Abstract
BACKGROUND:
Although widely studied, the role of HPV in the genesis of breast carcinomas remains elusive due to the diversity of results across studies, possibly caused by the wide methodological heterogeneity, some of them with inadequate methods.
OBJECTIVE:
To verify the association between HPV and breast cancer through the meta-analysis of studies that used the best-recognized techniques for viral detection and tissue conservation.
METHODS:
A systematic review and meta-analysis restricted to studies that detected HPV by PCR in fresh and frozen tissue from breast cancer were conducted to obtain greater homogeneity. PubMed, Scopus, Science Direct, Cochrane Library, and SciELO were searched until December 14, 2019. Search terms included “breast cancer” and “HPV” without language restrictions. Eleven studies were included in the meta-analysis. The pooled relative risks and 95% confidence interval (95% CI) were calculated, and heterogeneity was assessed using the I-squared (I2).
RESULTS:
The selected studies had very low heterogeneity (2%). There is a 2.15 times higher combined relative risk (95% CI = 1.60-2.89) of detecting HPV in breast cancer than in cancer-free breast controls with a statistically significant p-value (p < 0.0001).
CONCLUSION:
Our data support the association of DNA-HPV with breast carcinomas. Further studies are needed to find out which breast cancer subtypes this association is most frequent.
Introduction
Breast cancer (BC) is the most reported cancer in women worldwide causing significant morbidity and mortality [10,44]. Some risk factors (e.g., age, familial history, body mass index, hormones) are known; however, they may be missing in 50–80% of BC. For this reason, researchers have been looking for other factors, including the role of viral infections such as Epstein-Barr virus (EBV), mouse mammary tumor virus (MMTV), and human papillomavirus (HPV) as a potential etiology in BC [31].
Some HPV genotypes are more associated with carcinogenesis (16, 18, 31, 33, 35, 39, 45, 51, 56, 58, 59, 66, 68, 73, and 82) and are called high-risk (HR) HPV; while others, most associated with papillomatous lesions (e.g., 6, 11), although potentially carcinogenic are called low risk (LR) HPV[42]. HPV can integrate into the host genome and can cause genomic instability, increasing the risk of neoplastic progression due to E6 and E7 oncoprotein synthesis. These viral proteins degrade p53 and pRB tumor suppressor proteins leading to cell proliferation [2,41]. They also play a role in innate immunosuppression, which is critical for avoiding infection clearance [2,33].
Imagining that oncogenic HPV strains can reach breast tissues and cause or facilitate malignant transformation is not an exercise in futility. HPV requires the availability of proliferating epidermal tissue as occurs in the breast of patients [18]. Viral transmission in breast tissue is reasonable via virus-contaminated secretions through the nipple and microlesions on the breast skin to epithelial cells that are potentially receptive to HPV infection [5]. Most BCs originate from the mammary duct epithelium that communicates externally through the nipple-areola complex [40].
Cancer arises after a prolonged sequence of events involving oncogenes and tumor suppressor genes, and the HPV can act in specific stages of the carcinogenesis of some cases of BC [7,49]. There is new evidence reinforcing this possibility. Both HPV and its oncoprotein E6 have already been identified in BC. HPV can immortalize human mammary epithelial cells. Hyper-expressed estrogen receptors in BC correlates significantly with high viral loads by HPV. APOBEC3B protein, associated with increased risk of BC, may be positively regulated by an HPV infection [8,24].
While HPV is a well-established pathogen in oropharyngeal, anal, and cervical cancers, it remains controversial in BC [12,17,35]. There are discrepant results in several studies showing an HPV prevalence ranging from 1 to 86% in BC [21]. This variance may be due to inadequate methodologies, choice of less sensitive primers for HPV detection, the use of paraffin-embedded tissue studies that lower DNA integrity, and a geographically wide range of HPV prevalence [4,8]. More recently, a study detected HPV in 30% of intraductal papilloma samples, a result that may encourage HPV research in other benign breast diseases [4].
The aim of this systematic review and meta-analysis is to obtain good quality publication data comparing HPV detection in fresh and frozen BC tissues with benign controls using the polymerase chain reaction (PCR) technique and its variants.
