Abstract
BACKGROUND:
Despite the increase in chances of cure for early breast cancer (EBC) patients, approximately 20–45% of them will experience a disease recurrence, particularly bone metastases in 60–80% of cases, which occur more frequently in luminal subtypes. Endocrine therapy (ET) has always been the milestone of adjuvant treatment for hormone receptor-positive EBC patients, leading to indubitable reduction of disease recurrence risk. However, adjuvant aromatase inhibitors (AIs) therapy may promote a progressive decrease in bone mineral density (BMD), which can lead to osteoporosis. The increased bone resorption associated with osteoporosis may provide fertile soil for cancer growth and accelerate the development of bone metastases.
PATIENTS AND METHODS:
In this single-institution cohort study, we performed a retrospective analysis of “luminal-like” EBC patients who experienced bone recurrence after a subsequent disease free interval. The aim of the study was to evaluate the median time to skeletal recurrence (TSkR).
RESULTS:
143 patients experienced bone recurrence. Median TSkR was 54 months (95%CI: 45–65). Among patients who received adjuvant AIs median TSkR was 35 months (95%CI: 25–54), while among patients who did not was 61 months (95%CI: 50–80) (HR = 1.45 [95%CI: 0.97–2.17], p = 0.0644). After adjusting for TNM stage (AJCC 8th edition), adjuvant AIs treatment was significantly related to a shorter TSkR (HR = 1.60 [95%CI: 1.06–2.42], p = 0.0244). Adjuvant Tamoxifen, adjuvant AIs/Tamoxifen and no-treatment did not revealed to be associated to TSkR.
CONCLUSIONS:
In this cohort of EBC patients with bone recurrence, AIs treatment seems to be related to a shorter TSkR. AIs-induced bone resorption might represent the underlying mechanism.
Keywords
Introduction
Despite the increase in chances of cure for early breast cancer (EBC) patients, approximately 20–45% of them will experience a disease recurrence [1]; particularly bone metastases in 60–80% of cases [2], and bone-only metastases in 17–37% [3]. It is known that the most common metastatic site in luminal A (66.6%), luminal B (71.4%) and luminal/HER2+ tumors (65%) is the bone, while it is less frequently involved in basal-like tumors (39%) [4]. Therefore, we can assume that the expression of hormone receptors in breast cancer (BC) patients is related to a higher risk of developing bone metastases. On the other hand, endocrine therapy (ET), with either tamoxifen and aromatase inhibitors (AIs), has always been the milestone of adjuvant treatment for hormone receptor-positive EBC patients, leading to an indubitable reduction of disease recurrence risk [5].
In a previous “real life” study of bone metastatic breast cancer (BMBC) patients receiving denosumab, we performed an explorative analysis among the 40 patients with metachronous “luminal-like” disease, who experienced bone recurrence as first metastatic site. We found that the median time to skeletal recurrence (TSkR) was shorter for patients previously exposed to AIs compared to those who were not (53.0 versus 102.0 months, respectively; p = 0.0300) and longer for patients previously treated with tamoxifen compared to those who were not (102.0 versus 59.0 months, respectively; p = 0.0466) [6]. Both the results were not confirmed at multivariate analysis, but we drew inspiration to speculate on the effects of adjuvant endocrine therapy on bone health of EBC patients. Particularly, it is well known that AIs in post-menopausal women may promote a progressive decrease in bone mineral density (BMD), which can lead to osteoporosis [7]. Osteoporosis is a primary cause of morbidity in itself, but several studies have already revealed that the impairment of the bone microarchitecture/microenvironment related to the osteoporosis may provide fertile soil for the activation of the metastatic cascade, from the seeding of tumor cells to the expansion of bone lesions [8,9]. Enhanced osteoclast activity has been shown to exacerbate the growth and progression of bone metastases and osteoclast-derived proteolytic enzymes are able to promote angiogenesis and cancer cell invasion to metastatic sites [10–14]. In vivo pre-clinical data also showed how AIs-induced bone loss may increase breast cancer progression in bone [15]. Furthermore, untreated osteoporosis has been associated with accelerated development of bone metastases in EBC patients who experienced bone metastasis relapse [16].
On this basis, we conducted a single-institution retrospective analysis to further investigate the clinical characteristics and the TSkR of “luminal-like” EBC patients who experienced bone recurrence.
