Abstract
The rate of metastasis to the central nervous system is high in human epidermal growth factor receptor 2 (HER2)-positive metastatic breast cancer patients. Metastatic cauda equina tumors are characterized by rapid progression of symptoms, thus signifying the requirement of their early treatment. However, these tumors are rarely reported, and their optimal treatment options have not been established yet. Here, we report a case study of a patient with HER2-positive breast cancer that metastasized to the cauda equina. The patient underwent urgent surgery to relieve the spinal cord compression. The pain in her back and lower limbs was greatly reduced. Unfortunately, her ability to walk did not improve sufficiently. Overall, surgical treatment may be a favorable option to improve a patient’s quality of life.
Keywords
Introduction
Metastasis of cancer cells to the brain, bone, and spinal column has been frequently observed in patients affected with malignant tumors outside their central nervous system (CNS) [1–3]. However, the occurrence of intramedullary spinal cord metastasis (ISCM) is rare, accounting for only 0.1–0.4% of all malignant tumors. In addition, metastasis to the cauda equina has been seldom reported. To the best of our knowledge, there are only four reports of breast cancer metastasizing to the cauda equina [4–7]. Most cauda equina tumors are benign and occur as primary lesions [4]. The progression of metastatic cauda equina tumors is faster than that of primary benign cauda equina tumors, indicating high malignancy of the former and signifying the importance of early treatment [4]. Surgery in such cases aims to decompress the functional neural tissue and histologically confirm the tumor [5]. Herein, we report the case of a patient who exhibited metastasis of human epidermal growth factor receptor 2 (HER2)-positive metastatic breast cancer (MBC) to the cauda equina.
Case presentation
A 64-year-old woman who had a mass in her left breast was diagnosed with stage IV breast cancer, T4N1M1 (pulmonary), at Saitama Medical Center, Japan. Histopathological examination of a needle biopsy from the breast tumor revealed invasive ductal carcinoma. Immunohistochemistry showed that the tumor was estrogen receptor (ER)-negative, progesterone receptor (PR)-negative, and HER2-positive, with a Ki-67 labeling index above 80%. Systemic treatment with chemotherapy and anti-HER2 therapy was initiated. Partial response was obtained with 4 cycles of docetaxel plus trastuzumab followed by 4 cycles of fluorouracil, epirubicin, and cyclophosphamide. To achieve a complete response, the treatment regimen was changed to 6 cycles of vinorelbine plus trastuzumab (2nd line) with stable disease. Nab-paclitaxel plus trastuzumab was administered as 3rd line treatment. However, severe peripheral neuropathy appeared in the 4th cycle and was subsequently changed to lapatinib plus capecitabine (4th line) for 9 cycles. Surgery was performed for local disease control since the pulmonary metastasis disappeared on computed tomography. Histopathological examination of the surgical specimens revealed residual invasive ductal carcinoma in the breast tumor and absence of lymph node metastases (n = 0∕4). After surgery, lapatinib plus capecitabine therapy was resumed. However, new metastatic diseases appeared in her right axillary lymph node, liver, and left adrenal gland 6 months after surgery. The patient was treated with pertuzumab, trastuzumab and docetaxel (5th line) for 6 cycles. However, since the disease continued to progress, trastuzumab-emtansine (T-DM1) (6th line) treatment was started. Three years and 7 months post-surgery, the patient started experiencing lower back pain. Gradually, she started suffering from pain in the left lower extremity, which was eventually accompanied by her inability to walk, leading to her urgent admission to the hospital. After hospitalization, these symptoms were temporarily relieved with rest and painkillers. On the sixth day at the hospital, the patient developed paralysis in her left lower extremity, and magnetic resonance imaging (MRI) of her lumbar spine revealed a 44-mm lesion in the major axis mass within the conus medullaris with leptomeningeal involvement of the cauda equina at the third lumbar spine vertebra (L3). A postcontrast T1-weighted subtraction image of the MRI scan showed a low-intensity lesion. No bone destruction was observed (Fig. 1A,B). On the seventh day at the hospital, surgery was performed because the mass was believed to be compressing the spinal cord. The border between the mass and cauda equina was unrecognizable. It was observed that non-adherence to the arachnoid or dura mater led to metastasis in the cauda equina (Fig. 2). Further histopathological examination of the mass revealed metastatic adenocarcinoma (ER-negative, PR-negative, and HER2-positive). The patient underwent local spinal radiotherapy (3 Gy/10 fractions, 30 Gy) to prevent local recurrence because of the possibility of remnant tumors. After the surgery, an MRI of the brain revealed a mass in the right cerebellum (Fig. 3), and tumor resection was performed. Histopathological examination of the mass revealed metastatic adenocarcinoma. After the right cerebellum tumor resection surgery, an MRI of the brain revealed fifteen masses in the cerebrum (Fig. 4), and the patient underwent whole-brain irradiation therapy (3 Gy/10 fractions, 30 Gy). Subsequently, she was treated with gemcitabine combined with pertuzumab and trastuzumab (7th line) with a progressive disease (PD) as well as eribulin combined with pertuzumab plus trastuzumab (8th line) with a PD. However, she passed away six months after cauda equina surgery.