Methods
Search strategy
This manuscript followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [30]. We performed a comprehensive literature review using the following databases: PubMed, Scopus, Science Direct, Cochrane Library, and SciELO. The grey literature was searched using OpenGrey databases (http://www.opengrey.eu/) for unpublished research, and Google Scholar was searched for recent trials not yet indexed. Electronic literature was searched until December 14, 2019, with no language, publication year, or geographical restrictions. A search strategy was performed using the following keywords and Mesh terms: (“Breast Cancer” [All Fields] OR “Breast Neoplasms” [MeSH Terms]) AND (“HPV” [All Fields] OR “papillomaviridae” [MeSH Terms]).
Study selection and eligibility criteria
Two review authors (JEJr and GGO) independently examined each article based on its title and abstract. We initially excluded duplicates reports. Articles whose titles were not relevant were excluded. We also excluded articles whose abstract did not provide knowledge about HPV detection in women with BC. A flowchart with the summary of the study selection process is available in Fig. 1.
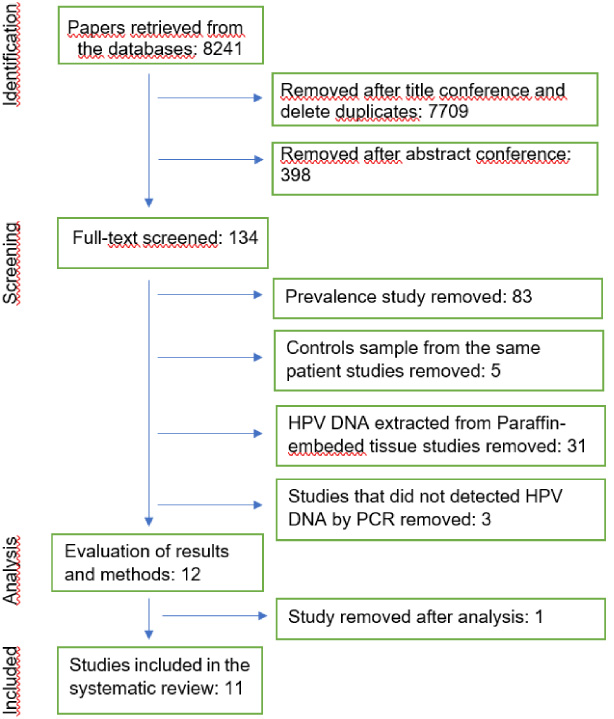
Flow chart of the study selection process.
In the screening phase, the full text of the relevant references was scrutinized, and their content was evaluated by two authors (JEJr and GGO) following the data extraction protocol. The inclusion criteria were case-control studies that detected HPV DNA in fresh and frozen tissue (FFT) by any PCR method. The exclusion criteria were (1) control tissue from the same breast as case tissue, (2) DNA obtained from formalin-fixed paraffin-embedded (FFPE) or cytology, (3) studies that do not report DNA quality testing, (4) cases reports, (5) reviews or systematic reviews articles, and (6) studies without HPV DNA-positivity in both groups. Cases of ductal or lobular mammary carcinoma in situ (CIS) were excluded. Any disagreements during the review were consensually resolved by listed authors.
The analysis phase included a critical evaluation of the results and methods by two authors (JEJr and GGO); any disagreement was solved by consensus. The Cochrane risk of bias assessment tool for nonrandomized studies of interventions (ROBINS-I) [46] was used to assess the risk of bias. The domains evaluated included the following: (1) bias due to confounding; (2) bias in selection of participants into the study; (3) bias in classification of interventions; (4) bias due to deviations from intended interventions; (5) bias due to missing data; (6) bias in measurement of outcomes and (7) bias in selection of the reported result. The categories for risk of bias judgments are “Low risk”, “Moderate risk,” “Serious risk,” “Critical risk”, and “No information on which to base a judgment about risk” of bias. All outcomes analyzed in the systematic review were classified according to the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) [19]. According to the GRADE scale, the overall quality of evidence was graded as “very low”, “low”, “moderate”, or “high”. Disagreements were resolved by consulting a third author (AKG).
