Abstract
INTRODUCTION:
Phyllodes tumours represent 0.3–1% of breast tumours, typically presenting in women aged 35–55 years. They are classified into benign, borderline and malignant grades and exhibit a spectrum of features. There is significant debate surrounding the optimal management of phyllodes tumour, particularly regarding appropriate margins.
METHODS:
This is a retrospective review of a prospectively maintained database of patients who underwent surgical management for phyllodes tumours in a single tertiary referral centre from 2007–2017. Patient demographics, tumour characteristics, surgical treatment and follow-up data were analysed. Tumour margins were classified as positive (0 mm), close (≤2 mm) and clear (>2 mm).
RESULTS:
A total of 57 patients underwent surgical excision of a phyllodes tumour. The Mean age was 37.7 years (range: ages 14–91) with mean follow-up of 38.5 months (range: 0.5–133 months). There were 44 (77%) benign, 4 (7%) borderline and 9 (16%) malignant phyllodes cases. 54 patients had breast conserving surgery (BCS) and 3 underwent mastectomy. 30 (53%) patients underwent re-excision of margins. The final margin status was clear in 32 (56%), close in 13 (23%) and positive in 12 (21%). During follow-up, 4 patients were diagnosed with local recurrence (2 malignant, 1 borderline and 1 benign pathology on recurrence samples).
CONCLUSION:
There are no clear guidelines for the surgical management and follow-up of phyllodes tumours. This study suggests that patients with malignant phyllodes and positive margins are more likely to develop local recurrence. There is a need for large prospective studies to guide the development of future guidelines.
Introduction
Phyllodes tumour (PT) is an uncommon fibroepithelial lesion accounting for 2–3% of all fibroepithelial breast tumours and 0.3% to 1% of breast tumours in general [1]. PTs display a spectrum of behaviours. Less aggressive tumours behave similarly to benign fibroadenomas while those at the opposite end of the spectrum may metastasize distally or degenerate into sarcoma like lesions [2]. Using various histological features, such as atypia, mitoses, surgical margin and stromal cellularity, the World Health Organisation (WHO) classifies PT into benign, borderline and malignant grades [3]. However, despite this classification system, obtaining a diagnosis can be challenging, particularly when differentiating between PT and fibroadenoma [4].
PT typically presents in middle aged women (years 35–55), approximately 20 years later than fibroadenoma [5]. Male PT is rare but cases have been reported [6]. In general they present with a mobile, multinodular lump ranging from 4 cm–7 cm without pain.
Optimal management of borderline and malignant PT is subject to debate [7,8]. Surgical excision with >1 cm margins has been described as “definitive primary therapy” [9]. The National Comprehensive Cancer Network (NCCN) and MD Anderson also recommend >1 cm margins [10,11]. However, various studies suggest margins less than 1 cm are sufficient in reducing recurrence, with 1 cm margins described as over treatment [8,12–14]. A 2020 meta-analysis by Thind et al. showed no significant difference in LR for borderline and malignant tumours excised with margins <1 cm versus margins >1 cm [7]. Results of the Second International Consensus Conference on lesions of uncertain malignant potential in the breast (B3 Lesions) showed that 98% of participants felt PT diagnosed on core-needle biopsy (CNB) should be excised. 72% felt that surgical excision should be performed, while 22% felt vacuum assisted excision was sufficient [15]. If negative margins are achievable, breast conserving surgery (BCS) is as effective as mastectomy in the treatment of PT [2,16,17]. In a comparison of 821 patients undergoing mastectomy or wide local excision (WLE) for malignant PT, WLE was associated with equivalent or improved cause specific survival regardless of tumour size [17].
The aim of this study was to outline clinicopathological characteristics, surgical management and outcomes of phyllodes tumours in a tertiary referral centre over a 10-year period.
Methods
Patient selection
All patients who underwent breast excision for PT from January 2007 to December 2017 were considered for this study. Any patients who had other high risk lesions were excluded. We also excluded patients who did not undergo surgery.
Demographics and procedure performed
Demographics and procedure performed
∗Mean (range).
Re-excision rates
Patients attending this unit with a diagnosis of PT were counselled on this condition and their prognosis and were offered BCS. Non-palpable lesions underwent wire-guided excision. Surgical resection size was based on the pre-operative size estimation on imaging. Intraoperative imaging or histopathological assessment was not routinely used to guide excision. Mastectomy was offered in cases where negative margins were otherwise unachievable.
