Abstract
BACKGROUND:
Transforming growth factor–β (TGFβ) has a dual function in breast cancer, having a tumor suppressor activity in early carcinomas while enhancing tumor metastasis in advanced breast carcinoma. Consequently, the prognostic role of TGFβ and its signaling cascade in breast cancer remain unclear.
OBJECTIVE:
To investigate the relationship between TβRII expression, clinic-pathological characteristics, and prognostic significance of TβRII expression in clinical stage III breast cancer.
METHODS:
Biopsy from the primary tumor was obtained from 30 newly diagnosed clinical stage III breast cancer patients before receiving any therapy. Expression of TβRII, ER, PR, Her2 and Ki-67 was assessed by immunohistochemistry.
RESULTS:
TβRII expression was positive in 66.7% of cases and was significantly associated with advanced nodal stage and distant metastases. After a median follow up of 42.3 months, TβRII was associated with poor disease-free survival and it was an independent factor for predicting the poor outcome for breast cancer patients, especially in node positive tumors, ER/PR positive and Her2-negative tumors.
CONCLUSIONS:
These findings suggest the usage of therapeutic drugs that target TGFβ in advanced breast cancer patients may be effective. Nevertheless, blockage of the tumor promoting and sparing of the tumor suppressor effect of TGFβ pathway should be taken into consideration. We suggest that these therapies might have more benefit in ER and PR positive tumors.
Introduction
Breast cancer is the most common diagnosed neoplasm worldwide. The highest incidence rates are reported in North America, Australia/New Zealand and western and northern Europe, while the lowest are reported in sub-Saharan Africa and Asia [1]. Similarly; the most common cancer in Egyptian women is breast cancer (32.04%), which is also the second most common cancer in both sexes in Egypt (15.41%) [2]. Breast cancer mortality in Egypt is 29% with 3.7:1 incidence to mortality ratio [3].
Transforming growth factor-β (TGFβ) is one of the most important regulators of development and function of normal breast tissue, as well as breast tumors. TGFβ has been proposed to play a dual role in breast cancer, initially acting as a tumor suppressor [4]. Nevertheless, cells become progressively resistant to the tumor suppressor effects of TGFβ with advanced stages of breast cancer development, and it then acts as a tumor promoter. So, TGFβ is thought to enhance tumor progression in advanced breast cancer stages with stimulating tumor cell motility, invasiveness and capacity of metastasis [5,6]. Two transmembrane receptors mainly mediate TGFβ signaling; TGFβ type I receptor (TβRI), and TGFβ type II receptor (TβRII). They act by phosphorylating and activating SMAD2 and SMAD3 after ligand binding to the receptors [7].
There is conflicting data in regards to the prognostic role of the TβRII expression in breast carcinoma [8]. Some studies have demonstrated that high TβRII expression was associated with good prognostic factors as low-grade tumors, decreased tumor size and ER positive status [9,10]. In addition, decreased TβRII expression in ER-positive tumors was reported to be associated with resistance to hormonal therapy [11]. Conversely, other studies reported that TβRI and TβRII expressions were associated with poor prognostic factors as advanced stage, LN metastasis and shortened survival [12,13]. This controversy was explained by several reasons including heterogeneity of patient populations in various studies, differences in sample sizes, and method/cut off of evaluating receptors expression [13]. Interestingly, most previous studies investigating the role of TGFβ expression in breast cancer formation and progression assessed TβRI and little information is available in regards to other isoforms particularly TβRII expression in breast cancer [14].
So, we aimed at the current study to investigate the rate of TβRII expression, its relationship to various clinico-pathological characteristics, and its prognostic significance in advanced breast cancer patients.
Material and methods
Patients and tissue samples
Thirty newly diagnosed, histologically confirmed, clinical stages III breast cancer patients (according to the AJCC TNM staging system 7th edition) were included into the study. They presented at the outpatient clinic of medical oncology unit at Oncology Center, Mansoura University between April 2011 and April 2015. Biopsies were obtained from the primary tumors of all patients before receiving any therapy and preparation of slides was done as previously described [14]. Histological typing was done according to the latest WHO guidelines [15]. Lymph node ratio (LNR) was defined as the ratio of positive lymph nodes to all excised lymph nodes [16]. The median follow up was 42.3 months (Range from 2.7 to 76.1 months).
