Abstract
The differential diagnosis for benign breast masses is broad and ranges from common lesions like fibroadenomas to rare masses like breast hamartomas. Fibroadenomas are proliferative benign masses made up of fibroglandular tissue. Hamartomas are neoplasms comprised of different tissues that are endogenous to the area where they originate. Breast hamartomas specifically, are rare, benign slow growing tumours comprised of fibrotic stroma, adipose, glandular tissue, and epithelial components. Both lesions are painless, firm, and are typically palpable on clinical exam. Given their similarities in composition, diagnosing these masses can be challenging, but may be confirmed with ultrasonography, mammogram, computed tomography, magnetic resonance imaging, or via histological specimen. Once diagnosed, surgical excision is the preferred treatment option. We present a 33-year-old woman with a large left breast mass that gradually increased in size and provide a review of the current literature regarding the challenge of distinguishing between breast fibroadenomas and hamartomas.
Introduction
Fibroepithelial masses of the breast encompass a broad spectrum of tumors ranging from common, benign fibroadenomas to rare lesions. Discerning between these entities can be challenging as both lesions have similar compositions, including glandular breast tissue, lobular fat, and fibrous stroma. For this reason, the incidence of breast hamartomas may be underestimated as they may be mistaken for other benign breast diseases like fibroadenomas or sclerosing lobular hyperplasia [17].
In this report, radiologic features on multiple imaging modalities (ultrasound (US), mammography, computed tomography (CT), and magnetic resonance imaging (MRI)) are described. Clinical photos taken pre-, intra-, and post-operatively are also included. Informed consent was obtained from the individual participant in the study.
Case presentation
A 33-year-old female presented with a 3-year history of a painless breast mass in the inferior pole of her left breast. The lesion was initially discovered by the patient on self-examination. There was no history of pain associated with the mass and it was not tender during her menstrual cycle. The mass was initially small and mobile, but progressively increased in size over time. The patient was otherwise healthy and had no history of breast-related trauma, radiation, previous surgeries, or previous breast lesions. She was taking oral contraceptive pills (OCPs) regularly for 12 years but stopped OCPs one month prior to her surgery. There was a maternal family history of breast cancer as the patient’s mother was diagnosed with breast cancer when she was 45 years old. The patient’s father had stage 3 lymphoma (diagnosed at 48 years old) and the paternal grandmother had a history of gastric cancer. No prior genetic testing for BRCA 1 or 2 had been performed.
On physical examination, there was a palpable, mobile, well-circumscribed mass that was approximately 8 × 12 cm in size located in the inferior pole of the patient’s left breast. Given the position and size of the mass, the skin envelope of the left breast had greater dimensions and laxity compared to the right breast. There were no signs of skin tethering and the mass was not fixed to the overlying skin, the underlying breast tissue, or chest wall. There were no abnormalities associated with the nipple areolar complex and the patient did not have any palpable axillary lymphadenopathy (Fig. 1).
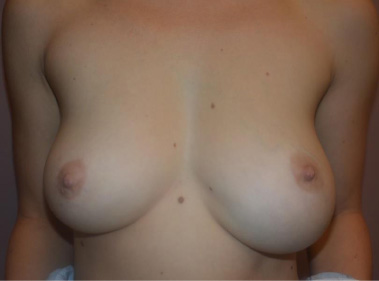
Preoperative photo (without markings). Note the increased volume and skin envelope of the left breast.
Given the increase in size and positive family history, the patient was concerned and had a previous diagnostic work-up at another hospital. According to the patient, a mammogram, CT, and biopsy were ordered and a fibroadenoma was reported. At the time, the patient decided to forego surgery as she was told that the excision would have resulted in a large defect/indent and significant breast asymmetry. Three years later, however, the patient had another CT scan, this time to further investigate a suspicious right upper lobe lung lesion seen on plain film x-ray. There was no significant lung disease, but the scan revealed a cluster of at least 10 oval shaped nodules within the lower pole of the patient’s left breast at the same location of the previously biopsied fibroadenoma. Initial reports described a discrete lesion that could be either a fibroadenoma or a giant hamartoma and suggested further investigation via ultrasound. At the time, the lesion measured 10.3 cm transversely × 7.1 cm craniocaudally × 4.8 cm on the anterior-posterior view. There were no appreciable adenopathies (Fig. 2).
