Abstract
Distant metastasis from malignant Phyllodes tumour (PT) is rare. They generally metastasize to the lung, bone, pleura, and liver. We present a very rare case of a 25-year-old woman with intraabdominal metastases from malignant phyllodes tumour of the breast. She presented with left breast lump and the biopsy was Phyllodes tumour. She underwent a two staged surgery; left modified radical mastectomy followed by left latissimus dorsi musculocutaneous flap cover. She received adjuvant radiation therapy to left chest wall. Following which she developed intrabdominal mass which was proven to be metastasis from Phyllodes tumour. She was then advised palliative chemotherapy. Malignant Phyllodes with distant metastases has a dismal prognosis. We propose it is preferable to have a CT Thorax and Abdomen as a staging workup for patients with malignant PT to identify those with poor prognosis.
Introduction
Phyllodes tumour (PT) accounts for less than 1% of all breast neoplasms [1]. They can be classified into benign (58%), borderline malignant (12%) and malignant (30%) subtypes based on histological findings [2]. The incidence of distant metastasis is rare and mostly occurs with the malignant subtype. Usual sites of distant metastases are lungs, bones, pleura and liver. We present a very rare case of a woman with omental deposits and mesenteric metastases from malignant phyllodes tumour of the breast.
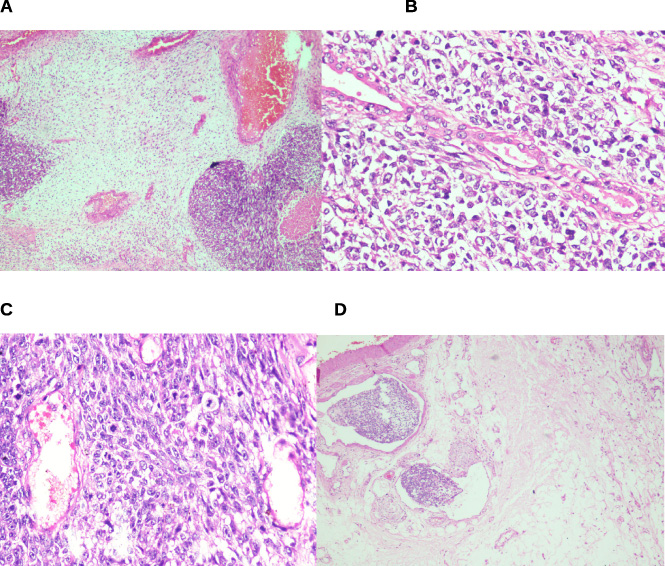
(A) Stromal heterogeneity and foci of necrosis (40×; H&E). (B) Epithelial and stromal components (100×; H&E). (C) Increase in mitotic activity (200×; H&E). (D) Lymphovascular emboli (100×; H&E).
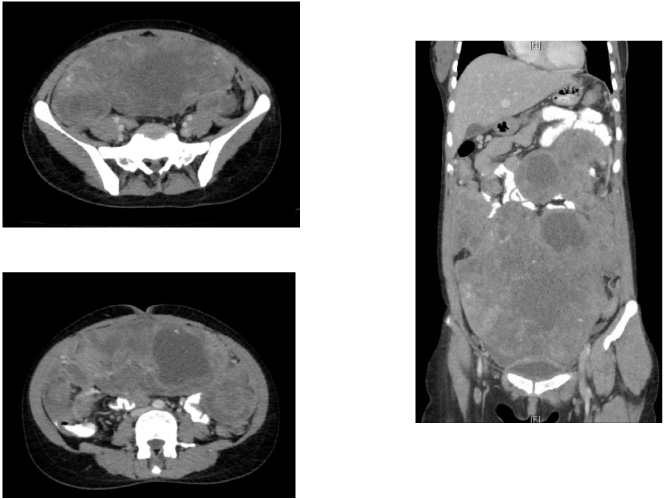
CT abdomen and pelvis axial and coronal sections showing the intra-abdominal metastasis from malignant phyllodes tumour of left breast.
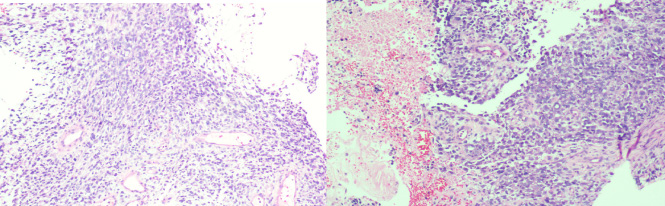
Core biopsy of the intra-abdominal mass with metastatic phyllodes tumour (100×; H&E.)
A 25-year-old unmarried lady presented with gradually progressive left breast lump for 1-year with no other associated symptoms in December 2018. She had no family history of malignancy. Left breast examination showed a 25 × 20 cm lump with stretched skin, engorged veins and variegated consistency involving all the quadrants. She had no palpable regional lymph nodes. Contralateral breast was normal.
Her Ultrasonogram of breasts showed multiple large well defined wider than taller heterogenous lesions with mild internal vascularity and cystic areas. The retroareolar region was normal and there were no axillary nodes. Biopsy from the lesion was reported as benign phyllodes tumour. She defaulted the advice to undergo wide local excision and reconstruction surgery.
She again presented in October 2019 with increase in size of left breast lump, associated with pain, ulceration and bleeding from the mass. The breast mass had enlarged to 30 × 25 cm with a 6 × 4 cm ulcer over the upper half of the breast with slough covered base and greenish yellow discharge. There were no palpable lymph nodes. She underwent a two staged surgery; left modified radical mastectomy on 21.10.2019 followed by left latissimus dorsi musculocutaneous flap cover on 14.11.2019 as she had large defect and the wound closure was difficult.
