Abstract
Background
Dementias, particularly Alzheimer's disease (AD), represent a global public health concern. Although its exact cause remains unknown, neuroanatomical and cognitive changes are crucial.
Objective
Finding biomarkers helping the early diagnosis of AD is crucial for developing early interventions.
Methods
In a sample of 402 cognitively healthy adults some of them with risk factors (family history of AD and APOE ε4 carriers), we examined the relationship between APOE ε4 genotype and other aspects of family history of AD with cognitive variables and brain volumetrics using magnetic resonance imaging (MRI).
Results
Key findings suggest that family history of AD may be associated with specific neuroanatomical markers, such as left hippocampal size and white matter integrity. However, no links were observed with cognitive performance, and furthermore additional analyses on the APOE genotype showed no significant differences in any neuroanatomical aspect.
Conclusions
This study reveals the importance of monitoring with MRI subjects with family history of AD, to assess early volumetric changes and use this information to stratify participants in future clinical trials.
Keywords
Introduction
Dementia affects 50 million people worldwide, with Alzheimer's disease (AD) accounting for 60–70% of cases. 1 Furthermore, dementia has been recognized as a public health priority as the prevalence rate of dementia is expected to increase gradually, doubling every 20 years. 2 In Europe in particular, the prevalence of dementia across all age groups is between 4.6% and 6.4%.3–5
The etiology of sporadic AD is currently unknown.6,7 Although patients with sporadic AD share common anatomopathological features, the mechanism triggering these neuropathogenic processes remains unknown. 2 Neuropathological changes are mainly localized in the cerebral cortex, especially in the temporoparietal lobes bilaterally, where the presence of brain deposits of amyloid-β (Aβ), neurofibrillary tangles, neuronal degeneration, and brain atrophy can be observed.8,9 Neurofibrillary tangles are composed of tau protein, which exhibits a distinct accumulation pattern between early and late AD, characterized by Braak stages.10,11 Similar to the tau protein, AD also results in a hierarchical accumulation of amyloid deposits in the brain. 12 These neuropathological changes are initially accompanied by the onset of memory impairment, which leads to impairment of other cognitive domains as the disease progresses. 3 There is evidence that neuropathological changes in the brain in AD can be observed more than ten years before the onset of cognitive symptoms. 2 Based on these findings, the National Institute on Aging and the Alzheimer's Association (NIA-AA) updated their diagnostic criteria in 2018, emphasizing three groups of biomarkers: A (amyloid); T (pathological tau); N (neurodegeneration) and recognizes three stages in the continuum of AD: (1) Preclinical AD, consisting of a cognitively asymptomatic phase but with the presence of biomarkers that pose a risk for the development of AD; (2) Mild cognitive impairment due to AD, a phase in which the first clinical symptoms appear in memory and other domains but do not interfere with activities of daily living; and (3) Dementia due to AD, a phase in which changes occur in at least two cognitive domains and affect behavior and activities of daily living. 13
Early diagnosis and associated early intervention are important to prevent or delay the transition from a preclinical state to a prodromal stage of the disease. 14 For this reason, the search for markers or biomarkers that inform us about the disease in a preclinical phase is crucial for early intervention. Therefore, in addition to age, one strategy for detecting early biomarkers of the disease is to evaluate populations with risk factors such as family history and/or being carrier of the APOE ε4 since both features have been identified as risk factors for the development of the disease. 15
The Apolipoprotein E (APOE) gene is the most important genetic factor associated with sporadic AD and has three polymorphic variants or alleles. The ε3 allele is the most common in the population with a prevalence of 78%, while the ε2 allele, which occurs less frequently at 7%, appears to have a protective effect against the development of AD. On the other hand, the ε4 allele, present in 14% of the Caucasian population, represents a risk factor for late-onset AD, especially after age 65. 16 It has been observed that patients with AD have a higher frequency of the ε4 allele, ranging from 36% to 52%, compared to 11% to 16% in the general population. 2 Furthermore, the risk of developing AD increases from 20% to 90%, and the age of onset decreases from 84 to 68 years in the presence of homozygosity of the APOE ε4 allele, which practically guarantees the onset of the disease at 80 years. 17 Carriers of the APOE ε4 allele show structural changes in their brains compared to healthy older adults, particularly in terms of a reduction in volume in the medial temporal lobe 18 as well as amyloid penetrance. 19 In addition, ε4 carriers exhibit a more pronounced cognitive decline in tests that measure attention, memory, language, and executive functions compared to non-carriers, as recent studies have shown.20–22
In addition to APOE ε4, research has focused on family history of AD as an independent risk factor for the development of the disease, even without the presence of the APOE ε4 gene.23,24 Pedigree-based studies have identified a relative risk for developing AD based on family history beyond first-degree relatives. 25 This line of investigation has found that cognitively healthy individuals with a first-degree family history of AD exhibit volume reductions in brain regions susceptible to AD, particularly the precuneus and parahippocampal gyrus. 26 Other studies have linked family history of AD to poorer subjective memory scores and cortical thinning of the left hippocampus. 27
Abnormalities in the function of the medial parietal regions have been associated with biological markers of AD in the cerebrospinal fluid in ε4 carriers and reduced memory in a group of asymptomatic individuals but were associated with a family history of AD. 28 Other studies also suggest that the APOE ε4 allele runs in families and is passed down independently, further contributing to the thinning of the hippocampal cortex. 29 Interestingly, the presence of a family history of AD in cognitively healthy adults can influence a progressive decline in gray matter volume in the precuneus and parahippocampal gyrus.26,30 Other longitudinal studies have found that greater global brain atrophy is associated with lower scores on verbal and visuospatial memory tests in healthy adults with a positive family history of AD than in those with a negative family history of AD, particularly in ε4 carriers. 31
Recent research has shown that early brain changes may occur in middle-aged adults with normal cognitive function and a family history of dementia. 32 These changes are characterized by abnormalities of white matter and cerebral perfusion, which are particularly pronounced in ε4 carriers.