Variables of the studies were used to create the following subgroups: authors, year of publication, country; study design; diagnostic method; case tissue type; control tissue type; high-risk (HR) HPV testing positivity; low risk (LH) HPV testing positivity; and mean cases and control age of patients. Data not provided by the articles and required for analysis or inclusion were requested via email from the corresponding author.
Statistical analysis
Statistical analysis and graphical analysis were performed with R© software Version 3.6.1 (R Foundation for Statistical Computing, 2019). Relative risk (RR) and 95% confidence interval (95% CI) were calculated for each article. The presence of heterogeneity in the meta-analysis was assessed using the I-squared (I2) value (%). Low heterogeneity was defined as I2 < 25%, moderate at I2 between 25–50%, and high heterogeneity when I2 exceeded 50% [37]. The fixed effect models were used for low and moderate heterogeneity while the random effect model was used for high heterogeneity. Subgroup analysis and meta-regression were performed to analyze the impact of covariates on the meta-analysis results in case of moderate or higher heterogeneity.
The meta-analysis result was reproduced graphically via a classic forest plot. An Egger’s test and funnel plot were also used to verify possible publication biases [13]. If Egger’s test had a significant result, we used Duval & Tweedie’s trim-and-fill procedure [11]. The Mantel–Haenszel method was used to analyze the data extracted from the selected studies and was combined in this meta-analysis [29]. The DerSimonian–Laird estimator was used to calculate tau square (𝜏2) [9]. A p-value <0.05 was considered statistically significant.
Results
A comprehensive literature review detected 8241 references in databases. Eleven studies met all the inclusion and eligibility criteria after rigorous selection and analysis (Table 1). Two studies [22,47] used both benign and adjacent breast tissue control samples and remained only with control samples from different patients. After applying the selection criteria, 12 studies were analyzed, and one study was also excluded for having used CIS (an email to the authors was not answered). The Forest Plot simulations showed that this study was also an outlier. Two studies also included CIS as BC, but contact via email allowed to exclude these cases from the BC group and keep maintaining these studies as eligible [15,22].
Variables of interest extracted from eligible studies
Variables of interest extracted from eligible studies
Abreviations: PCR: Standard PCR; SH: Southern Hybridization; DB: Reverse Dot Blot; MS-PCR: Multiplex Mass Spectrometry PCR; S: Sequencing; NB: normal breast samples; BBD: benign breast disease; BT: breast tenderness, Multiplex: multiplex-PCR; NG: Not genotyped; NA: Not available.
A critical bias risk analysis via the ROBINS-I tool was performed as described in the methodology. The overall effect of this quality analysis was classified as having a low risk of bias (Table 2). The body of evidence was shown to have a low quality of retrieved evidence via GRADE criteria—while there was an up-grade with a relative risk >2, probable publication bias was detected between studies nullifying this effect (Table 3).
Quality assessment studies and risk of bias using the ROBINS-I instrument
a used benign thyroid tumor specimens as control (not included in our study).
Summary of GRADE evidence
(Very Low ⊕; Low ⊕⊕; Moderate ⊕⊕⊕; High ⊕⊕⊕⊕).
Eligible studies assessed a total of 1857 samples, 1215 cases, and 642 controls. Overall HPV positivity in the case group was 29.5% (n = 359) while only 6.4% (n = 41) of the controls were positive. Standard PCR was used in six studies [20,22,23,32,34,47], and one of these studies genotyped via an INNO-LiPA commercial method [23]. One study used multiplex PCR detected by matrix-assisted laser desorption ionization-time of flight mass spectrometry (PCR-MS) [36], one study used a nested PCR [16], one study used multiplex PCR and sequencing [40], one study combined multiplex PCR and bead-based Luminex technology [14], and a final study used INNO-LiPA commercial kit [15]. The most widely used primer was L1 region MY09/MY11 followed by GP5+/GP6+. Two studies used primer combinations for the same DNA region [16,32]; only one study assessed the two different DNA regions [23].
