Abstract
BACKGROUND/OBJECTIVE:
The aim of this study was to identify the mammographic findings associated with malignancy in different age groups, taking into account breast composition (BC) and lesion size.
METHODS:
Preoperative mammograms of 1023 invasive ductal carcinomas were retrospectively evaluated. According to the American College of Radiology Breast Imaging Reporting and Data System, cancer mammographic findings were classified as mass, calcifications, architectural distortion and asymmetry, and breasts were assessed as non-dense (A or B BC) and dense (C or D BC). Patient cohort was subdivided into three age groups (group 1: <50 years of age; group 2: between 50 and 69; group 3: ≥70 years of age).
RESULTS:
Significant results of multinomial logistic regression were the association between mass and non-dense breast (p < 0.0001) and the association between mass and tumor size larger than 15 mm (p = 0.0049).
CONCLUSIONS:
Mass finding of invasive ductal breast carcinoma is associated with breast composition and tumor size.
Introduction
Breast cancer is the most common cancer in women, representing 30% of all malignant tumors, and it is the leading cause of cancer-related deaths for women [1]. It has two incidence peaks, the first at 45–49 years of age, and the second at 60–64 years of age, with a plateau after menopause. About a quarter of breast cancers occurs before age 50 [2,3].
It is known that younger women tend to have denser breast tissue than older women [4]. Moreover, it is known that increased breast density reduces mammography sensitivity [5]. However, few researchers investigated the relationship between breast cancer mammographic findings and patients’ age and breast composition [6–9]. Therefore, the aim of this study was to relate mammographic findings of invasive ductal carcinoma with women’s age (<50 years old, between 50 and 70 years old and >70 years old), breast composition and tumor size.
Materials and methods
Patient population
From January 2017 to December 2019, about 32,000 mammograms were performed in our institution, and 1716 invasive breast cancers were resected.
Inclusion criteria of this retrospective study consisted of surgical intervention and postoperative pathologic assessment performed in our institution.
Exclusion criteria consisted of: male patients (n. 17), personal history of breast cancer (n. 148), mammograms unavailability (n. 106) on our PACS (Picture Archiving and Communication System—Centricity GE PACS 4.0, General Electrics, USA), mammographically diagnosed cancer different from invasive ductal carcinoma (325), negative mammography (n. 97).
Therefore, 1023 lesions were included in the study and were divided into three groups according to patients’ age: under 50 years of age (group 1), between 50 and 69 (group 2), and aged 70 years or over (group 3).
Within the study cohort, 585 (57.2%) were asymptomatic women, while 438 (42.8%) showed breast symptoms such as palpable abnormality, pathologic nipple discharge, focal and noncyclical breast pain, focal skin thickening and/or retraction.
The study was approved by the institutional review board and ethics committee of our institution. Informed consent was obtained from all individual participants included in the study. All procedures were performed in accordance with the 1964 Helsinki declaration and its later amendments.
Mammographic examination
All women underwent mammography with a commercially machine (Selenia Value +, Hologic, Marlborough, Massachusetts, USA). Mammographic examinations included craniocaudal (CC) and mediolateral oblique (MLO) views of each breast.
Image assessment
Two radiologists with at least 10 years of breast imaging experience, according to the American College of Radiology Breast Imaging Reporting and Data System (ACR BI-RADS® Atlas 2013), retrospectively assessed breast composition and mammographic findings in separate reading sessions blinded to patient personal data, radiologic reports and each other’s assessments [10]. Disagreements were settled by consensus. When the two investigators did not reach consensus, mammographic images were evaluated by another radiologist with at least 30 years of experience, with the role of supervisor. For blinding purposes, the mammographic examinations included in the study were mixed into approximately two thousand other mammograms about different histological subtypes, benign findings and negative cases.
Image reading was carried out on standard views (CC, MLO).
According to the ACR BI-RADS® Atlas 2013, breast composition (BC) was categorized as: “A” if the breast was almost entirely fatty, “B” if there were scattered areas of fibroglandular density, “C” if the breast was heterogeneously dense or “D” if the breast was extremely dense.
Breast cancer mammographic findings were classified as: mass, calcifications, architectural distortion and asymmetries [10].
Histopathological findings
Breast-conserving surgery or mastectomy specimens were used as the reference standard in order to classify breast cancers as invasive ductal carcinomas and to evaluate lesions size. Histological findings were classified according to 2019 WHO classification [11].
Statistical analysis
Ordinal qualitative variables were analyzed using the non-parametric Mann–Whitney U test and the Kruskal–Wallis H-test. Quantitative variables were analyzed using the parametric t-test and ANOVA. Only the main mammographic findings were analyzed, whereas associated features where not. Breasts were evaluated as dense (C or D BC) or non-dense (A or B BC). A cut-off of 15 mm was chosen for tumor size. The variables found to be significant in univariate analysis were subsequently evaluated using multinomial logistic regression. Logistic regression with forward stepwise covariate selection was performed (with p values for entry and removal of 0.05 and 0.1, respectively). The statistical significance level was set at p < 0.05. All statistical analyses were performed using MedCalc Software v. 15.8 (Ostend, BEL).
Results
The mean age of the women in our study was 61.2 ± 15.6 years, range 32–89. The mean tumor size was 17.9 ± 8.3 mm.
Table 1 shows the tumor mean size in the three age groups, and Table 2 shows the mammographic findings related to BC in each age group.