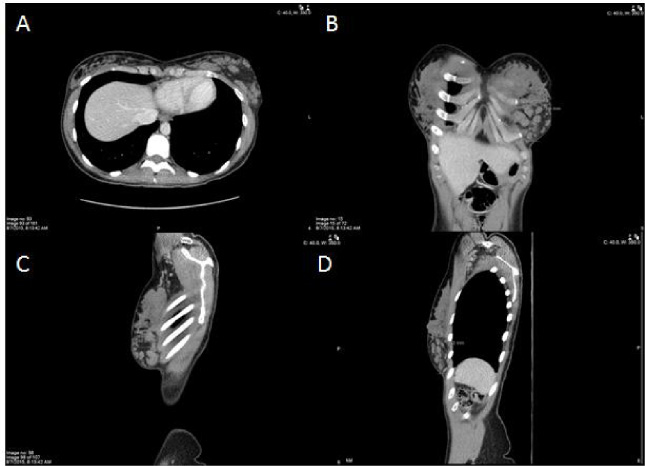
CT scan of the left breast in 2015 (a) axial view; (b) coronal view; (c, d) sagittal views.
Contradictory to previous findings, imaging by ultrasound, mammogram, and a dynamic gadolinium-enhanced MRI all revealed a giant solitary breast hamartoma. The mass was now a 12.6 × 7.7 × 2.2 cm. On ultrasound, the mass specifically had multiple circumscribed hypoechoic nodules scattered among lobules of adipose tissue (Fig. 3). On MRI, a large breast hamartoma composed of fat and breast tissue with increased density was reported (Fig. 4). A repeat CT scan further confirmed that the mass was a smooth-margined giant solitary hamartoma made up of soft tissue nodules interspersed among macroscopic fat (Fig. 5).
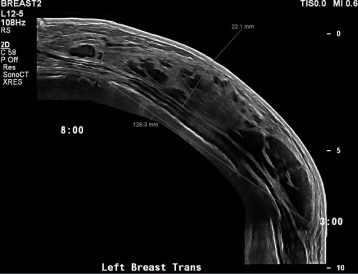
Ultrasound of the left breast (2018).
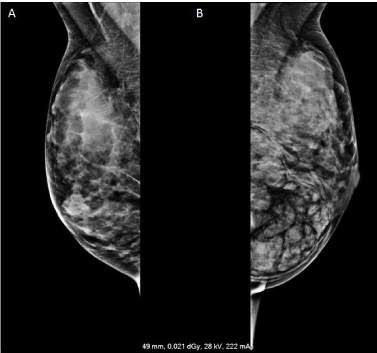
Mammogram comparing the (a) right and (b) left breasts.
Given the current size of the breast mass and its progressive growth, surgical excision of the mass was again offered. She was referred to our service by a General Surgeon/Breast Oncologist as they feared a disfiguring postoperative indent if removed in the usual lumpectomy fashion.
On the day of the surgery, preoperative markings were outlined in standing position. The right, unaffected breast inframammary fold-to-nipple distanced was measured as 7.5 cm. This distance was transferred to the larger left breast, measuring from the nipple inferiorly and marking 7.5 cm on the lower pole of the breast within the breast meridian. This mark was 2 cm superior to the left sided inframammary crease and a 2 cm crescent excision of skin was marked (Fig. 6).
