Abstract
BACKGROUND AND OBJECTIVES:
MRI is generally performed to assess response to Neo-adjuvant systemic therapy (NAST) in breast cancer.
OBJECTIVE:
To assess role of MRI in determining the probability of having residual disease in patients undergoing NAST. We also evaluated synchronous cancers diagnosed following MRI.
METHODS:
This is a retrospective study which included all patients who had pre-and post-NAST MRI between June 2014 and December 2019. Data on demographics, tumour characteristics and pathology were collected and analysed. Pre- and post-MRI probability were calculated and depicted on nomograms.
RESULTS:
The study included 205 patients. Overall pre-MRI probability of having residual disease was 55% (OR:1.2). The post-MRI probability was 78% (95% CI 72–83%; OR:3.5) if MRI showed residual disease and 23% (95% CI 16–31%, OR:0.3) if imaging showed complete response. The absolute benefit was higher in TNBC and HR-HER2. Additional cancers were identified in 8.78% of patients.
CONCLUSION:
MRI is beneficial in evaluating response to NAST specifically in TNBC and HR-HER2 cancers. Pre- and post-MRI probabilities of residual disease depicted on nomograms are a useful tool for clinicians. MRI can potentially impact the treatment decisions by identification of synchronous cancers.
Introduction
Neoadjuvant systemic therapy (NAST) has become the standard of care in patients with triple negative, human epidermal growth factor-2 (HER-2) positive and locally advanced breast cancer [1]. Patients who achieve pathological complete response (pCR) after NAST have superior overall and disease free survival compared to patients who do not achieve pCR [2]. Accurate assessment of tumour response to NAST is vital in deciding further treatment. This can be undertaken by clinical and radiological methods. Clinical examination is subjective [3] and therefore is not a reliable tool for monitoring tumour response to NAST. Although mammography and ultrasound are excellent diagnostic tools, they have limitations in monitoring treatment response due to low sensitivity [4–6].
Magnetic resonance imaging (MRI) evaluates tumour response by detecting the changes in tumour metabolism and vascularity and is now routinely used in many centres for monitoring during NAST. Most of the reported studies assessing the role of MRI have used sensitivity, specificity, and positive and negative predictive values to demonstrate its efficacy [1,7–9]. However, these values are influenced by population characteristics and therefore not easily translated to individual patients. Calculating likelihood ratio and post-test probability is a more practical way of interpreting a diagnostic test and will have more relevance in clinical practice [10–12].
The primary objective of our study was to identify the likelihood ratio and pre and post MRI probability of having residual disease in patients undergoing NAST for breast cancer. We have also evaluated the additional investigations undertaken and synchronous cancers diagnosed following MRI.
Patients and methods
This is a retrospective study which included all patients who underwent NAST between January 2014 and December 2019. All patients had histologically confirmed invasive disease on core biopsy. Patients underwent mammograms and ultrasound of breast and staging investigations as part of diagnostic assessment. Inclusion criteria: All patients who had two MRI scans, one before and one after the penultimate cycle of NAST. Exclusion criteria: Patients with Recurrent breast cancer Bilateral breast cancer Carcinoma of unknown primary with axillary metastasis Metastatic breast cancer.
The study protocol was submitted to the local governance committee and approval was obtained (Approval number: CA37820).
The decision regarding the NAST regimen was made by the multidisciplinary team, which includes surgeons, oncologists, radiologists and pathologists. Standard regimen for NAST was anthracycline and taxane based chemotherapy. Anti HER-2 treatment was given based on local protocol. Response to NAST was evaluated by MRI and was classified into two groups: radiological complete response (rCR) or radiological residual disease (rRD). rCR was defined as non-enhancement or faint enhancement equal to the background of normal breast tissue at the cancer site. Any amount of enhanced area was reported as rRD. The concordance of the MRI findings was compared with the post-surgical histology.
Pathological complete response (pCR) was defined as the absence of residual invasive and in situ cancer on haematoxylin and eosin evaluation of the resected breast specimen following completion of NAST irrespective of lymphnode status (pT0 N any). Tumours were classified as hormone receptor (HR) positive if the immunohistochemical (IHC) staining showed stained tumour cell nuclei ≥1% for estrogen receptor (ER) or progesterone receptor (PR). HER2 status was determined by IHC and/or Fluorescence in situ hybridization (FISH) analysis. On IHC analysis, 3+ was considered positive, and 0 to 1+ was considered negative. If a 2+ result was obtained, the tissue was evaluated by FISH for HER 2 gene amplification. When the HER 2 to chromosome 17 centromere ratio was ≥2.0, or the mean HER 2 copy number was ≥6, the result was defined as positive. Tumours were divided in to various subgroups using IHC as surrogate markers. They are HR+HER2-, HR+HER2+, HR-HER2+ and triple negative breast cancer (TNBC).