Immunohistochemistry
Four micrometer sections were cut from the paraffin block of each tumor. Slides were baked for 30 minutes at 55 °C, deparaffinized then rehydrated. Blockage of endogenous peroxidase was done by appling 0.3% hydrogen peroxide methanol for twenty minutes. EDTA was then used at maximum microwave power for ten minutes for induction of antigen retrieval. Sections were incubated overnight with polyclonal rabbit anti-human TβRII (sc-400; Santa Cruz), monoclonal mouse anti-human ER (sc-8002; Santa Cruz), monoclonal mouse anti-human PR (sc-166169; Santa Cruz), monoclonal mouse anti-human Her2/neu (sc-33684; Santa Cruz) and monoclonal mouse anti-human ki67 (sc-23900; Santa Cruz). Slides were finally incubated for thirty minutes with secondary mouse Envision (DAKO).
IHC scoring
A semi-quantitative scoring method was used to evaluate TβRII staining according to the percent of positively-stained cells as previously described [13]. A score of 0 was used if less than five percent of the cells were stained. A score of +1 was used if 5–20% of tumor cells were positive. A score of +2 was used if more than 20% of tumor cells were positive. Finally, scores 0 and +1 were considered negative, while +2 score was considered positive for TβRII expression. ER and PR expressions were interpreted using Allred scoring system as previously described [17]. Percent of positive cells was classified into five categories (score 0 for totally negative tumor cells, score 1 if <1% of tumor cells were stained, score 2 if 1%–10% of tumor cells were stained, score 3 if 11%–33% of tumor cells were stained, score 4 if 34%–66% of tumor cells were stained, and score 5 if 67%–100% of tumor cells were stained). Furthermore, the intensity of staining was categorized into 3 categories (0 for absent staining, 1 for weak staining, 2 for moderate staining, and 3 for strong staining). Summation of both scores devided the cases into 2 groups (negative staining for 0–2 score, and positive for score 3–8). HER2 expression was evaluated according to the American Society of Clinical Oncology/College of American Pathologists guidelines for HER2 testing in breast cancer as previously described [18]. Tumors with scores 0 or +1 were considered negative, +2 cases were considered equivocal, and cases with score +3 were categorized as positive. In regards to Ki-67 expression, a cutoff point of 14% was used to divide tumors into positive and negative groups (more and less than 14% of tumor nuclei positivity respectively).
Study endpoints and evaluation of outcome
The primary objective was to assess the disease-free survival (DFS) which is the time from surgery to time of recurrence either local or systemic in clinical stage III breast carcinoma patients. The secondary objective was to assess the association between TβRII expression and the clinico-pathological characteristics. Overall survival (OS) was also measured from the date of diagnosis to time of either death of the patient or the date of last contact in patients last known to be alive who censored.
Statistical analysis
Descriptive statistics including the mean (±SD) or median (range) were applied as appropriate for analysis of continuous variables “Shapiro–Wilk normality test was applied in order to find out if the values of the continuous variables were normally distributed” and frequency (%) was used for analysis of categorical variables. Mann–Whitney or Student’s-t tests were used for comparison of continuous variables between groups, while categorical data was compared between groups by using Chi-square and Fisher’s exact tests. Correlations were done using either Pearson’s or point-biserial correlation tests as appropriate. The differences were considered significant at P < 0.05. The probability of DFS and OS was defined with Kaplan–Meier’s method, and differences were compared with the log-rank test. Follow-up was defined from enrollment to the last follow-up. All statistical computations were performed using SPSS Software version 20 (IBM Corp., Armonk, New York, 2011).
Results
Patient’s and tumor characteristics
The mean age of patients was 54.9 (SD ± 12.6) years. Twenty-five cases (83.3%) had infiltrating duct carcinoma. High-grade histology (Grade III) was found in 25.0% (6 cases) of patients. The median largest primary tumor diameter was 3.5 cm (range 1.5–10). Lympho-vascular invasion was present in 25.0% (7 cases) of patients. ER was positive in 70.0% (21 cases) of cases (Fig. 1a). PR was positive in 63.3% (19 cases) of cases (Fig. 1b). Her2neu was positive in 26.7% (8 cases) cases. Ki-67 was positive in 46.7% (14 cases) of cases (Fig. 1c). Luminal B subgroup was the most common type (46.7%; 14 cases) (Table 1).