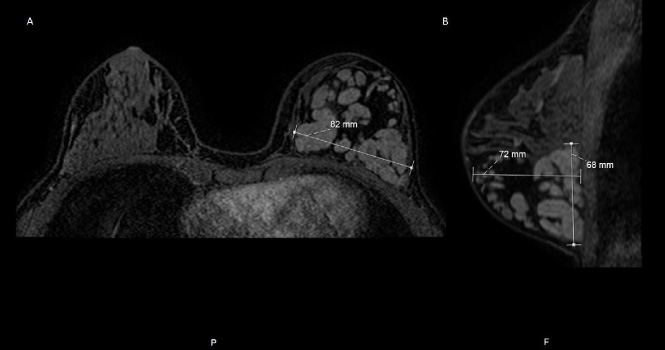
Repeat CT scan (a) axial view of bilateral breasts, comparing the left breast hamartoma with the right normal breast tissue; (b) sagittal view.
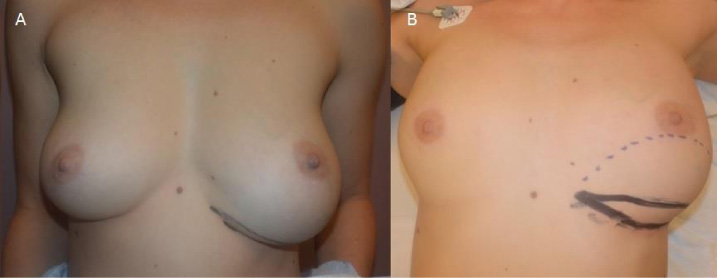
Pre-operative photos (with markings) in (a) standing position and in (b) supine position.
The superior aspect of the marked crescent was incised, and a superiorly-based skin flap was raised. During the dissection, the breast mass was easily identifiable and found to be encased within a pseudocapsule, which was visibly distinct from the surrounding tissue. The pseudocapsule around the mass was thin and was not fixed to any of the underlying structures. It was smooth, gray-pink in colour, and had varying areas of soft and firm lobules. The mass was circumferentially dissected, removed, and sent to pathology in a 10% neutral buffered formalin (Fig. 7). The crescent of skin within the lower pole of the breast (as described above) was then de-epithelialized to add volume to the inferior pole of the breast, partially replacing the excised mass. The remaining superior breast and skin was then advanced inferiorly to overlie the de-epithelialized crescent and the incision/scar was hidden within the inframammary crease. The 2 cm crescenteric skin excision also tightened the skin envelope to better match the contralateral right breast and aided in correcting the resultant defect after the mass was removed (Fig. 8).
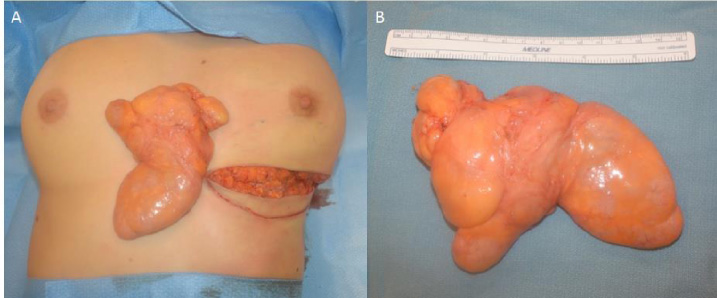
Hamartoma excised and compared relative to (a) bilateral breasts and to (b) a 15 cm ruler.
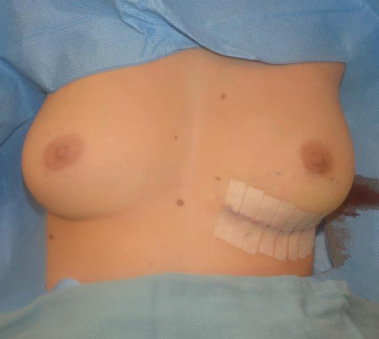
Immediate (on table) post-operative photo.
After the operation, the contradicting investigations were clarified and the diagnosis was confirmed by histopathological examination to be a fibroadenoma. The mass weighed 170 g and the dimensions were 12.7 × 9.2 × 4.8 cm; it comprised of lobulated, gray-pink nodules that ranged in size from 0.2–3.5 cm in size. The postoperative course was uneventful with no complications. Figure 9 shows her appearance two months after the operation.
