Abstract
Ectopic breast tissue (EBT) develops as the result of the incomplete embryologic regression of the mammary ridge, which extends bilaterally from the anterior axilla folds to the inguinal folds in the fifth and sixth weeks of gestation. EBT is encountered in 0.3%–6% of women and 1%–3% of men and can be found anywhere along the milk line. It is subject to the same pathologic changes that affect the orthotopic breast. Primary carcinoma arising on the ectopic breast (PEBC) is a very rare occurrence accounting for 0.3%–0.6% of all breast cancers and is most frequently found in the axilla. Due to the rarity and atypical presentation of the disease, the diagnosis is often delayed. We herein present a very rare case of PEBC in the axilla along with a review of the literature. Any subcutaneous hypoechoic nodule located along the milk line without evidence of inflammation should raise concern for ectopic breast carcinoma. Accurate staging and differential diagnosis with detailed immunohistochemical analysis are required. No specific guidelines on diagnosis and treatment are available and the tumor is treated similarly to the orthotopic breast cancer of a similar stage. Physicians should be aware of this rare entity to avoid treatment delays.
Introduction
Ectopic breast tissue (EBT), also known as accessory breast tissue, includes both supernumerary and aberrant breast tissue [1] and is the most common congenital breast abnormality [2]. EBT develops as the result of the incomplete embryologic regression of the mammary ridge, which extends bilaterally from the anterior axilla folds to the inguinal folds, and is encountered in 2% to 6% of females and 1% to 3% of males [3]. It is more frequently seen in Asians, especially Japanese females (5%), than in Caucasians (0.6%) [3].
In a retrospective review Fama et al. [2], found that EBT was observed in 327 (2.7%) out of a total of 12,177 patients attending a breast clinic. EBT is most commonly located in the axilla but it has also been reported in locations outside the distribution of the mammary ridge such as the face, posterior neck, chest, midback, buttock, vulva, flank, hip, posterior thigh, shoulder and upper extremities [4,5]. EBT may consist of breast parenchyma, areola, nipple, or any combination of these three components [6]. The first classification of congenital breast anomalies was published by Kajava in 1915, based on the presence of nipple, areola and, glandular tissue [7]. Although the Kajava classification is commonly used, it is often misquoted because the original classification included six categories [8]. In 2011 Bartsich and Ofodile [9] reported a four type classification of accessory breast tissue in the axilla and proposed a treatment algorithm. In this classification, however, the authors did not mention the accessory breast of the upper arm [9]. Finally, in 2018 Lee et all established a new classification, the Damsoyu-Lee classification in which the axillary accessory breast was classified into three categories, based on its appearance and severity [8].
EBT is subject to the same hormonal influences and pathological changes that affect the orthotopic breast [10–12].
The most commonly reported signs and symptoms of EBT include a palpable mass, pain, tenderness, restriction of arm movement, swelling, and secretion during pregnancy or lactation [5,12]. The most frequently seen benign histological change in EBT is fibrocystic disease [2]. Besides, various tumors have been reported arising from EBT, including benign entities such as fibroadenomas and phyllodes tumors, and malignant tumors mainly adenocarcinomas [12].
The primary adenocarcinoma of ectopic breast tissue (PEBC) is an extremely rare entity. Although it is most frequently seen in the axillary region, it has also been reported to arise in the subclavicular and anterior axillary regions, over the sternum, and in the upper abdominal skin outside the distribution of the mammary ridge [11].
The diagnosis of PEBC is made if intraductal and/or invasive carcinoma is detected in subcutaneous breast parenchyma situated beyond the normal extent of the breast [6,11]. We here describe a case of PEBC in the axilla with a rare clinical manifestation along with a review of the relevant literature.
Case presentation
A 77-year-old woman presented with a 12-month history of small non-tender left axillary mass (Fig. 1). She denied any symptoms. Her medical history was unremarkable and she had no history of breast or ovarian cancer.
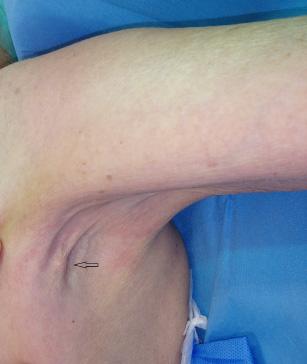
Preoperative photograph showing a small nodule in the midaxillary line. The nodule was visible only after retraction of the overlying skin (arrow).
Physical examination revealed a very small, non-tender hard nodule located in the midaxillary line, measuring approximately 0.7 cm, which was adherent to the overlying skin. The nodule could be seen only when the overlying skin was retracted. There was no palpable axillary and supraclavicular lymphadenopathy. The mammogram was unremarkable. An ultrasound that was performed at another institution was unremarkable for both breasts but had shown a left axillary nodule that had been diagnosed as an axillary lymph node.