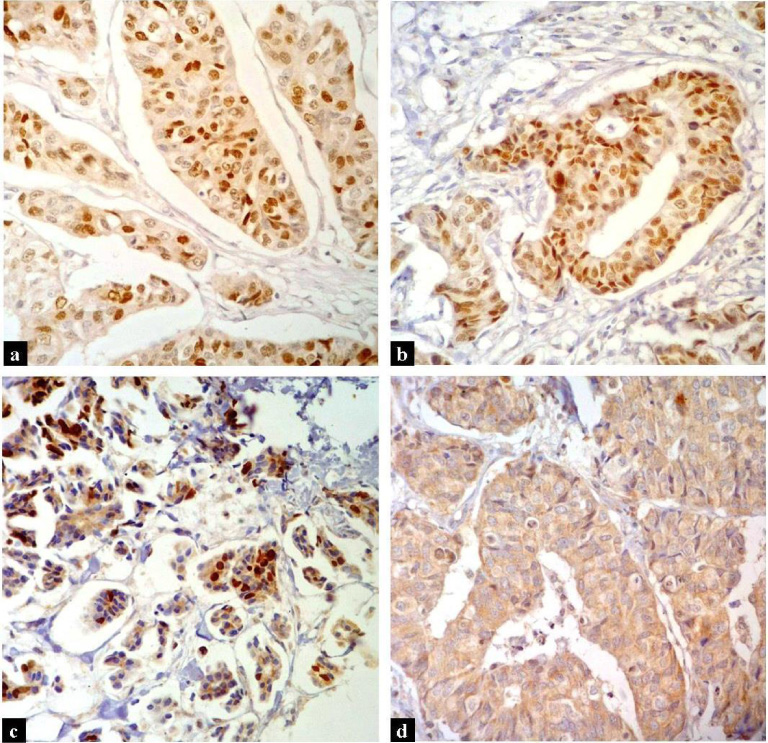
(a) Strong ER nuclear staining in 67–100% of tumor cells (×200) (b) Strong PR nuclear staining in 67–100% of tumor cells (×200) (c) Strong Ki-67 nuclear staining in >14% of tumor cells (×200) (d) Moderate TβRII expression in >20% of tumor cells (×200).
Clinicopathological features of the patients
Seventeen cases (56.7%) developed recurrence either local or systemic. Of these patients, 29.4% (5 cases) had local recurrence while 70.6% (12 cases) had systemic recurrence. Bone metastasis was the most frequent pattern of systemic recurrence in 41.2% (7 cases) followed by hepatic and brain metastasis in 17.6% (3 cases) then lung and/or pleural metastasis in 11.8% (2 cases) (Table 1).
TβRII expression was positive in 66.7% of patients (20 cases) (Fig. 1d). TβRII expression was significantly associated with advanced nodal stage. In patients presented with N2 or N3 stage, TβRII expression was positive in 85.7% (12 out of 14 patients), while in patients presented with N0 or N1 stage, TβRII expression was positive in 50.0% (8 out of 16 patients), and this was statistically significant (p = 0.038).
TβRII expression was significantly associated with development of distant metastasis (p = 0.037). In patients who developed metastasis, positive TβRII expression was detected in 82.4% of cases (14 out of 17 patients), while it was positive in 46.2% (6 out of 13 patients) of patients who did not developed metastasis (Table 2). In regards to specific metastatic sites, TβRII expression was positive in 71.4% (5 out of 7 patients) of patient who developed bone metastasis, 100% (3 out of 3 patients) of patient who developed brain or hepatic metastasis and 100% (2 out of 2 patients) of patient who developed lung and/or pleural metastasis (Table 2).
Association of TβRII expression in clinically stage III cases with tumor pathological characteristics and IHC markers
Association of TβRII expression in clinically stage III cases with tumor pathological characteristics and IHC markers
∗ P ≤ 0.05 is significant.
TβRII expression was not statistically associated with increased age, post-menopausal patients, large tumor size, advanced tumor stage, high lymph node ratio and lympho-vascular invasion (Table 2).
TβRII expressed more in ER positive, PR positive and Her2neu positive tumors. TβRII was positive in 71.4% of ER-positive tumors, 68.4% of PR-positive tumors and 75.0% of Her2neu positive tumors, however this was not statistically significant (p = 0.431, 1.000 and 0.682 respectively). On the other hand, TβRII expression was not associated with Ki-67 expression nor molecular subtypes (p = 1.000 and 0.846 respectively) (Table 2).
Relation of TβRII expression to survival of the patients
After median follow up with 42.3 months, the median DFS of clinical stage III TβRII-positive cases was significantly lower than TβRII-negative cases. Three and four years DFS was significantly prolonged in TβRII-negative (76% and 59% respectively) than in TβRII-positive cases (24% and 5%) (p = 0.003). However, 5 years OS was 100% for TβRII-negative and 83% for TβRII-positive cases, which was not statistically significant (p = 0.297).
