Abstract
Bilateral ectopic axillary breast carcinoma is extremely rare. Here, we report the case of a 68-year-old woman who presented with a palpable mass in the right axilla. After ectopic breast carcinoma diagnosis, the patient underwent partial mastectomy and sentinel lymph node biopsy, followed by radiotherapy of the whole breast. Adjuvant endocrine therapy was administered for 5 years. Seven years after the first carcinoma diagnosis, the patient noticed a second tumor in the left axilla by herself at the age of 75 years. Core needle biopsy revealed second primary breast carcinoma of the axilla. She underwent partial mastectomy and sentinel lymph node biopsy followed by chemotherapy, radiotherapy, and endocrine therapy. No recurrence has been observed so far. Therefore, ectopic breast carcinoma should be treated as typical breast carcinoma.
Keywords
Introduction
An ectopic breast tissue can be observed anywhere on the milk line from the axilla to the inguinal region, as a result of non-complete normal embryological development of the breast bud [1]. Primary breast carcinoma may arise from any ectopic breast tissue as accessory breast carcinoma. Accessory breast carcinoma is a rare disease. Furthermore, bilateral accessory axillary breast carcinoma development has been rarely reported. Here, we report the case of a patient with metachronous bilateral ectopic breast carcinoma in the axilla.
Case presentation
In 2010, a 68-year-old woman presented with a mass in the right axilla in the outpatient clinic at Uji Hospital, Kyoto, Japan. The patient was diagnosed with right ectopic breast carcinoma in the axilla and underwent partial mastectomy and sentinel lymph node biopsy. The tumor was invasive breast carcinoma of histological grade 2 (score 3 in tubular formation, score 2 in nuclear atypia, and score 1 in mitotic figures), and did not exhibit apocrine morphology (Fig. 1). Pathological staging was T1c(15mm)N0(0/6)M0. The first tumor was estrogen receptor (ER) positive, progesterone receptor (PgR) positive, and human epidermal growth factor receptor 2 (HER2) negative (immunohistochemistry (IHC) 1+). Radiotherapy of 50 Gy was administered to the right breast over 5 weeks. For adjuvant endocrine therapy, 1 mg of anastrozole was administered daily for 5 years. The patient underwent annual screening after the first surgery, and no abnormality was found until January 2017.
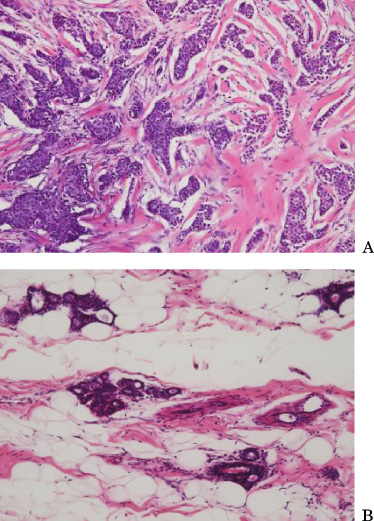
Hematoxylin and Eosin staining for the first tumor (A) and surrounding normal breast ducts (B). The tumor was invasive breast carcinoma of histological grade 2 (score 3 in tubular formation, score 2 in nuclear atypia, and score 1 in mitotic figures). The maximum diameter was 15 mm.
In May 2017, at the age of 75 years, the patient noticed a lump in the left axilla and visited the outpatient clinic at Shiga General Hospital, Shiga, Japan. On physical examination, a 1 cm mass was palpated in the left axilla, but no axillary lymph node was palpable. Mammography and ultrasonography evaluations confirmed an irregularly shaped tumor in the left axilla (Fig. 2 and Fig. 3). The tumor was 7.2 × 8.5 × 6.0 mm in size under ultrasonography. No swollen lymph node was detected. Core needle biopsy revealed that the tumor was an invasive ductal carcinoma of histological grade 2 with 99% ER positivity, 10% PgR positivity, HER2 negativity (IHC 1+), and 70% Ki67 positivity. Breast magnetic resonance imaging showed enhanced irregular tumor in the left axilla (Fig. 4). Computed tomography showed no distant metastasis.