Endpoints
Patient demographics, tumour characteristics and surgical treatments were recorded. Outcomes such as margin status, re-excision rates and local recurrence (LR) during follow-up were analysed. Margin status was classified as: positive (0 mm), close (≤2 mm) and clear (>2 mm). Margin width was defined as width of normal mammary tissue between tumour edge and specimen edge. Post-operatively, all patients were offered surveillance in the form of annual mammography for five years.
Initial margins
Initial margins
Final margins
Follow-up and recurrences
Continuous variables included patient age and tumour size. Categorical variables included tumour grade, margin status and rate of re-excision and local recurrence. Descriptive statistics was performed using frequency analysis. Continuous variables were reported as mean values with minimum to maximum range. Categorical variables were reported as numbers (n) and percentage (%). Statistical analysis was performed using IBM software SPSS Version 24. As this study involved retrospective analysis of a database, formal ethics committee approval was not required.
Results
From January 2007 to December 2017, 57 patients underwent surgical excision of PT. The Mean age was 37.7 years (range: ages 14–91) with mean follow-up of 38.5 months (range: 0.5–133 months). WHO classification was benign in 77.2% (n = 44∕57), borderline in 7.0% (n = 4∕57) and malignant in 15.8% (n = 9∕57). Of these, 94.7% (n = 54∕57) patients underwent BCS while 5.3% (n = 3∕57) underwent mastectomy (Table 1).
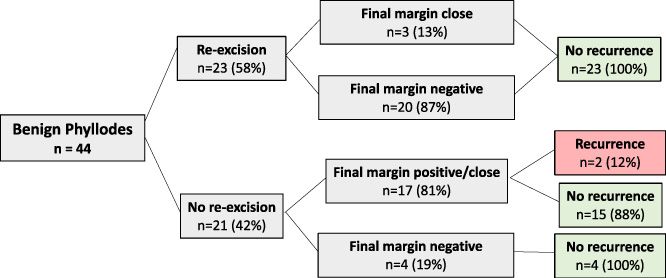
Outcomes of benign group.
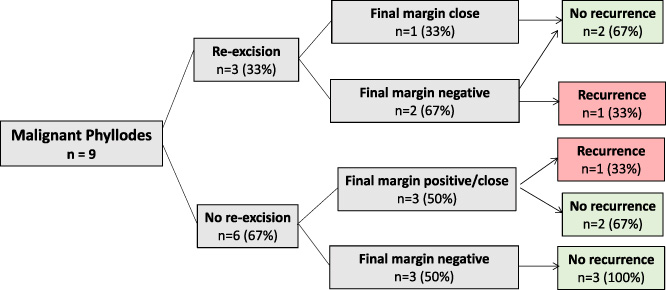
Outcomes of malignant group.
A total of 30 patients (52.6%) underwent re-excision of margins, all of whom initially underwent BCS. Three patients underwent a third excision and one patient required a fourth surgery (Table 2).
Initial surgical margins were positive in 61.4% (n = 35∕57), close in 24.6% (n = 14∕57) and negative in 14.0% (n = 8∕57). Final margins were positive in 21.1% (n = 12∕57), close in 22.8% (n = 13∕57) and negative in 56.1% (n = 32∕57). In cases of borderline or positive final margins, an individualised treatment approach was utilised. Patients were counselled on their diagnosis, margin status and prognosis. The decision not to further excise margins was based on tumour grade, location, patient comorbidities, patient preference and multidisciplinary discussion. All patients were followed up with yearly mammography and clinical examination for five years. Further data regarding margins is illustrated in Table 3 and Table 4.
Patients were followed up for an average of 38.5 months (range 0.5–133 months). Overall, 7% (n = 4∕57) of patients had LR. Within the benign and malignant groups there were LRs in 4.5% (n = 2∕44) and 22.2% (n = 2∕9) of patients respectively. There was no LR within the borderline group (Table 5). Three patients (one benign and 2 malignant) developed LR within 24 months of the first surgery. One patient had a recurrence within 60 months. Two of 12 patients with positive final margins (16.7%) had recurrence, one of thirteen patients with close margins (7.7%) and one of thirty-two patients with negative margins (3.1%) had a LR. Both patient with a benign diagnosis who recurred had positive final margins. Of the malignant cases that recurred one had a close final margin and one had a negative final margin. Two patients underwent BCS for their recurrences, one patient underwent mastectomy and one patient underwent a local excision having previously undergone mastectomy. None received adjuvant therapy. Local recurrence data as it pertains to margin status and tumour grade is illustrated in Figs 1 and 2.