By subset analysis, TβRII expression was associated with a significant decreased DFS in node positive tumors (p = 0.007) (Fig. 2a), ER-positive tumors (p = 0.012) (Fig. 2b), PR-positive tumors (p = 0.035) (Fig. 2c) and Her2neu-negative tumors (Fig. 2d) (p = 0.036).
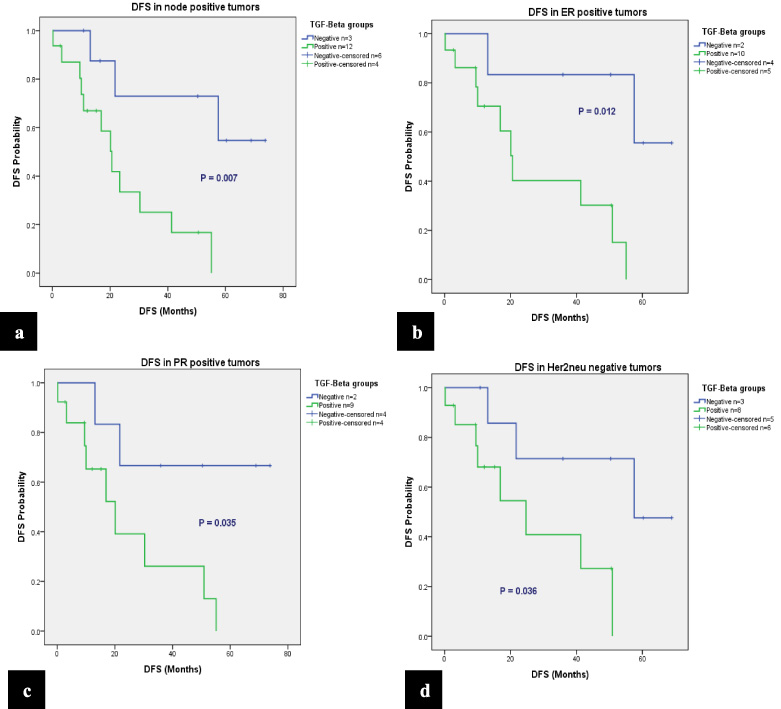
Disease free survival of TβRII negative and positive clinically stage III in: (a) Node positive tumors (b) ER-positive tumors (c) PR-positive tumors and (d) Her2neu-negative tumors.
TβRII expression was the only statistically significant factor that decreases the DFS in univariate analysis (HR = 7.215, p = 0.011). Although age above 50 years, lympho-vascular invasion, N2 and N3 lymph node stage, PR and Her2neu positive status have poor effect on DFS (HR = 1.748, 1.018, 1.927, 1.332, 1.225 respectively), this effect was not statistically significant (Table 3).
Univariate analysis of DFS in clinically stage III breast carcinoma cases
∗ P ≤ 0.05 is significant.
Breast cancer is the most common cancer in Egyptian women (32.04%) and the second most common cancer in both sexes (15.41%) [2]. TGFβ signaling controls many features of tumor evolution and metastasis. TGFβ has a pleiotropic effect which may be a tumor suppressor or tumor promotor according to cellular context and the course of the disease [19].
Till now, there is a contradictory data as regards the prognostic role of the TβRII expression in breast carcinoma. Some previous studies have demonstrated that high TGFβ expression was associated with good prognostic factors and better survival, in contrast to others [11]. Interestingly, most studies on the role of TGFβ in cancer formation and progression assessed TβRI and little information is available in regards to TβRII [14]. Therefore, we conducted this prospective randomized study to investigate the relationship between TβRII expression in locally advanced breast cancer patients (clinical stage III) and different clinico-pathological features, established biomarkers, and patient outcome.
Our results revealed that TβRII expression was positive in 66.7% of patients with clinical stage III breast cancer, denoting that TβRII is overexpressed in advanced breast cancer cases. This result is in harmony with the data of Wakefield and Roberts who reported that TGFβ signaling pathway acts as a tumor-promoter in later stage of human malignancies, which is associated with an aggressive tumor behavior and distant metastasis [20]. In concordance, we found that TβRII-expression was significantly associated with advanced nodal stage. Gao and colleagues also reported the significant association of TβRII expression with advanced tumor stage and lymph node metastasis [12]. A unique finding at the current study is the assessment of the nodal stage by further calculating the lymph node ratio (LNR) and correlating it with TβRII expression. TβRII expression was also associated with high LNR, but this was not statistically significant.