Intraoperatively there was a 20 × 20 cm large lobulated tumour replacing the entire left breast with multiple nodules on the surface and an ulcer in the upper part. There were multiple enlarged nodes in the left axilla. There was also a 2 × 2 cm nodule seen between the Pectoralis major and minor muscle which was also excised.
Surgical pathology of the mastectomy specimen was reported as a malignant phyllodes tumour. Grossly, the tumour was found to replace the entire breast with a maximum tumour dimension of 22 cm. Ulceration of the overlying skin was noted. Microscopically, the tumour had infiltrating borders with both epithelial and stromal components, the latter being the predominant component and composed of diffuse, moderate to markedly hypercellular areas with spindle shaped cells displaying high grade cytological atypia and mitotic activity of upto ∼10/10 hpf. There was evidence of stromal overgrowth and stromal heterogeneity. The spindled tumour cells were found to be negative for HMWCK, CD34, P63, SOX10 and GATA 3 immunostains. Lympho-vascular emboli were noted (Fig. 1). The tumour involved the posterior resection margin and was less than 0.1 cm away from the other resection margins. The dimension of the intermuscular nodule was 2.5 cm which also showed similar tumour morphology. One of the eleven lymph nodes was also infiltrated by the tumour from the perinodal adipose tissue, however definite evidence for metastasis was not seen.
She received adjuvant radiation therapy to left chest wall by medial and lateral tangents to a dose of 60 Gy in 30 fractions. In February 2020, patient had complaints of diffuse abdominal pain and multiple episodes of vomiting. There was diffuse tenderness over all regions of abdomen. Ultrasound abdomen showed a large mass extending from the epigastrium to the pelvis and right adnexa, with solid and cystic areas and internal vascularity. CT abdomen and pelvis (Fig. 2) showed large lobulated heterogeneously enhancing solid lesion with scattered areas of necrosis measuring 25 × 16 × 10 cm, seen along the infracolic greater omentum. There were multiple discrete soft tissue deposits in the supracolic omentum and in the mesentery, largest measuring 2 cm and 5 cm respectively. There was a small sub-pleural nodule of 6 × 8 mm in the posterior basal segment of lower lobe of left lung. Ultrasound guided biopsy from the intrabdominal mass was consistent with metastatic phyllodes tumour (Fig. 3). She was advised palliative chemotherapy with Doxorubicin.
Discussion
Malignant PTs of breast is pathologically characterised by high mitotic rate of 10/10 hpf, marked stromal cellularity, atypia, necrosis and infiltrative margins [3]. Most challenging question is predicting who will have local recurrence, metastatic disease or both. The local recurrence rates range from 15 to 40%. The axillary lymph nodal metastasis is rare (<1%) [4]. The most important factors which predicts local recurrence and metastasis are stromal atypia, mitoses, overgrowth and positive surgical margins (AMOS criteria) [5]. A nomogram based on this criteria can predict the recurrence-free survival in PT [5]. Other factors are nuclear pleomorphism, large tumour size, necrosis, grade and younger age [2,6].
Complete surgical excision with 1 cm margin is the standard approach. Mastectomy is indicated if negative margins cannot be achieved or in large tumours where breast conserving surgery leads to poor cosmetic [7]. Adjuvant radiotherapy has shown to reduce local recurrence in malignant PTs [8]. Meta-analysis by Zeng, et al. showed that adjuvant radiation reduced local recurrences in borderline and malignant phyllodes with hazard ratio of 0.31 (95% CI −0.10 to 0.72), but there was no overall survival benefit [9].
In malignant PT the incidence of distant metastases ranges between 9 to 27%. It could occur by hematogenous spread or via the vertebral venous plexus. The vertebral venous plexus drains into the intercostal veins, the vena cava, the azygos vein and pelvic veins. When the thoracic or the abdominal pressure increases, the tumour cells spread to these veins and metastasize to distant sites [10].
The most common site of distant metastasis is lung (66–85%) followed by bones (∼30%) and less commonly liver and brain. Other reported sites include skin, oral cavity, salivary glands, thyroid, heart, ovaries, pancreas, adrenal gland and spleen [11]. To our best knowledge we have come across only one case report mentioning the metastases to omentum and our case is the second such one. Metastases can occur either synchronously or within a few months.
Several biomarkers have been identified in multiple studies which may predict metastases from PT. Overexpression of CD10, Axl and ST6GALNAC2 were significantly associated with distant metastasis [12,13]. Mutations of MED12,FLNA,SETD2 and KMT2D also contribute to tumour progression in PT [14].
Chemotherapy has a very limited role and is given as palliation in metastatic setting. The various chemotherapeutic agents used are Ifosfamide, Cisplatin, Etoposide or Doxorubicin. Mitus, et al. found that Doxorubicin and Ifosfamide-based chemotherapies have some efficacy in the treatment of metastatic disease [15]. The NCCN recommends that treatment of Phyllodes tumour with distant metastasis should follow the NCCN Soft Tissue Sarcoma Clinical Practice Guidelines in Oncology [16]. The prognosis of patients with metastasis is very poor with an average survival of less than 2 years [11].
Our patient fulfils the AMOS criteria and had higher chance of developing distant metastasis. She developed rapid dissemination with intrabdominal mass over a period of 4 months after surgery. However, since she did not have an imaging of abdomen at diagnosis, one may argue that the lesions in abdomen could have been present during initial presentation. But given the rapid increase in size of abdominal lesion, we strongly believe that there could not have been such lesions during initial presentation. It is difficult to predict the rapid dissemination seen in our patient due to the limitations of our knowledge regarding this disease.
Conclusion
Malignant Phyllodes with distant metastases has a dismal prognosis. We propose it is preferable to have a CT Thorax and Abdomen as a staging workup for patients with malignant PT to identify those with poor prognosis.
Conflicts of interest
None.