Continuous variables such as age, tumour size on mammogram, ultrasound and MRI were expressed as mean, median, range and standard deviation. Student t-test and analysis of variance (ANOVA) were used to look for any statistical difference between the groups. Sensitivity, specificity, positive and negative likelihood ratios (LR), positive and negative predictive values and accuracy of MRI in detecting pathological complete response after NAST were calculated using true and false positive and negative values. Pre-MRI probability is the probability of residual disease which is calculated using the prevalence of residual disease in post-surgical histology based on pathological response. Post-MRI probability is the proportion of patients who truly have residual disease at the time of MRI scan and it is calculated based on LR. Post-MRI probabilities were calculated separately for rCR and rRD. Actual benefit of MRI was calculated by taking the difference between the pre and post-MRI probability of residual disease. Pre-MRI probability, negative and positive LR and post-MRI probability of residual disease was depicted on a Fagan’s nomogram. The Fagan’s nomogram is a graphical tool where pre-test probability is depicted on the left hand side and the LR in the middle. The nomograms were plotted for the whole cohort and each subgroup based on HR and HER-2 status.
Data regarding the tumour characteristics on various imaging modalities, further investigations following MRI and identification of additional cancers picked up was collected from the hospital database. Statistical analysis was performed using SPSS and medcalc.org software.
Results
During the study period, 266 women received NAST and 205 patients among them fulfilled the inclusion criteria. Median age was 53 years (range: 20–77). Invasive ductal carcinoma (86.34%) was the commonest type of cancer within this cohort. Majority of the patients (61.95%) underwent breast conserving surgery and 38.05% had mastectomy. The distribution of subgroups was: HR+ HER2-, 21 (10.24%); HR+HER2+, 36 (17.56%); HR- HER2+, 37 (18.05%) and TNBC, 111 (54.15%). Overall pCR rate after NAST was 45.36%. Highest pCR rate (62.16%) was seen in HER2 enriched group whereas lowest pCR (14.28%) was seen in HR+ HER2- group. The demographic and treatment details are mentioned in Table 1.
Predictability of treatment response on MRI
The median time interval between last MRI and surgery was 42 days. In the cohort, the sensitivity and specificity of MRI in predicting response to NAST were 82.10% (95% CI:73.78–88.74) and 72% (95% CI:61.78–80.86) respectively. The pre-MRI probability of having residual disease was 55% (95% CI 47.55–61.58; Odds ratio (OR) 1.2). Following NAST, if the MRI showed rRD, the positive likelihood ratio was 2.94 (95% CI 2.10–4.12) and the post MRI probability of having the residual disease increased to 78% (95% CI 72–83; OR 3.5). If the MRI showed rCR, the negative likelihood ratio was 0.25 (95% CI 0.16 to 0.38) and the post MRI probability of having residual disease decreased to 23% (95% CI 16–31; OR 0.3). Actual benefit of MRI in predicting the post-MRI probability was 23% and 32% in rRD and rCR respectively. Overall, the accuracy of MRI was 77.56% (95% CI 71.23–83.08) with a positive predictive value of 77.97% (95% CI 71.63–83.22) and negative predictive value of 77.01% (95% CI 68.83–83.56). Various parameters showing role of MRI in predicting response to NAST are described in Table 2.
Demographics and treatment details
Demographics and treatment details
HR: Hormone receptor, HER2: Human epidermal growth factor receptor-2, TNBC: Triple negative Breast Cancer, pCR: Pathological complete response.
Various parameters of MRI in predicting response to NAST
“*”: Hi means infinity. In this subgroup, MRI showed residual disease in 14 patients. All of them had residual disease in post-operative histology. HR: Hormone receptor, HER2: Human Epidermal Growth Factor 2, TNBC: Triple Negative Breast Cancer, CI: Confidence Interval, MRI: Magnetic resonance imaging, rRD: radiological residual disease, rCR: Radiological complete response.
In the sub-group analysis, positive likelihood ratio was highest (Hi) for HR+HER2− and the negative likelihood ratio was lowest (0.12) for HR–HER2+ cancers. The pre-MRI probability of having residual disease in various sub-groups was: HR+HER2−, 85.71%; HR+HER2+, 47.22%; HR-HER2+, 37.14% and TNBC, 56.76%. Post-MRI probability of residual disease in various sub-groups ranged from 60–100% in patients who had rRD. Highest post-test probability was seen in HR+HER2− (100%) and it was lowest in HR-HER2+ (60%). Post-MRI probability of residual disease in rCR ranged from 7% in HR-HER2+ to 57% in HR+HER2− subgroup. The actual benefit of MRI was highest in TNBC (26.24 in rRD and 34.76 in rCR) and HR-HER2+ (22.86 in rRD and 30.14 in rCR) breast cancers (Table 3). The values of pre-MRI probability, likelihood ratios and post-MRI probability for the whole cohort and various subgroups are shown in Fig. 1.