A new ultrasound was performed that revealed a hypoechoic irregularly-shaped nodule measuring 0.6 × 0.6 cm suspicious for malignancy (Fig. 2). No breast lesions were noted whereas in the axilla some reactive lymph nodes were also detected. There was no evidence of an ectopic axillary breast mass apart from the aforementioned lesion. Since the nodule was very small a decision was made not to proceed with a core needle biopsy and a local excision was then performed. Histopathological examination of the specimen revealed a primary ductal invasive axillary adenocarcinoma measuring 0.7 × 0.6 cm of grade I according to the Bloom- Richardson grading system. The tumor was found to invade the dermis and the subcutaneous fat. On immunohistochemical analysis, the tumor cells were stained positive for estrogen receptors (ER) and progesterone receptors (PR), whereas the expression of Her2 was negative. The ki-67 proliferation index was 5% (Fig. 3).
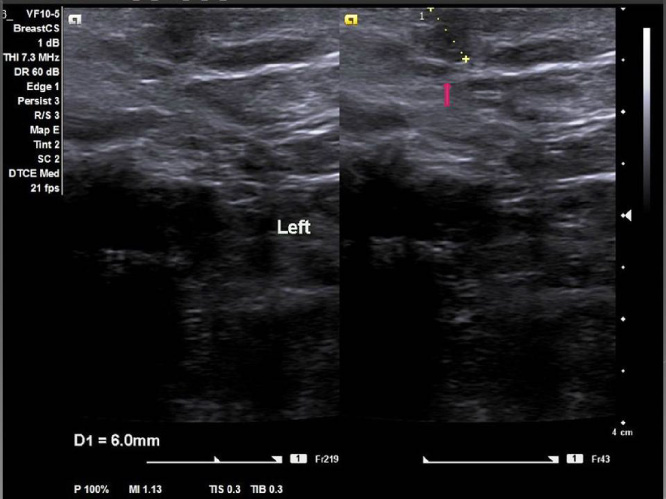
Axillary ultrasound showing a small hypoechoic superficially located mass with irregular margins measuring 0.6 cm (arrow).
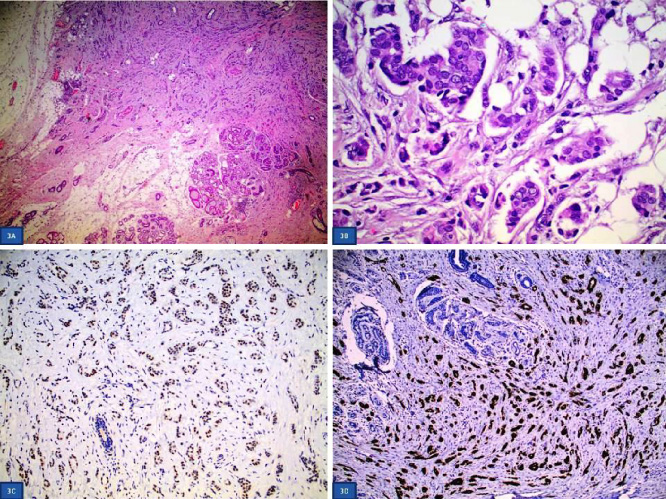
Histopathological and immunohistochemical findings of PEBC. 3A. The tumor cells are medium-sized with weakly eosinophilic cytoplasm and include ovoid, partly slightly irregular nuclei with fine-grained chromatin, small nuclei, and moderate polymorphism-atypia. (H&Ex600). 3B. Invasive carcinoma and breast tissue elements (left). (H&Ex40). 3C. Tumor cells stained positive for Estrogen receptors. (ERx200). 3D. Tumor cells stained positive for progesterone receptors (PRx100).
Following the histological examination, a complete staging investigation was performed including computed tomography scans of the abdomen and chest, and a breast MRI, which were all unremarkable. Sentinel node biopsy was not considered, and an axillary low-level node dissection was performed along with excision of the scar of the previous surgery. The ten axillary lymph nodes removed were free of metastatic disease. Foci of breast parenchyma were also detected. The patient underwent adjuvant radiotherapy and is currently under hormonal therapy with an aromatase inhibitor. She is well, without any evidence of recurrence, 28 months after surgery.
PEBC is a very rare occurrence accounting for 0.3% to 0.6% of all breast cancers [2,10,13], with a small number of cases reported mainly as case reports. The region containing PEBC is most commonly the axilla representing 91.5% of the cases [10].
In a literature review, Visconti et al. [5] found 171 cases of PEBC in the axilla, 6(%) of which occurred in male patients.