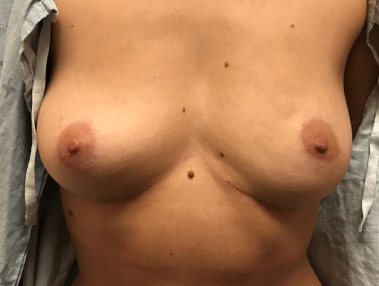
Two months after the operation.
Benign breast disease can be classified as non-proliferative lesions, proliferative lesions without atypia, or lesions with atypical hyperplasia. All other lesions are considered miscellaneous. Fibroadenomas (FA) are proliferative breast lesions without atypia and are the most common masses found in younger women between the ages of 15–35. These breast masses are typically palpable, well-defined, mobile, rubbery or firm in consistency, and are less than 3 cm in diameter. Fibroadenomas tend to proliferate and increase in size when exposed to female reproductive hormones because the fibrous and stromal connective tissue cells that they comprise of contain receptors for estrogen and progesterone [2].
Hamartomas are benign, developmental neoplasms comprised of tissues that are endogenous to the area where they originate [13]. Breast hamartomas (BH) are particularly rare, accounting for 0.1–0.7% of all benign breast lesions, and therefore fall into the miscellaneous category. On average, BHs present more often in premenopausal women (range = 13–88 years, mean = 45 years), and can vary in size from 1 to 20 cm (mean = 6 cm) [12]. They are well-circumscribed, slow-growing, painless tumors consisting of fibrotic stroma, adipose, and glandular tissue [5,6,13]. They are often lobulated, mobile, and surrounded by a pseudocapsule. Much like FAs, BHs may also increase in size due to presence of estrogen and progesterone receptors that are expressed on their epithelial and stromal components [17]. The exact etiology and pathogenesis of either FAs and BHs are not fully understood [2]. The current theory regarding the latter, however, is that these masses likely result from a developmental abnormality rather than a true neoplastic mechanism [13,17,18].
On clinical exam, asymmetry between the breasts may be detectable depending on the size of either breast masses [5,18]. The clinical sign that patients notice first is a painless, moderately firm breast mass that is most commonly located in the lateral quadrants of the breast [9,13]. The overlying skin does not tether, nor does the discrete mass fixate on the underlying fascia. In some cases, the masses may increase in size during the patient’s menstrual cycle. Palpable lymph nodes are not usually associated with either FAs or BHs.
Diagnostic tests that are typically used to characterize these lesions include ultrasound, mammogram, and MRI [18]. On ultrasound, FAs are oval in shape, homogeneous in texture, and can be iso- or hypoechoic relative to the surrounding fat [4,14]. Similarly, BHs are oval, lobulated masses that are hypoechoic consisting of sonolucent fat with a thin echogenic rim. Both lesions can displace the adjacent normal breast tissue. On mammogram, FAs have smooth margins and appear as well-defined iso- or hypodense oval masses and may sometimes contain calcifications. BHs have similar characteristics, presenting as ovoid encapsulated round masses that have mixed heterogenous density and mottled centres [5]. On T1 gadolinium-enhanced MRI, FAs vary in presentation, but typically appear as iso- or hypointense with non-enhancing internal septations. Meanwhile, BHs are encased by a dark, smooth, thin capsule and present with heterogenous gadolinium-enhanced centre [6,17]. Radiographic similarities between these breast masses were described by Georgian-Smith et al. [4], who reported indistinguishable differences in imaging features between FAs and BHs.
The utility of fine needle aspiration (FNA) as well as core biopsies have also been explored, but both tests were found to have limitations. Georgian-Smith et al. [4] reported a high discordant rate between core biopsy and excisional biopsy samples among 18 BH specimens that underwent both diagnostic tests. There were 12 false-negative diagnoses, 4 false-positive (all 4 were fibroadenomas), and 2 true-positive diagnoses. Tse et al. [6] also found that BHs were not correctly diagnosed using FNA because the aspirates lacked cytological features that are specific for BHs. This is likely due to the fact that hamartomas consist of tissues endogenous to the site and hence lack abnormal cytologic characteristics.
