Abstract
Breast carcinoma en cuirasse is a very rare form of cutaneous metastases of breast cancer. The clinical presentation is that of a diffuse indurated carcinomatous infiltration of the skin and subcutaneous tissues of the mammary region and the anterior aspect of the chest. In most cases, breast carcinoma en cuirasse develops post-mastectomy and represents a dramatic presentation of an aggressive tumor associated with a dismal prognosis. Because of the rarity of this type of malignancy, the optimal approach to treatment has not been clearly defined. The systemic treatment has been associated with limited efficacy, and the primary goal is palliative care and preservation of the quality of life through skin-directed therapies. Herein, a very rare case of primary breast carcinoma en cuirasse is presented, along with a review of the literature. Early diagnosis and prompt treatment of any potential skin metastases of breast cancer are essential to prevent the catastrophic natural progression of the disease.
Introduction
Breast carcinoma en cuirasse, also known as scirrhous carcinoma, pahydermia, and acarcine eburnee [18], is a rare manifestation of cutaneous metastases of breast cancer, characterized by the presence of diffuse cutaneous and subcutaneous carcinomatous infiltration of the mammary region that may extend to chest and abdomen [9]. In most cases, breast carcinoma en cuirasse develops after the treatment of the primary tumor, at the mastectomy site. However, in very rare cases it can either be the primary manifestation of breast cancer [12], or the result of the natural progression of untreated breast cancer [18]. We describe here, a very rare case of a primary breast carcinoma en cuirasse and we review the literature about clinical presentation and treatment approach of this aggressive malignancy.
Case presentation
A 60-year old woman presented with an extensive erythematous painful skin lesion of her left breast. She stated that she had noticed a painful mass on her left breast approximately five years ago causing nipple inversion, but she did not seek medical assistance. Two years before her current presentation, she underwent a core biopsy of the lesion at another institution that revealed a grade II invasive ductal breast carcinoma. Immunohistochemical analysis showed positivity for estrogen (ER) and progesterone (PR) receptors, negative expression of HER2, whereas the ki-67 proliferation index was 7%. She then refused any medical treatment for personal reasons. She did not have any history of breast or ovarian cancer.
Clinical examination revealed the presence of an extensive, very hard, shiny, erythematous, and indurated lesion involving the entire left breast causing a significant reduction of its volume. The erythematous lesion was relatively well-demarcated and was extending to the upper left arm and trunk (Fig. 1). On further examination, left supraclavicular and axillary lymphadenopathy associated with lymphedema of the unilateral arm were found. The primary tumor was associated with satellite lesions located on the left arm and left hypochondrium. Deep palpation of the mass was not possible due to the presence of the very hard woody mass fixated to the surrounding tissues. A provisional diagnosis of breast carcinoma en cuirasse was made.
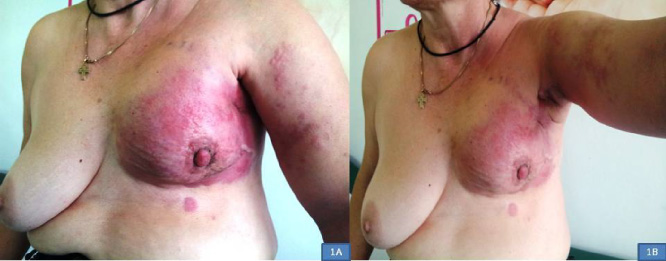
Breast carcinoma en cuirasse. The patient image at presentation showing an extensive, shiny, erythematous, and indurated lesion involving the entire left breast with satellite metastatic lesions of the upper left arm and left hypochondrium. Note the significant reduction of left breast volume and the presence of arm lymphedema.
Staging investigations followed. A bone scan revealed multiple foci of increased radionuclide uptake in the thoracic and lumbar spine and skull. Computed tomography of the abdomen was unremarkable, but the chest scan showed an extensive mass involving the left breast with infiltration of the underlying pectoralis major muscle. Pathologically enlarged mediastinal, supraclavicular and left axillary nodes were also detected, along with pulmonary metastases.
The patient started treatment with bevacizumab in combination with carboplatin and paclitaxel chemotherapy every 14 days along with the administration of zoledronic acid every 28 days. After six months, a significant response of the cutaneous lesions was observed (Fig. 2). She was then placed on bevacizumab with letrozole for one year, followed by a switch of letrozole to exemestane. Sixteen months after the initial presentation a relapse of the cutaneous lesions was observed. A skin biopsy was then performed and revealed that the tumor cells were positive for ER, PR, but there was overexpression of Her2 that confirmed with fluorescent in situ hybridization (FISH). She then received combination treatment with trastuzumab and vinorelbine for three months without clinical response. She was then placed on combination therapy with bevacizumab and capecitabine. At that time the patient was lost to follow-up.
