Abstract
BACKGROUND:
Despite the dominance of implant-based breast reconstruction after mastectomy, during recent years, it has been correlated to some complications. The aim of this study is to present the Greek experience about management of implant infections after breast reconstructions and to investigate the relationship between possible risk factors and breast pocket fluid cultures.
METHODS:
In total, 260 patients underwent implant-based breast reconstruction due to breast cancer in our center from 2016 until 2020. 46 patients, that underwent implant or expander replacement after breast reconstruction due to mastectomy were included in the present study.
RESULTS:
260 patients underwent breast reconstruction in our center and in 46 (18%) of them an implant replacement was required. 21 patients (8%) presented clinically with an implant infection, but 12 of them (5%) had positive cultures from the breast pocket fluid. On the contrary, 25 patients (10%) presented no clinical signs of implant infection, but 5 of them (2%) had a positive culture. In addition, we demonstrated a correlation between implant infection and positive cultures (p = 0.009), along with an association between chemotherapy before implant placements and negative cultures (p = 0.035). Finally, the most common pathogen was Staphylococcus epidermidis (29%), followed by Escherichia coli (24%) and Staphylococcus aureus (18%).
CONCLUSION:
Implant infection still remains a very serious complication after breast reconstruction surgery. The establishment of a therapeutic protocol, with specific antimicrobial and surgical targets seems as an effective strategy against implant infections.
Introduction
Approximately 1.7 million women are diagnosed with breast cancer every year. Mastectomy is required in up to 40% of these, and a breast reconstruction is offered to improve quality of life [1]. Almost 72% of all breast reconstructions is performed in the immediate setting, while 82% of all reconstructions is based on implants [2]. A common reconstruction method is a two-stage procedure involving initial placement of a tissue expander in a subpectoral pocket, followed by sequential outpatient expansions with saline until the desired volume is achieved, and replacement of the expander with a fixed-volume implant at a later stage.
Despite the increased level of satisfaction and quality of life of patients, this technique is accompanied by several complications such as cellulitis, hematomas or expander exposure [3]. Risk factors for postoperative complications after implant breast reconstruction are obesity, smoking, patient age, radiation therapy and implant volume [4–9]. Especially complications that require operative management seem to significantly increase the cost of breast reconstruction, prolong the reconstructive period, and decrease patient satisfaction levels [10–12].
A very serious postoperative complication after implant based breast reconstruction is implant infection, which is not rare, as its rates range from 5.8% to 28% [13,14], compared with 5% for autologous reconstruction and 0.1%–1.5% for breast augmentation [15,16]. Except from the devastating consequences of breast implant infection on the psychological well-being of the patients, it raises the cost over approximately 4000$ per patient, as it causes prolonged antibiotic treatment, need for hospital readmission and delay in oncologic treatment [17]. Less severe cases can be treated with outpatient oral antibiotics, while more severe cases necessitate inpatient admission and intravenous antibiotics and often result in a failure of reconstruction and implant loss. Nevertheless, attempts for reconstructive salvage, defined as the ability to keep an implant after infection, have also become more popular in recent years [18].
Aim of our study is to present the Greek experience regarding the findings and the management of breast implant infections after mastectomy and immediate or delayed breast reconstruction. In addition, we investigated any possible relationship between demographic and clinical data of the patients, that underwent implant based reconstruction and the breast fluid cultures that were obtained during the expander replacement process.
Material and methods
In the present study, we included all consecutive female patients that underwent implant replacement after breast reconstruction due to mastectomy at the 1st Propaedeutic Department of Surgery of the Medical School of Athens in “Hippocratio” General Hospital, from December 2016 until February 2020. Patients that underwent implant reconstruction in combination with a tissue flap, such as latissimus dorsi flap, were excluded. Patients were prospectively enrolled in our study; thus, a written signed consent for data use in terms of clinical research was obtained from each patient. In addition, the study was performed according to the World Medical Association Declaration of Helsinki and ethical approval had been received by the institutional ethics committee. Our study was based on the evaluation of the clinical data of these patients, that were kept in record, according to our institution’s policy.
