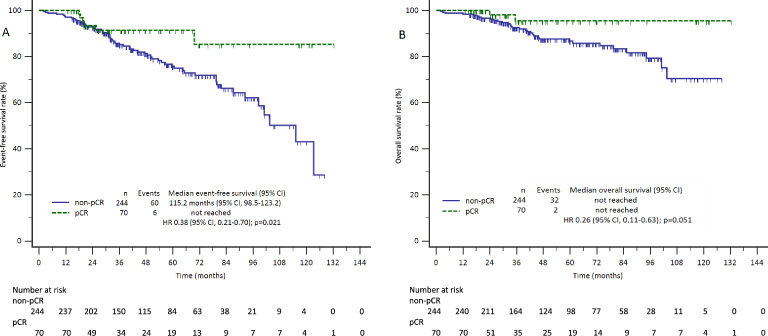
Kaplan–Meier curves of event-free survival (A) and overall survival (B) according to the achievement of a pathological complete response (pCR).
The role of obesity in cancer risk and prognosis has become a point of intense research in the last years. It is well recognized that obesity is linked with an increased incidence of at least 13 types of cancers, including postmenopausal BC [21]. There is also evidence about the negative effect of obesity on outcomes of patients diagnosed with BC. Results from a large meta-analysis of 82 clinical studies indicate that obesity is associated with poorer overall and BC survival in premenopausal and postmenopausal patients, been the impact greater in premenopausal women [22].
Despite these results, the level of evidence about the relationship of obesity with poorer outcomes in early BC is limited by the retrospective nature of most studies and a lack of full assessment of potential confounders. Obese patients tend to have larger tumors, with a higher proportion of nuclear grade 3 and TN phenotype, especially in younger patients [43,44]. Besides this, many oncologists routinely cap chemotherapy doses at a maximum body surface area of 2.0 or just reduce doses in OB/OW patients for fear of overdosing or toxicity [45–47]. Reduced doses of chemotherapy have been related to poorer outcomes in BC, and thus, may explain in part the adverse prognostic role of obesity in the adjuvant and neoadjuvant setting [27,33,48,49]. Another common weakness of these studies comes from the limitation of using BMI as a reliable measure of body fat, as it is a simple surrogate marker of obesity. The BMI does not distinguish fat mass from lean mass or the body fat distribution pattern. For example, higher visceral fat is associated with worse outcomes of BC after NAC, while sarcopenia (low muscle mass) has been associated with higher pCR after NAC in normal BMI patients [30,50]. Unfortunately, determining body composition and fat distribution requires complicated methods that are not readily available in clinical practice.
Regarding the role of obesity in the context of NAC, there are even more conflicting results. While some studies have found that BMI is an adverse prognostic factor for pCR and/or survival [25,26,30–34], others yielded negative results [27–29,35–37].
In a study with 1,169 BC patients receiving NAC from the M.D. Anderson Cancer Center, no significant differences in pCR between OB and UW/NW patients were observed, the progression-free survival (PFS) was not associated with OW or OB status, but OB patients experienced shorter OS than the other two BMI groups [25] In subsequent work, investigators from the same institution evaluated the role of sarcopenia and BMI comparing 67 patients achieving pCR with 62 matched controls that did not have pCR after NAC. In the multivariate model, only the negative HR was a significant predictor of pCR, and the PFS was significantly longer in the normal BMI group although it did not influence the OS [30]. Three reports from China [26], Mexico [31], and Turkey [32] concluded that a BMI ≥ 25 was associated with either worse pCR [26], shorter OS [31], or both pCR and OS [32] respectively. Results from a pooled analysis of eight prospective neoadjuvant BC trials from Germany with 8,872 patients showed that increasing BMI results in decreasing pCR and had an independent negative impact on DFS and OS, especially in patients with luminal-like tumors [34].
On the other hand, results from a retrospective evaluation of 110 BC patients younger than 35 years concluded that BMI was not correlated with pCR or survival [35]. A report from Korea involving 438 BC patients (4.3% OB) failed to find differences in pCR, DFS, or OS between NW, OW, and OB groups [37]. Similarly, the BMI was not associated with survival in two studies of NAC on TN BC patients conducted in Japan and at the Roswell Park Cancer Institute [28,36]. The importance of maintaining the dose intensity of NAC is highlighted in an Austrian study, where 120 BC patients were included only if they received full or uncapped doses of NAC. In contrast to previously described studies, the authors found that OB patients achieved better pCR rates and PFS than non-obese patients [27]. Finally, researchers from Germany present their results of a cohort of 324 BC patients undergoing NAC and concluded that there was no association between BMI and pCR or survival. In the same paper, the authors carried out a meta-analysis, and again found no association between BMI and pCR or survival outcome [29].
Several discrepancies in the design of these studies arise when interpreting their results. While some of them were restricted to younger patients or limited to a race subset, others focused on locally-advanced BC or specifically included only TN tumors. Also, different categories of BMI are employed and comparisons are usually made between two or three groups (NW, OW, or OB). Similarly, different end-points are reported among studies (pCR, EFS, relapse-free survival, or OS), and the results are sometimes significant for one but not for all of them.
We found that BMI did not influence the pCR, EFS, or OS in our unselected patient population of 314 BC patients, categorized as UW/NW (BMI < 25) or OW/OB (BMI ≥ 25). Significant predictors of pCR in the multivariate analysis were HR negative, HER2 positive, and cT1-2 tumors. Looking at survival, nuclear grade 3 was significantly associated with worse EFS, while HR negative and cT3-4 tumors conferred poorer OS. The only significant prognostic factor connected to higher EFS and OS in the multivariate analysis was achieving a pCR. These findings are consistent with those of earlier investigations, as stated above.
Our results should be regarded with caution because they are susceptible to some limitations. First, the retrospective nature of the study did not allow us to control for the imbalance in the distribution of some variables between both BMI groups. Second, as mentioned above, BMI is not the best method to estimate body fat. Another drawback of our study and others published on this subject is the lack of information about the reduction in chemotherapy doses.
Conclusion
Based on our experience in NAC for BC, we were unable to prove that BMI is an independent predictor of pCR or a significant prognostic factor for survival. Prospective and well-designed studies with more rigorous information about nutritional parameters, body fat distribution, physical condition, and accomplishment of full-dose chemotherapy are warranted in order to better understand the exact role of obesity in the response and prognostic of BC patients in the neoadjuvant setting.
Footnotes
Conflict of interest
The authors declare that they have no conflict of interest.