Absolute benefit (Difference between pre- and post-MRI probability) of MRI
HR: Hormone receptor, HER2: Human Epidermal Growth Factor 2, TNBC: Triple Negative Breast Cancer, MRI: Magnetic resonance imaging, rRD: radiological residual disease, rCR: Radiological complete response.
Mean size of tumour on mammogram and ultrasound was 27 and 26 mm respectively (p value = 0.82). The mean tumour size on MRI was 38 mm and this was significantly higher compared to the mammography and ultrasound (p value < 0.05) (Table 4). The mean tumour size on MRI done after NAST was 13.39 mm and it correlated well with the post-surgery histology size which was 11.40 mm (p = 0.26).
Secondary outcomes
Overall MRI identified new lesions leading to second look ultrasound in 68 patients (33.17%) on the ipsilateral side and in 24 patients (11.70%) on the contralateral side. In total, ipsilateral biopsies were performed in 46 patients (22.4%) and 13 had contralateral biopsies after a second look ultrasound. Additional malignancies were confirmed in 18 patients (8.7%) on the ipsilateral side and in one patient (0.48%) on the contralateral side (Table 5).
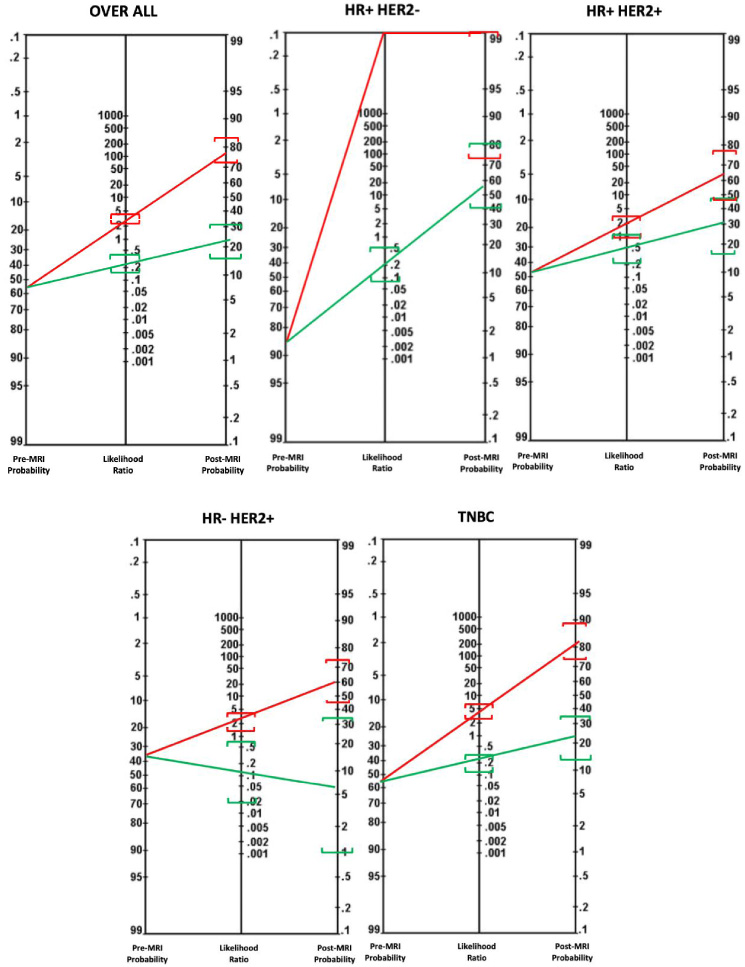
Nomograms showing the pertest probability, likelihood ratio and post-test probability of MRI with 95% Confidence intervals in overall cohort and various subgroups. Red line indicates positive likelihood ratio and post-MRI probability of having residual disease when MRI shows residual disease. Green line indicates negative likelihood ratio and post-MRI probability of having residual disease when there is radiological complete response. 95% CI values are shown in square brackets.