The meta-analysis for HPV showed a low heterogeneity with I2 calculated at 2% (Fig. 2). The combined risk ratios (95% confidence interval) for detecting HPV among women with and without BC using fixed-effect models were estimated at 2.15 (1.60-2.89), and the p-value was statistically significant (p < 0.0001). Despite the low heterogeneity founded, meta-regression was performed and showed R2 close to zero for all analyzable variables by the method. A Funnel Plot is shown (Fig. 3) exhibiting an apparent asymmetrical distribution. An Egger’s test showed a p-value of 0.0434 suggesting the existence of publication bias. Trim and fill analysis of adjustment for publication bias was applied, and it suggested a combination with five added studies, but there was no significant change in the risk ratio (RR = 1.9375, 95% CI 1.2105-3.1010, p-value = 0.0059). This indicates that the original result was similar to that of the adjusted value, so if there were publication bias in selected studies, this fact did not significantly influence the risk ratio.
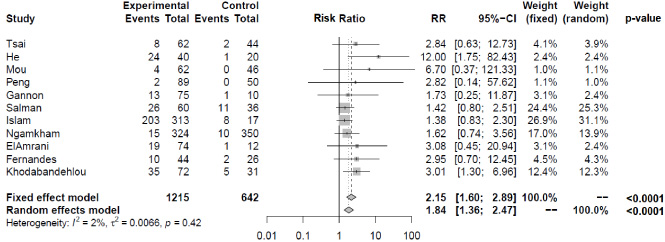
Forest plot of HPV positivity performed in R Studio®. Left: total events in each study, relative to the experimental and control groups; center: graphical representation of RR and 95% CI of each study; right: relative weight of each study when considering a fixed or random effect. Abbreviations: RR, Relative Risk; CI, Confidence interval.
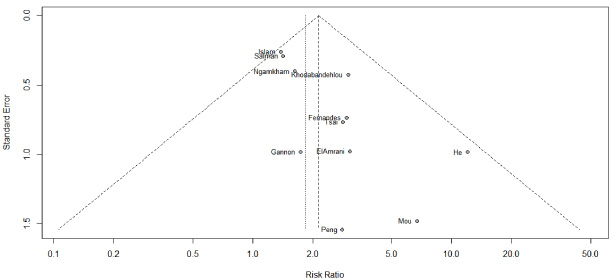
Funnel Plot (Egger’s test with p-value = 0.0434) A Trim and Fill analysis of adjustment for publication bias was applied, and it suggested there was no significant change in the risk ratio (RR = 1.9375).
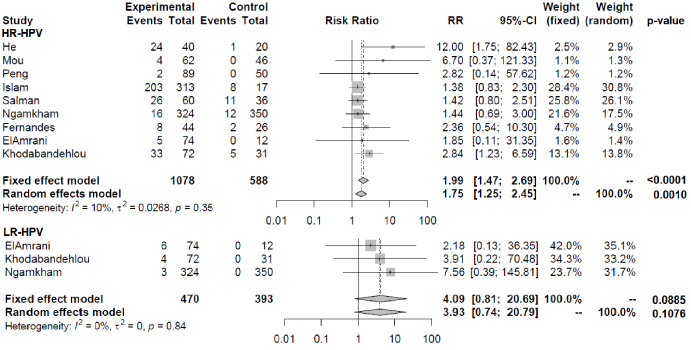
Sub-group by risk type of HPV. Above: Studies that detailed use of HR-HPV test. Below: Studies that detailed the use of LR-HPV test. Left: total events in each study, relative to the experimental and control groups; center: graphical representation of RR and 95% CI of each study; right: relative weight of each study when considering a fixed or random effect. Abbreviations: HR-HPV: High-risk HPV; LR-HPV: Low-risk HPV; RR: Relative Risk; CI, Confidence interval.
HPV 16 was the most common in both cases and controls followed by HPV 18. However, these data could not be subjected to meta-analysis because the two studies did not genotype, and the three others tested specific types. Rather, we divided the findings by HPV risk subtype (Fig. 4). Low-risk HPV was genotyped in three articles and high risk in nine. The relative risk of detecting HR-HPV in BC was 1.9886 (1.4673-2.6950; p < 0.0001). The relative risk for a LR-HPV was 4.09 (0.81-20.69; p = 0.0885).