In the current study, TβRII-expression was also found to be associated with less lympho-vascular invasion. However, this relation just approaches significance in the current study, these results are in accordance with Dave and colleagues who correlated lymphatic permeation with the serum protein level and mRNA of TGFβ2 in naive breast cancer patients. They observed that TGFβ2 negativity was also higher with lymphatic permeation in advanced stage patients and explained this by the tumor suppressor effect of TGFβ [21]. We also reported that TβRII-expression was significantly associated with development of distant metastasis, especially bone, brain, liver, lung and/or pleural metastases. This is further emphasizing the association of TβRII-expression with bad prognostic factors. Other investigators as Buck and colleagues failed to find such associations with nodal status and distant metastasis [15]. However, in line with our results, they reported that TβRII expression showed no statistically significant correlations with age, menopausal status, primary tumor size, histological types and histological grade. Regarding tumor size, we found that TβRII-expression was positive in 50.0%, 65.0% and 83.3% of T1, T2 and T3 tumors respectively, and although this was not statistically significant, it reflects the dynamic expression of TβRII-expression with tumor progression.
No significant relation between TβRII expression and ER, PR, Her2neu and Ki-67 status was found in the current study. Gao et al. and Hachim et al. also observed that TβRII expression was not significantly associated with ER, PR and HER2 expression levels [12,22]. Furthermore, we assessed TβRII expression in relation to different breast cancer molecular subtypes and found, in concordance to Hachim et al. [22], that TβRII-expression was expressed more in luminal A, luminal B and less expressed in her2neu enriched and basal type, however this was not statistically significant.
In regard to patient outcome, we observed that high TβRII expression was associated with statistically significant poor DFS. In subgroup analysis, TβRII expression was associated with a significant decreased DFS in node positive tumors, ER-positive tumors, PR-positive tumors and Her2neu-negative tumors. These results are exactly in agreement with the data of Gao et al. [13]. Conversely, other studies reported that patients with low TβRII mRNA levels had significantly shortened DFS [22]. They explained that by the tumor suppressor role for TGFβ signaling in the early stages of breast cancer. However, we focused at this study on advanced stage breast cancer patients.
Numerous studies have focused on the association between ER and TGFβ signaling. Previous studies stated that ER suppresses breast cancer metastasis by inhibiting TGFβ signaling [23]. Others observed that patients with ER-negative tumors with TβRII overexpression have been associated with reduced OS [24]. Conversely, we observed that patients with ER-positive tumors that expressed TβRII were associated with a significant shorter DFS.
There may be some possible explanations behind the contrasting results of the current and previous studies, such as the sample size, different races of studied patient population with varying clinico-pathological features including tumor histological type, tumor grade, clinical stage, type of adjuvant therapy and others. Nevertheless, the procedures of assessing TβRII expression by immunohistochemistry are different in various studies. Moreover, the cut-off values that consider the positive expression affect the study results. Different evaluation methods of TβRII expression can also result in the inconsistency found in different studies. Therefore, a better investigational design is needed for further studies. Furthermore, we believe that due to the small sample size at this pilot study, some of our results approached - but not reached - statistical significance. So, we recommend further functional studies with large sample size to validate our results about TβRII expression in advanced stage breast cancer.
In summary, our results indicate that high TβRII expression is associated with poor clinico-pathologic features in advanced breast cancer. This finding suggests the usage of therapeutic drugs that target TGFβ in advanced breast cancer patients may be effective. Nevertheless, blockage of the tumor promoting and sparing of the tumor suppressor effect of TGFβ pathway should be taken into consideration. In addition, expression profile of other ErbB receptors such as Her-3 and Her-4 should be also taken into account in predicting responsiveness to potential therapeutic targeting of TGFβ in advanced breast cancer patients as trastuzumab failure in some breast cancer patients may partly be attributed to co-expression of other ErbB receptors such as Her-3 and Her-4, these receptors should be considered more than HER-2/neu heterodimerization partners [25]. We suggest that these therapies might have more benefit in ER and PR positive tumors.
Conflict of interest
The authors declare that they have no conflict of interest. This work had no specific funding. There were no financial disclosures from the author.
Author contribution
All authors have contributed significantly and are in agreement with the content of the manuscript.