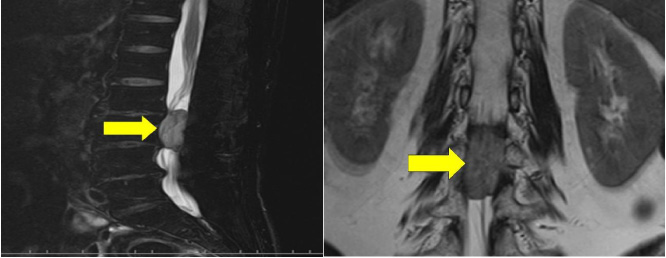
Magnetic resonance imaging (MRI) of the lumbar spine. (A) Sagittal T1-weighted and (B) coronal T1-weighted MRIs of the lumbar spine. Intramedullary mass at the level of L3 (arrows). No bone destruction is seen.
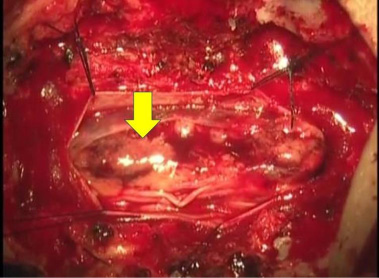
Intraoperative finding. The arrowhead indicates the tumor.
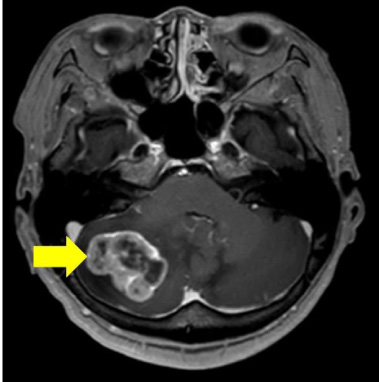
MRI of the brain after surgery of the cauda equina. The image shows a mass in the right cerebellum (arrow).
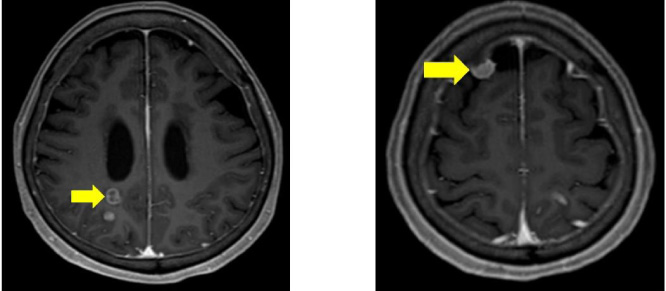
MRI of the brain after right cerebellar tumor resection surgery. The image shows masses in the cerebrum (arrows).
ISCM, particularly the metastasis to cauda equina, is rare. Metastatic cauda equina tumors do not exhibit typical initial symptoms and are difficult to diagnose. Additionally, the rapid disease progression requires early treatment [4]. In this case, the patient experienced sudden lower back pain that gradually evolved to pain in her lower left extremity. Eventually, she was diagnosed with ISCM when an MRI was performed due to lower-limb paralysis. Metastatic spinal cord compression is a serious oncological emergency as it potentially impacts the quality of life (QOL) of the patients. Furthermore, it was reported that no more than one-third of individuals with this condition survived beyond one year [2]. Therefore, aggressive treatment should always be considered for patients with an oncological emergency.