PEBC tends to affect patients at a younger age than orthotopic breast cancer. Zhang et al. reported 11 patients with PEBC of whom 8 (72,8%) were premenopausal and 3 (27.2%) were postmenopausal with a premenopausal to postmenopausal ratio of 2.6:1 [12]. The pick incidence occurs between 40 and 45 years with a median age of 51 years [14], which is 10 years less than the median age of patients with orthotopic breast cancer [5]. Rare cases of synchronous development of invasive and in situ carcinomas and bilateral invasive carcinomas with accessory breast cancer have been reported [6,10,15].
The most frequent histologic type of PEBC is ductal carcinoma (72%–79%) followed by lobular, and medullary carcinoma (12%), and other subtypes (16%) [5,16]. Extremely rare cases of Paget disease, cystosarcoma phyllodes, papillary carcinoma, leiomyosarcoma, and secretory carcinoma have been reported [1]. Most patients present with stage II or III disease [12]. The pathological stage for PEBC is the same as that for orthotopic breast cancer [17].
The clinical presentation is of PEBC is that of a palpable immobile axillary nodule with irregular boundaries which is subcutaneously located in 45% of the cases [12]. PEBC does not arise from subcutaneous fat but from mammary gland tissue. However, when the mammary gland volume is small, and the supramammary fat layer is thin, the ultrasonographic findings may suggest subcutaneous fat.
Patients present after 3–24 months period from the onset of the disease [12]. The average tumor diameter at presentation is 2.8 cm [18]. The overlying skin can be normal, erosive, ulcerated, or discolored. Bleeding and discharge may also co-exist [19]. Lim et al. [20], reported a case of PEBC with clinical manifestations of a breast abscess.
PEBC is associated with a more frequent superficial development, being therefore more likely to involve the skin [19], as seen in our case. Axillary metastases have been reported in 46%–51.8% of the patients at presentation [18,20]. Clinical examination should include evaluation of the contralateral breast [10] since in 13% of the cases a bilateral ectopic breast tissue has been reported [15]. In males, the incidence of PEBC is higher than that of the usual form of breast cancer whereas male patients more often present with advanced-stage or inoperable tumors [21].
It is most important to distinguish PEBC from other diseases in the axilla. Main differentials include lymph node metastases, axillary tail breast cancer, lymphomas, metastatic carcinomas, carcinomas of adnexal origin, and benign entities such as fibroadenomas, lipomas, hidradenitis suppurativa, follicular cysts and lymphadenopathies [4,5,12]. Differentiating a PEBC from an axillary sweat gland adenocarcinoma or axillary lymph node metastasis may be difficult [5,10]. A thorough ultrasonographic and mammographic evaluation is essential. Ultrasonographic features such as an irregular hypoechoic mass, increased vascularity and the presence of a suspicious mass or microcalcifications on a mammogram may suggest a PEBC thus requiring adequate tissue sampling for a definitive diagnosis [10]. Specific histological and immunohistochemical findings may suggest a PEBC. These findings may include the presence of primary breast carcinoma in situ, the presence of normal breast tissue adjacent to the lesion, and the positivity of the immunohistochemical markers ER, PR and gross cystic fluid protein GCDFP-15 [5,10]. On the contrary, Her2/neu, CEA, and glandular keratins are not helpful in discriminating a PEBC from a skin adnexal tumor [5].
Due to the rarity and atypical presentation of PEBC the establishment of diagnosis is often delayed. Visconti et al. [5], reported an average delay of 40.5 months in obtaining a diagnosis. The patient described here experienced one-year delay to her diagnosis because in the first ultrasound examination the tumor was initially misdiagnosed as a lymph node.
The diagnostic work-up of PEBC is similar to that of orthotopic breast carcinoma. The triple diagnostic assessment is essential [15]. The mammogram may be unremarkable due to the position of the tumor [13,19], but oblique and exaggerated craniocaudal views may exhibit the tumor [18]. Zhang et al. [12], reported an unremarkable mammogram in 40% of the cases with axillary PEBC. The mammogram may demonstrate findings indicative of malignancy like in the orthotopic breast cancer as well as clusters of microcalcifications requiring further evaluation with a biopsy [5,14,17]. The mammographic findings appear separated from the main breast parenchyma [16].
On ultrasonography, PEBC most commonly appears as a hypoechoic, irregularly shaped heterogeneous mass without evidence of inflammation [12,16]. The tumor may be misdiagnosed as axillary lymphadenopathy, as in the case presented here. The superficial location of the mass along with the presence of accessory breast tissue is indicative of a possible malignancy [12].
Fine needle aspiration biopsy has been used in the diagnostic evaluation of PEBC [22], but the definitive diagnosis can be made only with the histologic confirmation of the presence of benign mammary glandular parenchyma beyond the normal extent of the tumor [11,18].