Patients and methods
In this single-institution, retrospective cohort study we evaluated consecutive “luminal-like” EBC patients who experienced bone recurrence, after had underwent radical resection of the primary tumor. Patients were eligible if they had histologically confirmed diagnosis of “luminal-like” BC, and a subsequent disease free interval. Patients were followed at the Medical Oncology department of St. Salvatore Hospital in L’Aquila. The study period ranged from September 1986 to November 2017; the data cut-off was May 2019. Estrogen and Progesterone Receptor expressions were evaluated by immunohistochemistry (IHC), using Dako monoclonal antibodies. HER2 analysis was performed by IHC on paraffin embedded tissue from the primary tumor and/or metastatic site (Hercept-Test®, Genentech Inc. subject to licenses held by Dako Denmark A/S and F. Hoffmann-La Roche Ltd.). Fluorescence in situ hybridization (FISH) and silver in situ hybridization (SISH) were used for cases of doubtful interpretation. “Luminal-like” disease was defined in any case of Estrogen and/or Progesterone Receptor expression. Clinical follow-up was performed by radiographic imaging (X-ray, computed tomography scan, ultrasound scan, magnetic resonance), according to clinicians choice in clinical practice.
The aim of the study was to evaluate the median time to skeletal recurrence (TSkR), which was defined as the length of time from the surgical radicalization of the primary tumor to the diagnosis of first skeletal metastasis. The following patients characteristics were evaluated: age, menopausal status (post- versus pre-), histology (ductal, lobular, others), “luminal-subtype”, baseline TNM staging (updated according to the 8th AJCC version) [17], grading, adjuvant and neoadjuvant chemotherapy, adjuvant endocrine therapy (tamoxifen, AIs, none). TSkR was evaluated according to the adjuvant ET as follow: patients who received exclusively AIs (yes versus no), patients who received exclusively tamoxifen (yes versus no), patients who received both AIs and tamoxifen (sequential) (yes versus no), and patients who did not received adjuvant ET (yes versus no). Chi-square (𝜒2) and binary logistic regression were used to evaluate the correlation between age, menopausal status and AIs adjuvant ET because baseline clinical features which were significantly related to AIs therapy were not used in the multivariate analysis [18]. Considering the long interval of patients evaluation, the only baseline factor available for every patients was the updated TNM, which was used for the multivariate analysis of the TSkR. Median TSkR, median DFS were evaluated using the Kaplan–Meier method. Median period of follow-up was calculated according to the reverse Kaplan–Meier method. Cox proportional hazards regression was used for the univariate and multivariate analyses of TSkR. All the statistical analyses were performed with MedCalc Statistical Software version 19.0.4 (MedCalc Software bvba, Ostend, Belgium; https://www.medcalc.org; 2019).
Demographic and disease characteristics of the patients
Demographic and disease characteristics of the patients
Luminal-A: hormone receptors positive, HER2 negative, Ki-67 ≤ 20%. Luminal-B: hormone receptors positive, HER2 negative, Ki-67 > 20%. Luminal-HER2: hormone receptors positive, HER2 positive, any Ki-67. HER2: human epidermal growth factor receptor 2.
143 consecutive patient were considered eligible in the study period. All patients characteristics are summarized in Table 1. Among post-menopausal patients, 28 (30.1%) received exclusively AIs, while among pre-menopausal patients 5 (10.0%) received exclusively AIs (p = 0.0067). There also was a significant association between age and exclusive AIs treatment (p < 0.0001; OR = 1.08 [95%CI: 1.04–1.12]). At a median follow-up of 178 months, median TSkR was 54 months (95%CI: 45–65). Among patients who received exclusively adjuvant AIs median TSkR was 35 months (95%CI: 25–54), while among patients who did not received exclusively adjuvant AIs was 61 months (95%CI: 50–80) (HR = 1.45 [95%CI: 0.97–2.17], p = 0.0644) (Fig. 1). After adjusting for TNM stage, adjuvant AIs was significantly related to a shorter TSkR (HR = 1.60 [95%CI: 1.06–2.42], p = 0.0244) (Table 2). Adjuvant Tamoxifen, adjuvant AIs/Tamoxifen and no-treatment did not reveal to be associated to TSkR.