In this review of 57 women undergoing surgical excision of PT, four patients (7%) were diagnosed with local recurrence during follow-up. Recurrence rates for benign, borderline and malignant PT were 4.5%, 0% and 22.2% respectively. With regard to margin status, 16.7% (n = 2∕12) of the margin positive group, 7.7% (1/13) of the close margin group and 3.1% (1/32) of the negative margin group developed LR (Figs 1 and 2).
These outcomes contrast WHO data which reports an overall LR rate of 21% for PT and LR of 10–17% for benign and 14–25% for borderline cases. The WHO rates for malignant PT range from 23–30%, similar to the rates in this study (22.2%) [18]. A large 2019 meta-analysis examining 9,234 cases reported LR rates of 12% overall 8% for benign, 13% for borderline, and 18% for malignant PT [19]. Our results for overall, malignant and benign cases are similar. However, particularly noteworthy from our study is the 0% recurrence rate for borderline cases which is not in keeping with published literature. This is explained by the small sample size of just four cases.
There is a lack of definitive guidelines for the excision and surveillance of phyllodes tumour. Surveillance is of utmost importance but is not sufficient alone. Patient counselling and education is a key aspect of management in order to ensure adherence to surveillance schedules and lifestyle modifications. In our centre, patients with PT who undergo diagnostic excision undergo 5 year surveillance with yearly mammography and outpatient follow-up in the symptomatic outpatient department. After this patients may be followed in the national breast screening service if they fall within the age range of 50–70 years.
Complete surgical excision with negative margins is the standard of care. The vast majority of studies have shown high LR rates associated with positive margins [20–23]. The results of this study are no different with 17% of patients with positive margins developing LR. Recent studies however, have suggested that negative margins may not be associated with high LR for borderline or benign PT [19].
Adequate margin size is also subject to debate. In general, margins of ≥1 cm are considered negative, with studies showing an association with significantly lower LR [2,16,24]. Multiple guidelines recommend at least 1 cm margins [10,11]. As previously stated, multiple studies have suggested that margins <1 cm are sufficient, particularly for patients in the benign category [8,12–14,25,26]. Thind et al.’s recent meta-analysis has suggested that margins <1 cm are sufficient for all PT [7]. Our study aimed for a surgical margin of ≥2 mm with LR rates that are comparable to those within the literature. A 2017 study by Moo et al. examining benign phyllodes tumour reported excellent outcomes with this margin size [26]. Similarly, Wen et al. utilised margins of ≥2 mm with acceptable outcomes [27]. A recently published, 50-year retrospective study of 124 patients with borderline or malignant PT defined margins of ≥1 mm as negative. This study by Spanheimer et al. had an overall LR of 12%, with 9% for negative margins or 33% for close/positive margins [28]. It is clear that there is a lack of consensus in the current literature and guidelines regarding adequate margin size. In situations where there is a lack of clarity, as is the case for PT, it may seem safer to opt for a >1 cm margin. However, such an approach may render breast conservation impossible, result in poorer aesthetic and patient related outcomes, and lead to increased re-excisions and costs [7,29]. It is also helpful to draw parallels from the management of other disease processes. As suggested by Wood in a 2013 review, there is a lack of definitive evidence for a particular margin width for invasive cancers that is associated with lower rates of recurrence [29,30]. Furthermore, guidelines for the excision of ductal carcinoma in-situ recommend margins of >2 mm [31].
Axillary disease is rare with PT and none of the subjects in this study had axillary disease, in keeping with other published reports [17,32,33]. The roles of systemic chemotherapy and hormone therapy are limited. While some small studies have reported successful treatment with chemotherapy, there have been no randomised controlled trials examining its adjuvant use in PT. Hormonal therapy has been shown to be ineffective [9,34]. Radiotherapy was shown to significantly reduce LR in borderline and malignant PT with no effect on overall or disease-free survival [35]. Interestingly, use of adjuvant radiotherapy is not widespread as per a National Cancer Database review [36]. None of the patients in this study received adjuvant treatment.
This study has some limitations. It is a retrospective review with a small sample size in comparison to other published studies. It is also lacking data on important risk factors such as family history, smoking, BMI.
Conclusion
PT accounts for approximately 2% of all primary breast cancer. Currently, there are no clear guidelines for their surgical management and follow-up. Our data suggests that patients with malignant phyllodes and positive final margins are more likely to develop local recurrence. This is in keeping with published data and suggests that such cases may require further excision to ensure a negative (<2 mm) margin. In contrast, patients with borderline and benign phyllodes and positive margins may avoid further surgery. These results highlight the need for large prospective studies in order to steer the development of guidelines.
Footnotes
Conflict of interest
All authors declare no conflict of interest.