This patient complained of pain in her back and left lower extremity when she was urgently admitted to the hospital. After hospitalization, these symptoms were temporarily relieved with rest and painkillers. However, on the 6th day in the hospital, the patient suddenly developed paralysis in her left lower extremity. We recognized that she was suffering from spinal cord compression, and she underwent a spinal MRI. We decided to perform the surgery within 24 h of the onset of paralysis, as we believed that there was a possibility of recovering nerve functions. If this patient had undergone surgery at the time she developed back and leg pain instead of paralyzing symptoms, she could have avoided paralysis. Therefore, we consider that if a patient with recurrent breast cancer undergoing long-term chemotherapy complains of low back pain, spinal cord compression syndrome should be suspected, and an MRI scan should be performed immediately.
There is no consensus on the indications for surgery for ISCM in patients with MBC since it is a form of terminal stage cancer treatment. This may depend on the wishes of the patients and their family. A previous study reported that the prognosis in patients with ISCM is in the range of 6–9.4 months [8]. However, treatment of MBC with cauda equina syndrome does not aim to prolong the prognosis but to relieve pain and maintain the ability to walk as much as possible. Patchell et al. verified the efficacy of direct decompressive surgery in patients with metastatic spinal cord compression in a phase-III randomized trial. This trial compared radiotherapy alone (standard 30 Gy in 10 fractions) to decompressive surgery within 24 h of diagnosis followed by the same radiation (within two weeks of surgery) [9]. The study also demonstrated a significant advantage of surgery for every endpoint. For non-ambulatory patients, the combined treatment led to a significant rise in their chances of regaining the ability to walk (surgery and radiotherapy versus radiotherapy alone = 62% versus 19%, P = 0.01). For ambulatory patients, the combined treatment led to a significant rise in the chances of maintaining ambulation (surgery and radiotherapy vs radiotherapy alone = 94% versus 74%, P = 0.024). In addition, Gasser et al. reported that surgery should be carried out quickly while the patient is still ambulatory and should be abandoned when symptoms are associated with paraplegia for over 24 h or in case of the development of flaccid paraparesis [2]. In this case, the patient was operated upon to relieve the spinal cord compression urgently. Unfortunately, her ability to walk did not improve sufficiently. However, the patient and her family were quite satisfied with the surgery because the pain in her back and lower limbs was greatly reduced. In such cases, even if the prognosis is poor, surgical treatment may be a favorable option to improve QOL [10].
Summary of previous case reports in relation to breast cancer metastases to cauda equina
Summary of previous case reports in relation to breast cancer metastases to cauda equina
Abbreviations: HER2, human epidermal growth factor receptor 2; ER, estrogen receptor; PR, progesterone receptor; NR, not recorded; RTx, radiotherapy; CTx, chemotherapy.
Previous case studies of breast cancer that metastasized to the cauda equina are summarized in Table 1 [4–7]. All patients underwent surgery for metastatic cauda equina tumor, and three out of four patients underwent local spinal radiotherapy. All patients had complained of lower extremity pain or paralysis, which later improved after treatment. However, only one patient survived for approximately six months post-surgery. Three of the four patients (75%) exhibited lung metastasis, and one (25%) exhibited brain metastasis. Although five routes have been hypothesized for the dissemination of ISCM from outside the central nervous system [4], these previous case studies indicated that the main cause of cauda equina metastasis is the hematogenous route from lung metastasis [4–7]. The patient in this case also exhibited metastases to the brain. It was reported that metastatic intracranial tumors are concomitantly detected in 90% of patients with ISCM [4]. Therefore, if ISCM is detected, brain metastases must be examined. Two out of four patients had ER-positive and HER2-negative MBC. Generally, the treatment period for ER-positive MBC is longer than that for ER-negative MBC because chemotherapy and hormonal therapy are both valid for ER-positive MBC treatment. This is the first case report of HER2-positive breast cancer metastasis to the cauda equina. Although it has been reported that HER2-positive breast cancer has a poor prognosis, novel HER2-targeting agents including trastuzumab, lapatinib, pertuzumab, trastuzumab emtansine (T-DM1), and trastuzumab deruxtecan were recently shown to significantly improve progression-free survival in patients with HER2-positive MBC [3,11–15]. In this case, the patient received several combined HER2-targeted, long-term chemotherapy regimens. In addition, another study reported that overexpression of HER2 is an independent factor for the development of brain metastasis in patients with MBC [1]. It is possible that long-term treatments and aggressiveness of HER2-positive breast cancer may have contributed to brain metastasis and ISCM. Therefore, the brain and intramedullary spinal cord should be considered as potential metastatic sites, particularly in HER2-positive MBC.