The imaging findings of MRI have not been clearly defined due to the small number of reported cases. They include the presence of a subcutaneous and poorly defined mass within an accessory breast tissue which is separate from the breast parenchyma showing a hypointense signal on T1 weighted images and an intense signal on T2 weighted images [16,18]. MRI is a useful imaging tool to delineate the tumor extension preoperatively.
After a diagnosis of PEBC is made a thorough staging evaluation is mandatory including chest, abdomen and thoracic CT scan, bone scintigraphy, and breast MRI, according to the existing guidelines for breast cancer management [5].
The treatment of PEBC is similar to that of orthotopic breast cancer including surgery, chemotherapy either in the adjuvant or neoadjuvant settings, radiotherapy, and hormonal therapy.
Wide local excision of the tumor including the overlying skin and regional axillary dissection is the preferred treatment [4,5,11,12,23].
Although the exact role of sentinel node biopsy is unclear [5], it however has been successfully performed in some cases [19,24]. However, the proximity of the injection sites to the axilla may make the intraoperative sentinel node detection very hard by the surgical gamma probe due to the high background [24]. The excision of the primary tumor should be preferably performed before the search of the sentinel node to allow the detection of the sentinel node by the gamma probe [19]. When the primary tumor has already been excised the radiotracer should be injected in both ends of the surgical scar [24,25].
Ipsilateral mastectomy is not required provided that a thorough imaging evaluation has excluded a concurrent breast tumor [5,10–13,23,26,27]. Zhang et al. [12] suggested that if the PEBC is closely located to the normal breast tissue, then the indications for surgery should be similar to those for orthotopic breast cancer. If the tumor is located far away from the breast parenchyma, representing a separate anatomical structure, the additional resection of normal breast parenchyma is not necessary. A long-term follow-up is mandatory especially in patients not undergoing mastectomy [23].
Chemotherapy is used either in the adjuvant or the neoadjuvant settings on the same principles as for orthotopic breast cancer [11,23]. The administration of anthracyclines has been reported to decrease the annual cancer recurrence risk by 2%, whereas taxanes are administered to the patients with positive axillary nodes [27]. The available data for neoadjuvant chemotherapy in locally advanced PEBC are scarce [21].
Hormonal therapy should be offered to patients with a receptor-positive disease [11,19], whereas the use of radiotherapy is controversial [23,27]. Some authors suggest that radiation should be used for the tumor site, the axilla, and the breast, while others suggest the tumor site and axilla, whereas some suggest radiation should only be reserved for the tumor site. Indications for radiotherapy generally depend on the prognostic factors and final staging of the tumor [25].
The prognosis of patients with PEBC is difficult to obtain due to the limited number of cases reported, but it is generally acknowledged that it is similar to matched cases with orthotopic breast cancer [5]. Some authors suggested a worse prognosis since the tumor is located near the axillary lymph nodes thus resulting in early metastases [12]. However, it has not been clearly defined whether the increased incidence of axillary metastases is due to the low proximity of the tumor to the axillary lymph nodes or is the result of delayed diagnosis [16]. Other authors have reported that PEBC does not appear to have a poorer prognosis compared with the orthotopic disease of similar stages. Ninon-Yagani [18], in a retrospective review of 68 of 94 Japanese patients with available data, reported that all but one patient were alive after a median follow-up of 28.3 months (range 2–156 months). In another review of 11 Chinese patients, Zhang et al. [12] reported a median survival of 20 months whereas the 3-year overall survival was 54.5 months. The number of involved nodes has been reported as the strongest predictor of survival [18]. Further research is needed to assess the long-term survival of patients with PEBC.
In accordance with the majority of cases reported in the literature [4,5,12–14,21,23], our patient had an invasive ductal carcinoma of luminal A subtype, that was immunohistochemically characterized by ER and PR positivity, negative expression of Her2, and a low ki-67 proliferation index. The rarity of our case lies in the fact that the tumor affected a 77-year-old patient and it was initially misinterpreted as an axillary lymph node, thus leading to a twelve-month treatment delay. Adjuvant chemotherapy was not considered in our patient. On the contrary, many reported cases required the administration of adjuvant chemotherapy [10,12–14,17,21,23,27]. Zhang et al. reported that all eleven patients with PEBC received adjuvant chemotherapy, whereas local relapse or metastasis occurred in 7 (64%) of the patients [12].
In conclusion, a rare case of a PEBC developed in axillary ectopic breast tissue is presented. The possibility of a PEBC should always be considered in the differential diagnosis of patients females, or males, presenting with hypoechoic superficially located axillary masses without evidence of inflammation. Core needle biopsy or excisional biopsy should be performed to establish an accurate diagnosis. Awareness of the clinicians is needed to avoid a delayed diagnosis with negative consequences. Due to the limited data, the optimal treatment approach has not been clearly defined and is currently similar to that of patients with orthotopic breast cancer.