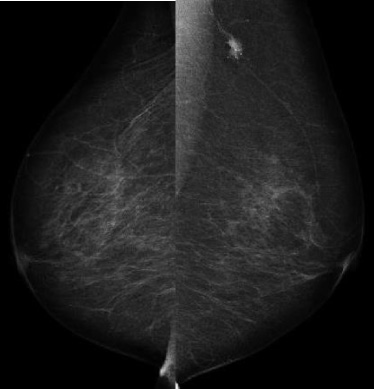
Mediolateral oblique view of mammogram for the second tumor. A spiculated mass was observed on the left upper breast.
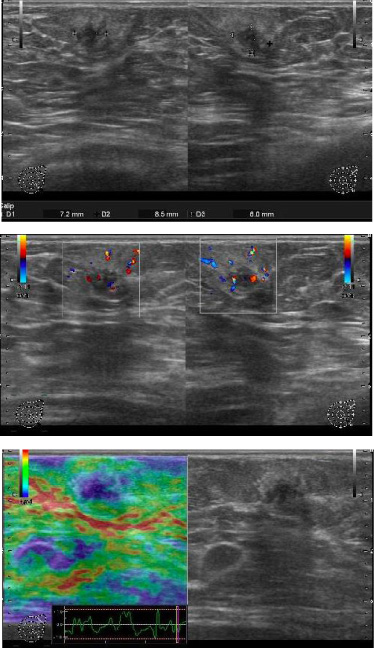
Ultrasonography findings for the second tumor. An irregularly shaped mass was observed on the upper lateral part of the left breast. The size was 7.2 × 8.5 × 6.0 mm. Vascularization was observed around the tumor. The elast score was 5. No mass was found in the two breasts. No axillary lymph node was swollen.
In July 2017, the patient underwent left partial mastectomy and sentinel lymph node biopsy (Fig. 5). Sentinel lymph node was detected using indocyanine green and indigo blue dye. The pathological findings showed that the second tumor was invasive ductal carcinoma (Fig. 6) of histological grade 3 with 95% ER positivity, PgR and HER2 (IHC 1+) negativity, and 70% Ki67 positivity. The tumor did not exhibit apocrine morphology. Pathological staging was T1c(18mm)N0(0/4)M0. For adjuvant chemotherapy, paclitaxel (90 mg/m2) was administered on days 1, 8, and 15, and the cycle was repeated every 4 weeks for 4 cycles. After the chemotherapy, radiation therapy of 50 Gy was administered to the remnant left breast over 5 weeks. For adjuvant endocrine therapy, 2.5 mg of letrozole was recommended for daily administration for 5 years. As of July 2020, no sign of recurrence has been observed.
The patient had a medical history of hypertension and surgical history of thyroid goiter at the age of 26 and lower limb varicose vein at the age of 65. In addition, she had a family medical history in which her eldest daughter and second daughter had thyroid carcinoma at the age of 40 and breast carcinoma at the age of 42, respectively.
The mammary gland develops from the ectodermal layer during embryogenesis. The ectodermal ridge or the milk line which extends from the axilla to the inguinal region appears in the sixth week of gestation. The milk line normally disappears, except for in the thoracic region where the normal breast tissues will develop. However, breast tissues will develop in the ectopic area if the milk line remnants fail to regress during embryogenesis [2]. Ectopic breast tissue is frequently localized in the axilla [3–5]. Ectopic breast carcinoma is a rare neoplasm that accounts for 0.3%–0.6% of all cases of breast carcinoma and has a higher incidence in Asians than in Caucasians [3,6,7]. Modified radical surgery offers no advantage over local excision combined with axillary dissection or radiation with respect to outcomes. Accessory breast carcinoma is surgically treated and administered with preoperative or postoperative chemotherapy, radiotherapy, and endocrine therapy.
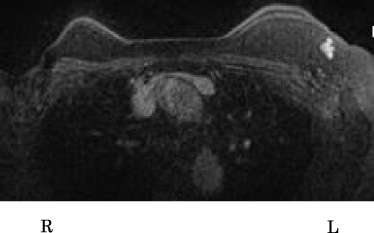
MRI imaging for the second tumor. The MRI showed an enhanced tumor with irregular margins at the level of the left axilla. The size was about 1 cm.