Since trastuzumab is a monoclonal antibody with a large molecular weight, it does not penetrate the blood-brain barrier well. Thus, trastuzumab administration was considered ineffective for brain metastases [16]. However, Tamura et al. have shown that trastuzumab accumulates in the brain by PET using 64Cu-DOTA-trastuzumab [17]. In addition, lapatinib can penetrate the blood-brain barrier and may be effective for treating cancers that have metastasized to the brain due to its small size. The LANDSCAPE trial investigated the efficacy of a lapatinib and capecitabine combination regimen in breast cancer patients with brain metastases. The trial reported improved clinical outcomes, with 65.9% of patients exhibiting reduced brain tumor volume by 50% or more, a time to progression of 5.5 months, and a time to whole-brain irradiation of 8.3 months [18]. Although the clinical outcomes and prognostic factors of HER2-positive breast cancer patients with brain metastases are not well known due to the relatively small frequency of occurrence, a previous multicenter retrospective study reported that treatment with trastuzumab before brain metastases was not associated with survival duration after developing brain metastases [19]. However, this previous study also showed that the patients treated with both trastuzumab and lapatinib after developing metastasis survived significantly longer than patients treated with trastuzumab alone, lapatinib alone, and those not given HER2-targeting agents (p < 0.001). It suggests that treatment with both trastuzumab and lapatinib might improve prognosis, regardless of the use of trastuzumab before developing brain metastasis. An exploratory analysis in the EMILIA trial reported that T-DM1 may confer a survival advantage over the lapatinib plus capecitabine regimen in patients with treated central nerve system metastases [20]. In addition, another study reported that the efficacy of T-DM1 for leptomeningeal and brain metastases in patients with HER2-positive MBC [21]. Therefore, HER2-targeting agents in HER2-positive breast cancer may be efficacious for treating metastasis to CNS, and further studies are required to investigate their efficacy.
Although ISCM in HER2-positive MBC is rare, it is possible that its frequency will increase in the future due to the improved prognosis in this population. In case of sudden lower back pain or lower extremity paralysis in patients with HER2-positive MBC during long-term systemic therapy, ICSM should be considered and immediately examined and treated. Although the prognosis for ISCM is poor, surgical treatment should be considered if the patient has severe pain that prevents an acceptable QOL. Further development of treatments for ISCM in patients with HER2-positive MBC is warranted.
List of abbreviations
Central nervous system (CNS)
Intramedullary spinal cord metastasis (ISCM)
Human epidermal growth factor receptor type 2 (HER2)
Metastatic breast cancer (MBC)
Estrogen receptor (ER)
Progesterone receptor (PR)
Trastuzumab-emtansine (T-DM1)
Magnetic resonance imaging (MRI)
Progressive disease (PD)
Quality of life (QOL).
Footnotes
Ethics approval and informed consent
The study obtained approval from the ethics committee of the Saitama Medical Center.
Consent for publication
The patient has already passed away, and we were unable to contact her family. We cannot obtain informed consent for publication, and thus, we take the responsibility for anonymization of the patient.
Conflict of interest
The authors declare that they have no conflict of interest.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Funding
This study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors’ contributions
NO and HS conceived the study. NO and HS drafted the manuscript. NO and HS carried out the literature search. NO, HS, TS, YH, KK contributed to the clinical management of the patient. KS contributed to the pathological diagnosis of the patient. TS and YH performed the surgery on this patient. HS revised the manuscript critically and approved the modified text. All authors read and approved the final manuscript.