A possible implant infection was defined by the presence of criteria such as: fever, minimal local edema and inflammation; cellulitis, leukocytosis, systemic inflammation requiring intravenous or oral antibiotic treatment in the outpatient or inpatient setting; echographic evidence of inflammation or peri-prosthetic liquid accumulation; presence of purulent discharge and/or microorganism isolation [19]. The follow-up period was at least one-year in order to audit all possible postoperative complications.
All patients had skin or nipple-sparing mastectomy followed by immediate or delayed implant based breast reconstruction. Silicone implants or tissue expanders were placed under the pectoralis muscle (subpectoral) with the aid of a biological or synthetic mesh. Tissue expanders and permanent implants had the same textured surface and external envelope made by three overlapped layers of silicone rubber. According to our department antimicrobial protocol, the breast pocket was irrigated with povidone–iodine, the surgical field was draped with new sterile towels and all surgeons changed their gloves before implantation of expanders, implants or meshes. Expanders were positioned in a submuscular pocket after mastectomy and progressively inflated; permanent implants were positioned as a replacement of a previous expander after its inflation or during a contralateral augmentation mammoplasty. The antibiotic prophylaxis consisted of intravenous administration of Amoxicillin-Clavulanic acid (1 g every 12 h) and Gentamicin (80 mg every 12 h) for 2 days, followed by per os administration the same drugs for 5 more days. If a patient was allergic to beta-lactams, Clindamycin was used [20].
During the expander replacement or implant insertion process, cultures were obtained from the fluid inside the expander or the implant pocket. All meshes and biologic materials were briefly soaked in gentamycin solution before use [21]. In addition, the breast pocket was irrigated with the povidone–iodine solution. All members of the surgical team changed gloves and the surgical field was draped with new sterile towels [22]. Finally, minimal handling of implants was followed by subcutaneous placement of a closed suction drain, that was kept for approximately 5–7 days [23].
In case of an implant infection (Fig. 1), patients were inserted in our surgical department and initially administered with the empirical combination of Meropenem (500 mg every 8 h), Linezolid (2 mg every 12 h) and Metronidazole (500 mg every 8 h), followed by the appropriate de-escalation according to blood or breast pocket fluid cultures [24]. If no clinical improvement was observed after 5–7 days, the patients were guided to the operation room. During surgery, a capsular debridement up to capsulectomy was performed, depending on breast tissue thickness, following by an implant pocket irrigation with saline solution, povidone–iodine and hydrogen peroxide. A complete exchange of surgical instruments, towels and surgical coats was performed during the procedure before implant replacement, accompanied by patient disinfection. The new implant was then inserted in the breast pocket with minimal handling procedures and placement of a closed suction drain was performed. In case of a second infection after implant exchange, the implant infection protocol was followed only in the terns of antibiotic administration; a third operation using a prosthetic material (mesh or implant) was not performed in any case.
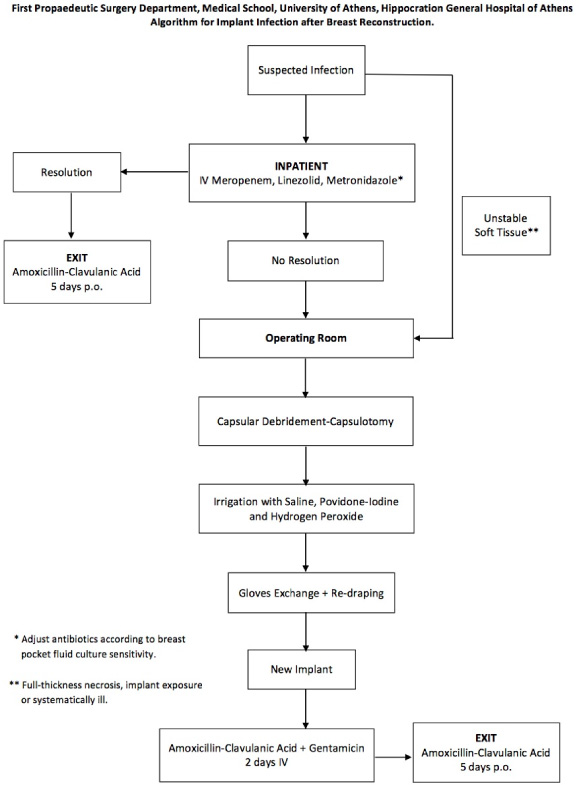
Implant infection after breast reconstruction algorithm.