Patients, age groups and tumor size, taking into account breast composition
Patients, age groups and tumor size, taking into account breast composition
SD, standard deviation.
Mammographic findings and breast composition in each age group
Tumor size showed to be significantly smaller in age group 2 (p < 0.001). Tumor size did not showed statistically significant differences between dense and non-dense breast groups (p = 0.3583).
Mass and architectural distortion showed to be significantly associated with non-dense breast, whereas calcifications showed to be significantly associated with dense breast (p < 0.0001). Within age groups 2, mass showed a statistically significant association with non-dense breast (p < 0.0001).
Considering only the three age subgroups of patients with non-dense breasts, mass showed a statistically significant association with age group 3 (p = 0.0060). Considering only the three dense breast age subgroups, mass showed a statistically significant association with age group 1 (p < 0.0001), and calcifications with age group 2 (p < 0.0001).
Mass findings showed a statistically significant correlation with age group 3 (p < 0.0001), mass showed a statistically significant association with non-dense breast (p < 0.0001), and mass showed a statistically significant association with a tumor size larger than 15 mm (p = 0.0083). Calcifications showed a statistically significant inverse correlation with age group 3 (p < 0.0001), and a statistically significant association with dense breast (p < 0.0001). Architectural distortion showed a statistically significant correlation with age group 2 (p = 0.0007). Asymmetry showed a statistically significant association with a tumor size of 15 mm or less (p = 0.0097).
Statistically significant results of multinomial logistic regression were the association between mass and non-dense breast (p < 0.0001) and between mass and tumor size larger than 15 mm (p = 0.0049) (Fig. 1).
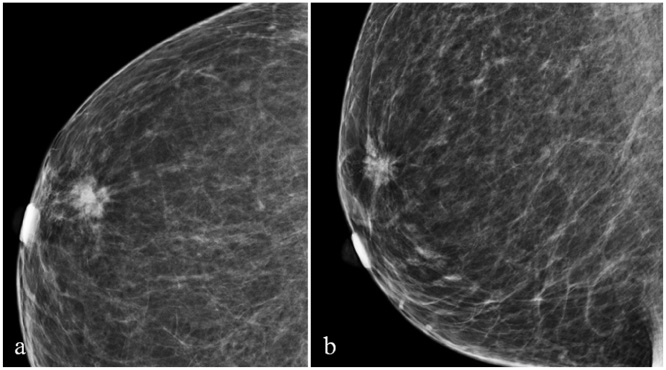
72-year-old woman with a 1.8 cm irregular spiculated mass in upper-outer quadrant of right breast: craniocaudal (a) and mediolateral oblique (b) views.
It is known that breast cancer mammographic sensitivity is mainly influenced by breast density, which has an inversely proportional correlation with age [5]. In fact, young women higher breast density requires sometimes the use of other diagnostic modalities such as ultrasound and magnetic resonance.
However, it is less clear whether breast cancer mammographic findings are related to patients’ age, breast composition and tumor size.
In the present study, in which the correlation between invasive ductal carcinoma mammographic findings and patients’ age, breast composition and tumor size was evaluated, mass was found to be significantly associated with non-dense breast and tumor size larger than 15 mm. Moreover, mass was found to be the most frequent mammographic finding in all three age groups.
Previous studies showed that a mass is the most frequent finding of IDC [7]. However, Jiang et al. reported a significantly higher proportion of masses occurring in postmenopausal women [7]. Moreover, Zhao et al. reported that patients who are less than 40 years of age showed less frequent mass finding [6]. On the contrary, after multinomial logistic regression we did not find a statistically significant association between mass and a specific age group. However, Zhao et al. included all breast cancer histological subtypes [6]. Gajdos et al. also reported that mass is the most frequent breast cancer finding; this study included lobular carcinoma and ductal carcinoma in situ, showed a higher prevalence of calcifications than our study, and breast composition was not evaluated [8].
Ferranti et al. reported that calcifications are most commonly associated with invasive breast cancer in patients aged less than 40 years. All breast cancer histological subtypes were included in this study [9]. On the contrary, after multinomial logistic regression we did not find a statistically significant association between calcifications and a specific age group. In our study, because we included only invasive neoplasms, we cannot confirm that calculations are more often associated with intraductal carcinoma as highlighted in previous studies [8,9].
Our study did not show statistically significant tumor size differences between dense and non-dense breast groups. Bertrand et al. reported that lesion size is larger in the case of higher breast density. However, in this present study breast density was assessed using two computer-assisted mammographic density softwares; therefore, ACR BI-RADS® Atlas 2013 was not used in order to evaluate BC [12,13].
In our cohort of patients, women aged less than 50 and women aged 70 or older showed statistically significant larger tumors. This trend agrees with other studies, and the reason could be the absence of routine check-ups for these patients [14–19].
The main limitation of the present study is the retrospective design. The relatively limited number of patients did not allow a more detailed analysis of the mammographic findings. It was not possible to evaluate the morphology and the distribution of the calcifications or the characteristics of the masses. It is considered that in order to perform a more-in-depth examination of these findings, a larger multicentric study is needed. However, to our knowledge this is the first study in which a statistical analysis was performed in order to assess mammographic findings of patients with IDC, taking into account BC, patient’s age, and tumor size.
In conclusion, this study emphasizes the heterogeneity of invasive ductal carcinoma mammographic findings, particularly showing that mass is significantly related to breast composition and tumor size.