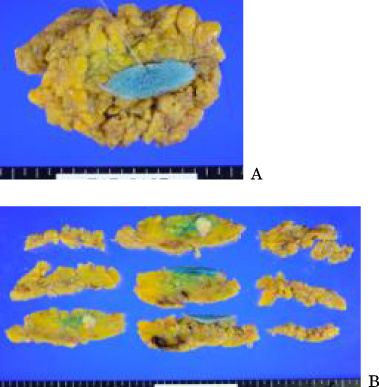
Lumpectomy specimen (A) and the dissection method (B) of the second tumor.
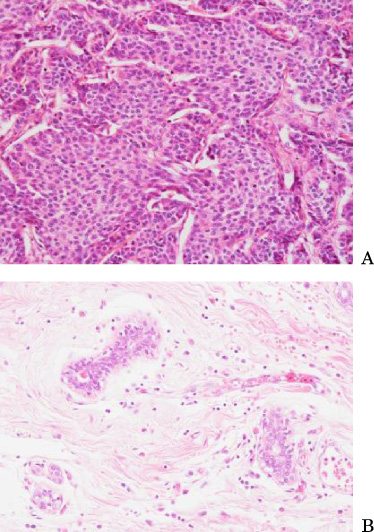
Hematoxylin and Eosin staining for the second tumor (A) and surrounding normal breast ducts (B). H&E staining showed invasive ductal carcinoma with a solid tubular and scirrhous pattern. Histological grade was 3 (score 3 in tubular formation, score 2 in nuclear atypia, and score 3 in mitotic figures). The maximum diameter was 18 mm.
In this case, the patient noticed the lump on both occasions which led to the early diagnosis of carcinoma. Ectopic breast carcinoma was reported to have a poorer prognosis than carcinoma developed in the normal breast parenchyma because early diagnosis is difficult [8]. Visconti et al. calculated the delay was 40.5 months on average from the available data [4]. Physicians should highly suspect primary ectopic breast carcinoma when they see the presence of a subcutaneous mass along the milk line with the clinical features with persistent unilateral growing subcutaneous nodule, irregular, firm and not tender, red and painless. Primary ectopic breast carcinoma should be ruled out first [9]. Ultrasonography is useful for diagnosis. The presence of a hypoechoic, not well-defined heterogeneous mass, without signs of inflammation, is suspicious [10].
The patient received lumpectomy and sentinel lymph node biopsy followed by irradiation to the ipsilateral breast and axilla. Some authors recommend irradiating the tumor cavity, axilla, and ipsilateral breast [11]. Axillary dissection could result in significant morbidity. Since no swollen lymph node was observed before surgery, sentinel lymph node biopsy was selected in this case. Some studies have reported the usefulness of sentinel lymphadenectomy in the treatment of ectopic breast carcinoma [12,13]. The first tumor was of luminal A-like subtype, and the second tumor was luminal B-like subtype, according to immunohistochemistry. The patient was treated according to the breast cancer treatment guidelines of the National Comprehensive Cancer Network (NCCN) and Japanese Breast Cancer Society. No specific treatment protocol is available in the literature for axillary primary ectopic breast carcinoma. Fama et al. reported that ectopic breast carcinoma should be treated as typical breast carcinoma [3]. Furthermore, there is no consensus on whether to excise the ectopic breast tissue prophylactically to prevent malignancy [4].
This patient had a family medical history of breast carcinoma. According to the NCCN guidelines, a close relative diagnosed with breast carcinoma at less than 50 years of age and more than two primary breast carcinomas are criteria for genetic cancer risk evaluation. The patient was referred to undergo testing for the possibility of familial breast carcinoma, but she refused genetic testing.
There has been no sign of recurrence so far, but physicians should continue to follow-up with the patient regularly, and annual mammograms need to be taken. This case suggests that ectopic breast carcinoma should be treated as typical breast carcinoma.
Footnotes
Acknowledgements
The author appreciates Dr. Norio Kageyama and Uji Hospital for providing medical data of the first ectopic breast carcinoma in the axilla. Also, the author appreciates Dr. Yoko Iwasa for providing H&E images and diagnosis.
Conflict of interest
The author declares no competing interests.