Surgical excision is the curative treatment for both FAs and BHs and is indicated when the masses are growing in size and/or causing symptoms like pain [9]. At extirpation, hamartomas typically appear as smooth, greyish-white to yellow masses on gross examination [5]. Histologically, FAs are made up of stromal and epithelial components that are interspersed around glands and ducts [1,2]. BHs have lobules of adipose tissue that can be found interspersed between fibrotic tissue and can be surrounded by a pseudoangiomatous stroma [6]. The challenge with diagnosing BHs is that they can often be mistaken for FAs or other benign lesions like sclerosing lobular hyperplasia. Currently, pathologists discern BHs from other breast lesions based on several factors: the lobular distribution of adipose tissue; the presence of lobules within fibrotic stroma [6]; the presence a pseudoangiomatous capsule; and whether the lobules are adherent to the surrounding tissue [12]. However, the cited proportions of fat within hamartomas varies widely in the reported literature (e.g., 5–90% in a study by Wang et al. [18], vs 10–20% by Tovar et al. [9]). This variation may explain the challenge associated with diagnosing these lesions; when there is a large volume of adipose tissue, a hamartoma may be mistaken for a lipoma, but when there is a small volume, it may be mistaken for a FA [12]. Hamartomas may also be challenging to diagnose due to its indolent nature as they are typically not biopsied or excised. For these reasons, the reported incidence of BHs may actually be an underestimation.
Of note, there are FA variants (giant FA, juvenile FA, and cystic fibroadenoma) and BH subtypes (myoid, adenolipomas, and chondrolipomas). The latter may be associated with certain syndromes. If patients are found to have a rare phyllodes tumour, the breast lesion should be excised because it can be locally aggressive, and it has higher potential for malignancy [15]. Compared to FA variants, BH subtypes are extremely rare and may require additional investigations like immunohistochemistry to arrive at the diagnosis [16]. In patients with multiple breast hamartomas, Cowden syndrome may be an important consideration because, much like a phyllodes tumour, these lesions tend to have a greater risk for malignant transformation. Eighty-five percent of females with Cowden syndrome, for example, will develop breast carcinoma at some point in their life. As such, if this diagnosis is present, the treatment option for these patients may be more extensive than local excision and would require a multidisciplinary approach to management [3].
Once the breast mass has been removed, the surrounding normal breast tissue can re-expand and resume its normal growth rate [12]. If the lesions are fully excised, recurrence of FAs or BHs is low [13,15]. A case series by Amir and Sheikh [13] found a recurrence rate of 8% among breast hamartomas, but it was hypothesized that these patients likely had multifocal disease rather than a true recurrence. The risk of malignant transformation among FAs is usually low ( <1%) but may be higher in those with a more complex variants [15]. For BHs, malignant transformation into invasive ductal or lobular carcinomas has also been found to be low and approximates that of the general population, which makes sense as hamartomas contain normal endogenous breast tissue and are not themselves premalignant masses [8]. Patients with known BHs who developed a carcinoma were typically in their 50s and the tumour size ranged from 1.5 to 10 cm [7,10]. Mammographic findings of malignant changes typically include microcalcifications or spiculated masses within the hamartoma [11]. Malignancies arising from or within hamartomas are treated in the same manner as those originating from normal breast tissue [7].
FAs and BHs are composed of similar tissue components and share many features clinically and radiographically. This case presentation is unique because of the discordance between the findings reported among multiple imaging modalities and those that were reported on pathology. Future investigators should seek to characterize the prevalence of this discrepancy. Establishing concordant and consistent radiographic and pathologic features for these solitary breast masses could help decrease unnecessary diagnostic tests and reduce patient anxiety.
Conclusion
FAs and BHs currently are diagnosed based on radiologic and pathologic features. There may be discordance between these findings, however, and clinicians should be mindful that the diagnosis may only be accurate when the whole mass is surgically removed and visualized. Surgical excision is recommended especially among patients with symptomatic breast lesions that are growing in size.
Footnotes
Acknowledgements
The authors would like to thank our patient who consented for the dissemination of these images.
Conflict of interest
The authors have no conflicts of interest or financial disclosures to declare.