Demographics including patient age, body mass index (BMI), active tobacco use, diabetes mellitus, history of chemotherapy before or after the implant placement and exposure to radiotherapy before the implant placement were assessed as possible risk factors for a positive culture. A smoker was defined as someone who had smoked in the 6 months leading up to surgery or in the 3-month period following surgery. However, all patients stopped smoking one month before and at least one month after implant implantation. Patients were considered as diabetics, if their glycated hemoglobin (HbA1C) was over 6.5%. Moreover, we evaluated the relationship between breast inflammation and positive breast pocket fluid cultures.
Statistical analyses were performed using SPSS version 25.0 software for Macintosh (SPSS Inc., Chicago, IL, USA). The non-parametric Pearson chi-square test of independence and the nonparametric Mann–Whitney U test were performed on categorical and continuous variables, respectively. The significance level was set at a value of p < 0.05.
During the study period, 260 patients underwent immediate or delayed breast reconstruction after mastectomy in our Breast unit. A biologic mesh had been used in only 4 patients (1.5%) and no implant replacement was required in none of them. Replacement of the expander or the implant was required in 46 patients (18%), and a pocket fluid culture was obtained. Among them, 21 patients presented clinically with an implant infection (8%). Twelve of them (5%) had positive cultures from the breast pocket fluid, while the rest 9 of them (3%) had negative culture.
On the other hand, 25 patients (10%) that underwent an expander or implant replacement after breast reconstruction presented with no clinical signs of implant infection. However, 5 of them (2%) had a positive culture that was obtained from the breast pocket during the replacement process, while the rest 20 patients (8%) had a negative culture (Fig. 2). A significant capsular contracture was observed in 13 patients (5%) and in the rest 12 patients (5%) the expander was replaced by an implant according to the two-stage breast reconstruction protocol of our unit.
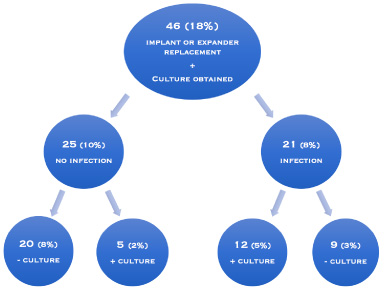
The patients, that underwent replacement of expander or implant after breast reconstruction, and were included in our study, divided according to the clinical manifestation of implant infection and the breast pocket fluid culture.
In total, positive cultures from the breast pocket fluid were found in 17 patients. The most common pathogen was Staphylococcus epidermidis, which was found in 5 patients (29%), followed by Escherichia coli, which was found in 4 patients (24%). Staphylococcus aureus was found in 3 patients (18%), Klebsiella pneumoniae and Pseudomonas aeruginosa were found in 2 patients (12%). In addition, Staphylococcus warneri, Enterococcus casseliflavus (vancomycin resistant), Enterobacter aerogenes, Pantoea species, Stenotrophomonas maltophila and Enterococcus faecalis were found in 1 patient (6%) (Fig. 3). Two or more pathogens were found in cultures from 5 patients (29%). On the other hand, the pathogens that were found in the cultures of the patients that underwent expander replacement without any clinical sign of infection were the following: Staphylococcus warneri, Klebsiella pneumoniae, Escherichia coli and Staphylococcus epidermidis.
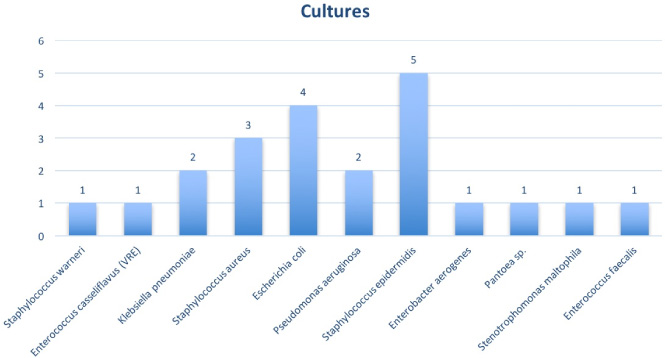
The bacterial species that were isolated in the breast pocket fluid cultures from the patients that underwent replacement of expander or implant after breast reconstruction due to mastectomy.