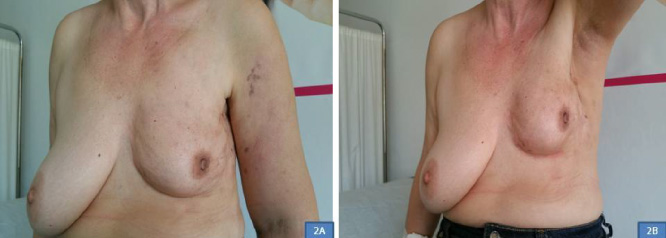
Image showing the clinical response after six months of treatment with bevacizumab in combination with carboplatin and paclitaxel chemotherapy.
Carcinoma en cuirasse was first described by Velpeau in 1938 based on its resemblance to the breastplate worn as body armor by a cavalry soldier [19]. Although carcinoma en cuirasse is most commonly seen in breast cancer patients, it has also been reported in lung, kidney, and gastrointestinal malignancies [12]. It has very rarely reported in male patients [10].
The exact pathogenesis of carcinoma en cuirasse has not been clearly defined [5]. It has been suggested that pleiotrophin, a multifunctional tumor promoter is a causative factor in the transition of cancers to the scirrhous subtype of invasive carcinoma [10].
Cutaneous metastases may result from dissemination of tumor cells via lymphatic and hematic routes and by contiguity. In breast carcinoma en cuirasse the spread of tumor cells is through the lymphatics.
Histologically, carcinoma en cuirasse is characterized by interstitial infiltration of tumor cells in single rows exhibiting a characteristic “single file” pattern in the presence of dense stromal fibrosis [12]. Macroscopically the tumor is characterized by an extensive infiltrated plaque with papules and nodules [6].
Breast carcinoma en cuirasse is an extremely rare presentation of cutaneous metastases of breast cancer accounting for up to 3% of the cases [6]. In a retrospective cohort study including 31 patients with cutaneous breast metastases, only 3.2% presented with clinical manifestations of carcinoma en cuirasse [1].
Carcinoma en cuirasse initially appears as discrete nodules that later enlarge and coalesce forming diffuse sclerodermatic indurations with a well-defined edge that encase the chest wall and abdomen [4]. The tumor surface may be pruritic, painful, ulcerated, bleeding with foul-smelling discharge, and is prone to infections [17,18,20].
Differential diagnoses of breast carcinoma en cuirasse include inflammatory breast cancer, radiation-induced morphea, radiation-dermatitis. The tumor may be misinterpreted as benign dermatitis [4].
Due to the limited number of cases, there is no consensus on optimal treatment. Surgical excision is not feasible due to the tumor’s size, whereas other therapeutic methods such as chemotherapy, hormonal therapy, and radiotherapy are associated with limited efficacy [20].
The treatment approach is aiming at palliation and preserving the quality of life [3]. The treatment approach for cutaneous metastases including carcinoma en cuirasse includes electrochemotherapy, photodynamic therapy, intralesional therapy, hormonal therapy, and local therapies [4,12,17].
Because of the presence of extensive fibrosis and decreased vascularity the chemotherapeutic agents cannot obtain effective concentrations in the tumor area thus making the tumor resistant to chemotherapy [5,8,18]. Resistance to both chemotherapy and radiation can also be due to the impaired microvasculature of the tumor area because of previous surgery or radiation [8].
A meta-analysis of 47 prospective studies including 915 patients with 4313 cutaneous metastases evaluated the efficacy of five skin directed therapies, electrochemotherapy, photodynamic therapy, radiotherapy, intralesional therapy and topical therapy [17].
Among all metastases, the complete response rate was 35.5% and the objective response rate was 60.2%. The objective response rate was for electrochemotherapy 75.4%, for photodynamic therapy 67.8%, for radiation therapy 62.4% and for intralesional therapy 21.4%. Grade >3 toxicity was reported in less than 6% of the cases. Breast carcinoma and melanoma represented 96.8% of the cases of the meta-analysis [17].
Oldenborg et al. [8], studied 196 patients with irresectable carcinoma en cuirasse that were treated with reirradiation and hyperthermia and found that the overall clinical response was 72% (complete 30% and partial 42%). The local progression-free rate was 24% at 1 year. The authors concluded that reirradiation and hyperthermia may provide sustainable tumor control despite refractory extensive tumor growth. The results of the study indicated the superiority of the method compared to systemic treatment in terms of effectiveness and toxicity [8].
Electrochemotherapy (ECT) is the latest therapeutic approach for cutaneous metastases that was introduced in 2006 [2]. It is based on the combination of reversible electroporation caused by the application of an electric field and chemotherapy thus increasing permeability of the cell membrane to chemotherapeutic agents [20]. It is usually used with palliative intent for the treatment of patients with nonresectable tumors.