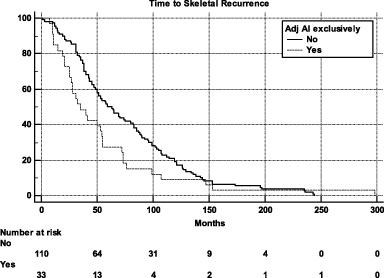
Kaplan–Meier survival estimate for Time to Skeletal Recurrence according to adjuvant endocrine therapy with AIs (exclusively yes versus no).
Univariate and multivariate analyses of Time to Skeletal Recurrence
This explorative analysis was aimed to evaluate the clinical characteristics and the TSkR among luminal-like EBC patients in our single-institution case series and our purpose was merely descriptive. As mentioned above, one of the major AIs side effects is the progressive decrease in BMD [7,19,20], which may lead to osteoporosis. Furthermore, we have already discussed about the possible role of the osteoporosis-induced impairment of the bone microarchitecture in the promotion of bone metastases expansion [8,9]. Therefore, it is reasonable to think that AIs-induced osteoporosis might be the underlying mechanism of the shortened TSkR in patients who exclusively received AIs.
The study population comes from a historic cohort and most of the EBC patients did not received antiresorptive treatments during adjuvant ET, which to date have already proved to be effective in this setting. The role of denosumab in the prevention of AIs-induced osteoporosis was confirmed in a phase III trial [21], while several randomized trials showed an improvement in both disease-free survival and overall survival for EBC patients treated with adjuvant ET plus bisphosphonates compared to ET alone [22–25]. The recent evidences regarding the role of denosumab in improving disease-related outcomes are contradictory: a phase III study showed that denosumab significantly improved disease-free survival respect to placebo in postmenopausal EBC patients receiving adjuvant AIs [26], while another study revealed bone metastasis-free survival was not significantly different among the two groups [27]. However, we have to emphasize than in the latter a consistent proportion of enrolled patients were hormone-receptor negative (22% and 23% in the denosumab and placebo arm, respectively), and only the 54% among the receptor positive patients received adjuvant AIs [27].
Of course, we are not questioning the role of AIs as adjuvant ET and we must not fail in recognizing the limits of this analysis. The long study period exposed us to the risk of data dispersion, while the small sample size and the retrospective design have further biased the analysis. Moreover, we must take into account the lack of data regarding measurements of BMD (such as computerized bone mineralometry). This study was planned with a selection bias because we evaluated only patients who experienced bone recurrence, without a control group; therefore the results cannot be applied to the general population (including patients without disease recurrence) as they are.
Conclusion
We revealed that in a population of luminal-like EBC patients who all experienced bone recurrence, an adjuvant ET exclusively with AIs seems to anticipate the clinical emergence of bone metastases (which would occur in any case).
In our opinion, this phenomenon might be related to the AIs-induced osteoporosis, and it gives back further relevance to bone-oriented preventive treatments/strategies which should be promoted for every EBC patient candidate to receive adjuvant ET.
Footnotes
Acknowledgements
None.
Ethics approval and consent to participate
All patients alive at the time of data collection provided an informed consent for the present retrospective analysis. To guarantee the confidentiality of personal data for deceased patients, all the available procedures to ensure anonymity have been used. The procedures followed were in accordance with the precepts of Good Clinical Practice, and the declaration of Helsinki. Being a retrospective update of data previously collected, approval by institutional review boards was not required, although a notification was sent (normative ref. Gazzetta Ufficiale della Repubblica Italiana n. 76 of 31-3-2008) to the local responsible committee on human experimentation (Comitato Etico per le province di L’Aquila e Teramo).
Consent for publication
Not applicable.
Conflicts of interest
The authors declare no conflicts of interest.
Availability of data and materials
The datasets used during the present study are available from the corresponding author upon reasonable request.
Funding
No funding was received.
Authors’ contributions
All authors contributed to the publication according to the ICMJE guidelines for the authorship.
Study conception and design: KC. Acquisition of data: FP, SR, VC, AP, GP, TS, LV, GP. Analysis and interpretation of data: FP, AP, GP, CF, KC. Drafting of manuscript: FP, AP, GP. Critical revision: CF, KC.
All authors read and approved the submitted version of the manuscript (and any substantially modified version that involves the author’s contribution to the study). Each author have agreed both to be personally accountable for the author’s own contributions and to ensure that questions related to the accuracy or integrity of any part of the work, even ones in which the author was not personally involved, are appropriately investigated, resolved, and the resolution documented in the literature.