A meta-analysis on this subject is a very challenging task due to the considerable clinical and methodological heterogeneity among references. Targeting homogeneity, our selection strategy sought studies with more accepted laboratory methods. Therefore, only molecular detection in FFT was included. Detection in FFPE may lead to an increased number of false negatives by the higher possibility of DNA destruction. There is also a greater possibility of false positives due to the higher possibility of contamination during the DNA extraction process [3,50]. Cases of breast carcinoma in situ in the experimental group were excluded due to its uncertain nature regarding the evolution of invasive carcinoma [48].
Two problems permeate HPV detection in breast tissues: no molecular method is universally accepted as the gold standard [45], and there is low HPV viral load in these tissues (2,000-fold less than in cervical cancer) [25]. The most widely used method is PCR. It stands out among other options not only for providing higher sensitivity and specificity of detection but also for requiring small amounts of DNA templates [1]. PCR can be considered the most effective method to detect HPV in BC [51]. For this reason, our meta-analysis was restricted to studies using PCR as a detection technique.
To our best knowledge, this is the meta-analysis on HPV detection comparing tissues with and without BC with the least heterogeneity published. It showed significantly higher HPV detection in BC tissues compared to benign or healthy breast tissue controls. These are consistent with findings in others meta-analyzes [3,6,26,38,43,51]. Except Simões et al. [43], all reviews demonstrated moderate or high heterogeneity. Ren et al. [38] also performed a subgroup analysis with 11 FFT studies (with three different studies by our meta-analysis) and found more heterogeneity, probably due to different search criteria. Another finding was a significant HR-HPV detection in BC but not LR-HPV detection, explained by the few studies about LR-HPV subtypes.
Although data from this and other meta-analyses support higher HPV positivity in BC samples, further studies are needed to establish a causal relationship even if only for some specific neoplastic subtypes. HPV is a ubiquitous virus, and its presence in these tissues may not have a direct etiological relationship and may be the result of comorbidity facilitated by tumor immunological and inflammatory changes. It is also possible that HPV only initiates, facilitates, or participates in the process chain leading to tumorigenesis. HPV infection can, as with cervical cancer, precede malignant transformation by decades [28,43].
Lack of uniformity in control tissues has also been challenging. Some previous studies used benign tissue adjacent to BC as control; similar to Delgado-Garcia et al. [8], we understand this type of study as methodologically inappropriate that can lead to a selection bias. Our control tissue samples were several benign pathologies such as phyllodes tumor, fibroadenoma, and even healthy tissue were included, but this did not appear to influence detection, which was low regardless of the control tissue type. Another point to look for is research that includes recent advances in HPV detection. Recently, new variations of the PCR, such as the Droplet Digital PCR Method and nanoPCR method, have shown better sensitivity and specificity, even with small viral loads, as happens in the breast, but there are no studies with these methods in BC [27,39].
Notwithstanding HPV is not a necessary and sufficient agent for BC, the results of this meta-analysis can be useful to the mastologist thinking about the prevention of this possible causal agent. Because HPV infects the breast through direct sexual contact, a hygienic sexual practice can prevent this infection. Vaccination prophylactic against HPV should also be encouraged. Cytology with HPV testing to detect infection in ductal lavage of the breast, discharge from the nipple, and breast milk could be considered as a potential screening for early diagnosis of BC [21].
Conclusion
This meta-analysis used a selection criterion that sought better methodologies for current knowledge, and once again reinforced HPV’s role in BC. Further studies were needed on how this role is fulfilled, what histological types of cancer are involved, what role HPV plays in precursor lesions, and which HPV genotypes are involved.
Footnotes
Acknowledgements
We would like to thank Dr. Ricardo Ney Oliveira Cobucci, Professor of the postgraduate program in health sciences at the Federal University of Rio Grande do Norte (UFRN), and Ayane Cristine Alves Sarmento, doctoral student in the Health Sciences program at UFRN, for their expertise in assisting this work.
Conflict of interest
The authors have no potential conflict of interest.
Funding
The authors declare there are no grants or other funding for this study.
Authors’ contributions
LGPP, GGO, and JEJr. developed the review protocol. GGO and JEJr. performed the literature search and extracted the data. All authors contributed to writing the manuscript; they also read and approved the final manuscript for publication.