Mean age of the patients that were included in our study was 52 ± 10 years (range, 38–79 years), while their mean BMI was 28.8 ± 4.3 kg/m2 (range, 23.1–42.9 kg/m2). Patient age and BMI were not associated to a positive breast pocket fluid culture after implant or expander replacement (p = 0.227 and p = 0.331, respectively).
Nevertheless, a statistically significant relationship was outlined between a clinically suspected implant infection and a positive culture from the fluid of the breast pocket (p = 0.009). In addition, chemotherapy administered before implant placements was associated with negative cultures from the fluid of the breast pocket (p = 0.035) (Fig. 4). However, no statistically significant relationship was observed between fluid cultures and chemotherapy after implant placement (p = 0.606). In addition, preoperative radiotherapy was not correlated to positive breast pocket fluid cultures (p = 0.177). Finally, smoking and diabetes mellitus were not associated to positive breast pocket fluid cultures after implant or expander replacement (p = 0.760 and p = 0.106, respectively) (Table 1).
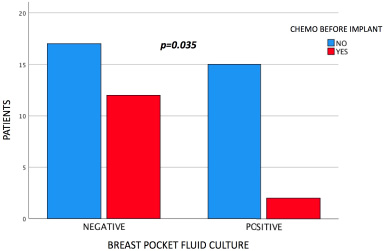
Negative breast pocket fluid cultures were associated with chemotherapy administration before implant placement.
Demographic data between positive and negative cultures after implant replacement
Demographic data among patients with positive and negative breast pocket fluid cultures after expander or implant replacement. Statistical significance was set at a level of p < 0.05. BMI, body mass index.
The present study describes our findings and therapeutic protocols for the management of implant infection after immediate or delayed breast reconstruction due to mastectomy. In addition, we outline that only half of the patients that underwent implant or expander replacement after breast reconstruction, presented clinical signs of implant or expander infection. Furthermore, only half of these patients had a positive breast pocket fluid culture. From the rest of the patients that presented no clinical signs of infection, only one fifth had positive breast pocket fluid pocket cultures.
We also demonstrated a correlation between implant infection and positive cultures, along with an association between chemotherapy before implant placements and negative cultures. Nevertheless, there was no association between positive breast pocket fluid cultures and patient age, BMI, chemotherapy after implant placement, preoperative radiotherapy, smoking and diabetes mellitus. Finally, the most common isolated pathogen from the breast pocket fluid was Staphylococcus epidermidis, followed by Escherichia coli and Staphylococcus aureus.
The establishment of a standardized therapeutic protocol for the management of implant infection after breast reconstruction due to mastectomy is essential for every breast unit. Implant infections has been reported to subside after the initial empirical antibiotic administration with vancomycin and extended spectrum penicillins at a rate of 80% [25,26]. In cases that implant infection remains, surgical intervention cannot not be avoided; explantation (40%) has been the most common procedure, followed by secondary implant insertion (20%) and operative salvage (20%) [27]. However, several preoperative, intraoperative and postoperative actions, that reduce the risk of postoperative implant infection after breast reconstruction have been described in the literature. Preoperative chlorhexidine patient wash and intranasal mupirocin administration, intraoperative use of intravenous cefazolin or clindamycin, chlorhexidine preparation, glove changing, re-draping, minimal touch handling, betadine, triple-antibiotic pocket irrigation and postoperative administration of a cephalosporin, along with drain removal after 20 days, are some of the previously mentioned actions [28,29].