In a multicenter study, 47 ECT procedures were performed in 38 patients with skin metastases of breast cancer. At 12 weeks after the procedure, 71% showed a positive response and a 29% partial response resulting in significant improvement in the quality of life. Estrogen receptors status and tumor size were predictive of the overall response. The most important factor influencing the effectiveness of ECT is the choice of the right cytotoxic agents. The most commonly used chemotherapeutic agents are bleomycin and cisplatin [20].
In multicenter cohort analysis, including 125 patients with breast skin metastases who underwent electrochemotherapy, the overall response rate after 2 months was 90.2% whereas the complete response rate was 58.4%. In multivariate analysis, the small tumor size, the absence of visceral metastases, the estrogen receptor positivity, and the low ki-67 index was significantly associated with complete response [2]. The one-year local progression-free survival was 86.2%, and 96.4% in the subgroup of patients with complete response. The complete response rate was significantly higher in cancers with luminal A, or B breast cancer subtype compared to triple-negative and HER2 overexpressed tumors [2].
In another multi-institutional study by Campagna et al. [3] that evaluated the efficacy of ECT in 376 patients with superficial metastases, the tumor response rate at 60 days was 88%. The one-year local free-progression survival was 73.7%. In patients with breast cancer, that comprised 8% of the cases of the study, the relevant rate was 89.5%. Tumor size was a predictor of tumor shrinkage [3].
ECT with reduced bleomycin dosages has been reported to be as effective as currently recommended dosages thus making the method suitable in patients with impaired renal function [15].
A retrospective analysis of 201 patients with locally recurrent breast cancer who were treated with contact-free thermograph-controlled w-IRA-HT followed by hypofractionated re –Rt resulted in a high clinical response rate even in patients with large tumors. The authors suggested a novel classification (rClasses 0-IV) that can allow for stratification into distinct prognostic groups [16].
Surgical excision of cutaneous metastases is the preferred treatment with a positive impact on the quality of life [20]. However, it is feasible only in small-sized lesions [6]. Most cases are unresectable due to tumor size, tumor resistance, and poor performance status [3]. Shimozuma et al. [14], reported that in patients with advanced or recurrent breast cancer cutaneous metastases, heavy body weight, and bone metastases had a strong negative influence on the quality of life, whereas endocrine therapy and more extensive first surgery had a strong positive influence on it.
Intravenous bleomycin has been administered with palliative intent in 39 patients with cutaneous metastases with an overall response rate of 66.6% especially in patients with nodules smaller than 2 cm [16].
Correlation of the immunohistochemical findings between the primary breast tumor and cutaneous metastases is mandatory, because any discordant findings may alter the therapeutic approach and the prognosis. Bastard et al. [1], reported that 23.8% of the patients of their study had discordant results for hormone receptors and her2 expression between the primary breast tumor and the metastatic skin tumor.
The prognosis of patients with cutaneous metastases depends on the type and histological characteristics of the underlying primary tumor and its response to treatment [1]. In a retrospective study by Shoenlaub et al. [15], including 228 patients the median survival was 6.5 months. Patients with breast cancer exhibited the best median survival (13.8 months), whereas patients with lung cancer had the poorest prognosis. Survival beyond 10 years was observed in only 3% of breast cancer patients [15]. Survival is also influenced by the age of the patient [11]. The shortest survival has been reported in the group of patients with simultaneous cutaneous and visceral metastases [1].
Despite the reported data suggesting limited efficacy of systemic treatment in the management of breast carcinoma en cuirasse, our patient exhibited a significant clinical response of her dramatic presentation after chemotherapy. The complete clinical response lasted sixteen months. At the time of relapse, the immunohistochemical analysis showed overexpression for Her 2, contrary to the negative Her 2 status at the biopsy that had been performed at the time of presentation. In addition, while the first value of ki-67 index was 7%, the biopsy at the time of relapse revealed an increased new ki-67 index of 20%. We believe that despite the low initial ki-67 index the patient had this dramatic presentation because the tumor had been left untreated for approximately two years.
In conclusion, an extremely rare presentation of breast carcinoma en cuirasse is described. This type of metastatic cutaneous cancer represents a dramatic presentation of the disease and is associated with poor survival and significant impairment of the quality of life. Early recognition and prompt treatment of any potential local recurrence of breast cancer are mandatory to avoid the local progression of the disease. Due to the rarity of the tumor, the optimal treatment has not been clearly defined whereas the current treatment approaches are associated with limited efficacy. Electrochemotherapy appears to be a promising approach in achieving local control of the tumor but needs to be further evaluated. Palliative therapy aiming at the preservation of the quality of life is of great importance.