Dimensions of tumour on mammography, ultrasound and MRI
Extra investigations and new cancers picked up due to MRI
Our study has demonstrated that MRI is useful in predicting response to NAST specifically in HR-HER2+ and TNBC subgroups. The actual benefit was more than 20% when MRI showed rRD or rCR in these two subgroups (Table 3). A study by Loo et al. which included 188 patients showed similar results as of our study with MRI being useful in predicting response to NAST in TNBC (Pearson’s r = 0.605; p < 0.001) and HER2-positive disease (Pearson’s r = 0.426; p < 0.01) but is inaccurate in ER-positive/HER2-negative breast cancer (Pearson’s r = 0.074; p = 0.458) [13]. On the contrary, the contribution of MRI in assessing NAST response in HR+ and HR+HER2+ cancers is minimal. This is supported by a study by Kuzucan et al. who reported on the effect of hormonal receptor and Ki-67 in HER2- breast cancers and demonstrated that the accuracy of MRI was worse in HR+ and low proliferative tumours [14].
Showing various studies evaluating the sensitivity and specificity of MRI in evaluating response to NAST
Showing various studies evaluating the sensitivity and specificity of MRI in evaluating response to NAST
PPV: Positive predictive value, NPV: Negative predictive value, pCR: Pathological complete response, NAST: Neo-adjuvant systemic therapy, NA: Not available, “*”-Calculated rates based on pCR.
The use of MRI in evaluation of response to NAST has been extensively reported in literature [8,9,15–20] (Table 6). Most studies have assessed sensitivity, specificity, PPV, NPV and accuracy of MRI in predicting response to NAST. In these studies, the sensitivity of MRI ranged from 32.2% to 92%, specificity from 50–97.6% and the accuracy varied from 72.4% to 78.6% [8,9,15–20]. There is limited information in literature regarding LR and pre- and post-MRI probability of residual disease after NAST. These parameters are more reliable to describe the diagnostic accuracy of MRI and are increasingly becoming popular as they are easy to interpret and can be depicted in a pictorial form such as a nomogram [11,21–23]. A systematic review has shown that visual aids improve the understanding of post-test probability amongst health care professionals [24]. Therefore, we have depicted the pre-MRI probability, LR and post-MRI probability for each subgroup based on tumour biology on Fagan’s nomogram which is a user-friendly interface for clinicians to interpret and communicate the likelihood of residual disease to their patients. The clinician can use this information to discuss the prospects of postoperative radiotherapy and implications on surgery including breast reconstruction. The only study which reports on the use of a nomogram is the meta-analysis from Cheng et al. which showed slightly different results. In this study, the pre-MRI probability was 25% and increased to 62% with a positive LR of 5 when the MRI showed residual disease. It reduced to 7% with negative LR of 0.24 when MRI showed complete response [25]. However, there was no subgroup analysis based on hormone receptors and HER2 status. In our study, the overall positive LR was 2.94 and negative LR was 0.25. The difference in the outcomes could be explained by low pre-MRI probability as also reported by Cheng et al. [25].
The benefit of MRI in neoadjuvant setting was seen in all subgroups, but it was highest in TNBC in identifying residual disease and in HR-HER2+ subset when identifying complete response (Table 3). The contribution of post-NAST MRI is minimal in HR+HER2− cancers as the pre-MRI probability is high. Our results are consistent with studies by Hayashi et al. and G Werutsky et al. who showed high residual disease in Luminal/HER2 negative which was 86% and 79.6% respectively [26,27].
Various imaging modalities have been studied in assessing the accuracy of determining the tumour size. In our study, there was a significant difference between the preoperative tumour size measured by mammogram or ultrasound scan and MRI. The post NAST MRI size was comparable with the histopathological as previously reported in literature [28–30].
In our study, pre-operative MRI led to the identification of new cancers in 18 (8.29%) patients. Similar results have also been reported in COSMICE study with an identification rate of 6.2% new cancers [31]. However, a higher detection rate (16%) of additional cancers were reported by a metanalysis which included 19 studies with 2610 patients [32]. This is not entirely unexpected, considering the aggressive nature of cancers in our study cohort.
The potential limitation of our study is its retrospective nature with relatively small numbers in each sub group. In addition, owing to the study design, the results do not demonstrate the superiority of parameters such as likelihood ratios over conventional ones like sensitivity and specificity. Further prospective well-designed studies with higher numbers in each sub group would strengthen the validity of the nomogram in predicting response to NAST.
MRI is a useful investigation in evaluating response to NAST in TNBC and HR-HER2+ cancers. Pre- and post-MRI probabilities of residual disease depicted on nomograms are a useful tool for clinicians when counselling patients. MRI in preoperative setting can potentially impact the treatment decisions by identification of synchronous cancers.
Footnotes
Acknowledgements
None
Conflict of interest
None
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Financial disclosures
None