Implant infection after breast reconstruction has been associated with several “modifiable” and “unmodifiable” risk factors. Firstly, Boustany et al. demonstrated that longer operative times, use of tissue expanders, drain use and elevated BMI have been associated to increased risk of implant infection [30]. Moreover, Kato et al. described that diabetes mellitus, repeated expander insertions, larger expander size (≥400 cc), postoperative hormone therapy before silicone implant surgery, preoperative chemotherapy and nipple-sparing mastectomy lead to elevated implant infection rates [31]. Furthermore, surgical site infections after implant based reconstruction following mastectomy have been correlated to raised BMI (OR, 1.1 per 1 BMI point increase; 95% CI, 1.0–1.2; p = 0.02), hypertension (OR, 6.5; 95% CI, 1.9–22.3; p = 0.002), neo-adjuvant chemotherapy (OR, 2.6; 95% CI, 1.0–6.3; p = 0.04), axillary lymph node dissection (OR, 7.1; 95% CI, 1.7–29.2; p = 0.006), seroma formation (OR, 15.34; 95% CI, 3.7–62.5; p = 0.0001) and wound healing complications (OR, 23.91; 95% CI, 6.1–93.4; p < 0.0001) [32]. On the other hand, Hanna et al. presented prolonged drain use as an independent risk factor for implant infection (OR, 3.3; p = 0.002) and indicated that earlier expansion was correlated with earlier post-operative drain removal (r = 0.3, p = 0.001), that avoided infections and improved outcomes [33].
The bacteriology of breast implant infections after reconstruction has remained stable over years and represents the source of pathogen contamination. Most commonly Gram-positive organisms, such as coagulase-negative staphylococci, Cutibacterium species, Staphylococcus aureus, and streptococci, gain access through deeper breast tissue during surgery. Gram-negative bacilli (especially Pseudomonas aeruginosa) were noted to be the second leading cause of microbiologically confirmed breast implant infections after S. aureus in a single referral center in France [33]. However, the contamination of the implant or saline or skin marking solution, and the possible hematogenous seeding of the implant, cause rare infections by nontuberculous mycobacteria, such as Mycobacterium fortuitum, gram-negative bacteria like Pseudomonas, and anaerobes [34–38]. Our study revealed a similar pathogen distribution from the cultures that were obtained from the breast pocket fluid during the implant or expander replacement process.
Implant infection rate after breast reconstruction is still significant and all implant infections are not microbiologically proven totally. This phenomenon of “sterile inflammation”, that was observed in our study as well, could be explained by local alterations in microvasculature due to surgery and altered tissue healing processes, that might be impaired by chemotherapy or foreign body (mesh) reaction [39]. The overall breast implant infection rate (8%) of our center is near the infection rate of the majority of breast units [40]. In addition, Franchelli et al. reported an 8% implant infection rate after breast reconstruction, with 71% of these infections microbiologically confirmed [41]. A recent systematic review demonstrated an overall implant infection rate that ranged from 0.9% to 21.6%, with a mean pooled infection rate of 5.4%, and the positive breast pocket fluid culture rate ranged from 45.9% to 100%, with a pooled positive culture rate of 74.5% [42]. On the other hand, a single-center study at the Mayo Clinic described that 9 of 27 (33%) breast implants that were removed not due to infection, but due to significant capsular contracture, had ≥20 CFU bacteria/10 mL sonicate fluid [43], similarly to the increased positive culture rate from non-infected implants of our study. Finally, the iBRA-study revealed a high overall implant infection rate (25%, 95% CI 23–27), that was higher than the National Quality Standards of the UK (<10%) [44].
Research has been guided towards “modifiable” and “unmodifiable” risk factors, that could decrease the possibility of an infection after breast reconstruction [45,46]. Our study contributes towards that direction, as it investigated the relationship between clinical infection, diabetes mellitus, smoking, radiotherapy, chemotherapy before or after implant insertion, age, BMI, and positive breast pocket fluid cultures. In addition, it highlighted that some implant infections were not microbiologically confirmed, while there was proven pathogen development in implants that were not infected. These facts might lead future studies to the search of unknown factors that may cause or protect against implant infection after breast reconstruction. Until now, adoption of a successful therapeutic protocol that consists of a pharmaceutical and a surgical plan still remains the only effective strategy against implant infections after breast reconstructions.
Conclusion
Breast implant infection after reconstruction due to breast cancer is a frequent and serious complication of breast surgery. Women’s psychology is furtherly burdened after breast surgery and sometimes their life may be in danger. Therefore, an implant infection protocol like the one suggested in our study, should be followed in every breast unit. In addition, risk factors such as BMI, previous chemotherapy, smoking etc., that are associated with breast implant infection should always be taken into account. Finally, bacteriology of implant infection and cultures are very important for targeted antibiotic administration and effective treatment.
Footnotes
Disclosure
The authors report no conflict of interest.
Source of funding
None to disclose for all authors.