All previous studies where mainly analyzing one data-point without describing the curse of cognitive and neuroanatomical variables in a longitudinal design. The purpose of this study was to examine the association between family history of AD, APOE genotype, cognitive performance, and brain volume, particularly the medial temporal lobe, determined by magnetic resonance imaging (MRI) in a sample of individuals over 70 years of age. Since participants were followed for five years and remained cognitively healthy throughout the study, our work focused on analyzing the natural course of cognitive performance and brain volume as a function of family history of AD and APOE genotype. We hypothesized a progressive involvement of the neuroanatomical features across time, with a potential penetrance of being carrier of APOE genotype.
Methods
Participants
The study was conducted in the city of Madrid, Spain, as part of an ambitious scientific program called “Study of the anatomo-functional connectome of AD-relatives: An early intervention on cognitive al lifestyles (CONNECT-AD)” to early detect and prevent subtle cognitive changes associated with AD. A total of 402 cognitively healthy individuals were recruited through community awareness campaigns and collaborations with local health centers and followed for five years. The participants were home-dwelling older adults volunteers, 70–88 years of age, and without relevant psychiatric, neurological, or systemic disorders. The participants signed informed consent and undertook a systematic clinical assessment, including medical history, neuropsychological examination, and brain MRI scan. APOE genotype was also studied, with total DNA isolated from peripheral blood following standard procedures.
Ethical approval was granted by the Research Ethics Committee of Hospital Clínico San Carlos, Madrid, Spain, and written informed consent was obtained from all the participants. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation, and with the Helsinki Declaration of 1975 and its later amendments.
Variables
Two independent variables are of interest to this study. On the one hand, the family history of AD with a focus on first-degree relatives (i.e., parents and/or siblings), which was collected by a practitioner during a medical interview and coded dichotomously for each participant (yes/no). On the other hand, the APOE haplotype ε4, also coded dichotomously (carrier versus non-carrier). APOE gene polymorphism status was studied with total DNA isolated from peripheral blood following standard procedures. Genotyping of APOE polymorphisms (rs429358 and rs7412) was performed by Real-Time PCR. 33
In addition, the dependent variables included the following cognitive tests: Mini-Mental State Examination (MMSE) 34 ; Free and Cued Selective Reminding Test (FCSRT) 35 ; verbal fluency, symbol-digit from the Wechsler Adult Intelligence Scale (WAIS-IV); 36 Functional Assessment Questionnaire (FAQ); 37 Geriatric Depression Scale 15-items version (GDS-15); 38 and Clinical Dementia Rating (CDR). 39 Moreover, the following neuroanatomical parameters were considered in this study: white matter volume, gray matter volume, whole brain volume, left hippocampus volume and right hippocampus volume. These brain areas were chosen because of their known association with cognitive decline and neurodegeneration characteristic of AD.
MRI data acquisition and preprocessing
The imaging data were acquired on a 3T General Electric scanner (GE Milwaukee, Milwaukee, WI, USA) utilizing the following parameters: T1-weighted inversion recovery, a supine position, a flip angle 12°, 3-D pulse sequence (echo time Min. full), a time inversion 600 ms, a receiver bandwidth 19.23 kHz, a field of view = 24.0 cm, a slice thickness 1 mm, and freq phase 288 × 288. The preprocessing of MRI 3 Tesla images in this study consisted of generating an isotropic brain image with non-brain tissue removed. We used the initial, preprocessing step in the computational segmentation tool FSL pipeline (fsl-anat). 40
FSL allows the processing of structural MRI with the following tools: the Brain Extraction Tool (BET), the FMRIB's Automated Segmentation Tool (FAST), and the FMRIB's Integrated Registration and Segmentation Tool (FIRST). The BET routine separates brain tissue from non-brain tissue.41,42 BET estimates the total brain volume after bone, fat, and muscle has been removed in the image. The FAST routine 43 routine can classify the voxels of the brain into white matter, gray matter, and CSF. And FIRST 44 uses Bayesian modeling to segment the different subcortical structures of the brain. The model training set includes both normal and pathological brains (including cases of schizophrenia and AD) and the age range is between 4 and 87. FIRST segments the following brain structures: putamen, thalamus, amygdala, caudate, pallidum, hippocampus, accumbens, and brainstem.
The stages in the FSL pipeline are, in order: alignment of images to standard orientation (MNI), automatic image cropping, bias field correction (RF/B1 inhomogeneity correction), registration in standard space (linear and non-linear), brain extraction, tissue type segmentation and subcortical structure segmentation. We run the pipeline in a Mac OS X operating system, product version 10.14.5 and build version 18F132. The version of the BET tool for FSL was v2.1 - FMRIB Analysis Group, Oxford, and the FIRST tool version is 6.0.
Statistical analysis
We conducted a preliminary analysis of demographic, cognitive, and neuroimaging variables at baseline to find out their distribution. Association between categorical variables were also analyzed with the Fisher's exact test. In addition, since most variables were not adjusted to the parametric assumptions, mean differences between individuals with negative vs. positive familial AD history (Table 1), as well as ε4 non-carriers vs. carriers (Table 2), were based on non-parametric Welch's tests. Demographic, cognitive, and neuroimaging data at baseline and last follow-up were used as dependent variables.
Baseline demographics, cognitive and neuroimaging data by family history of AD.
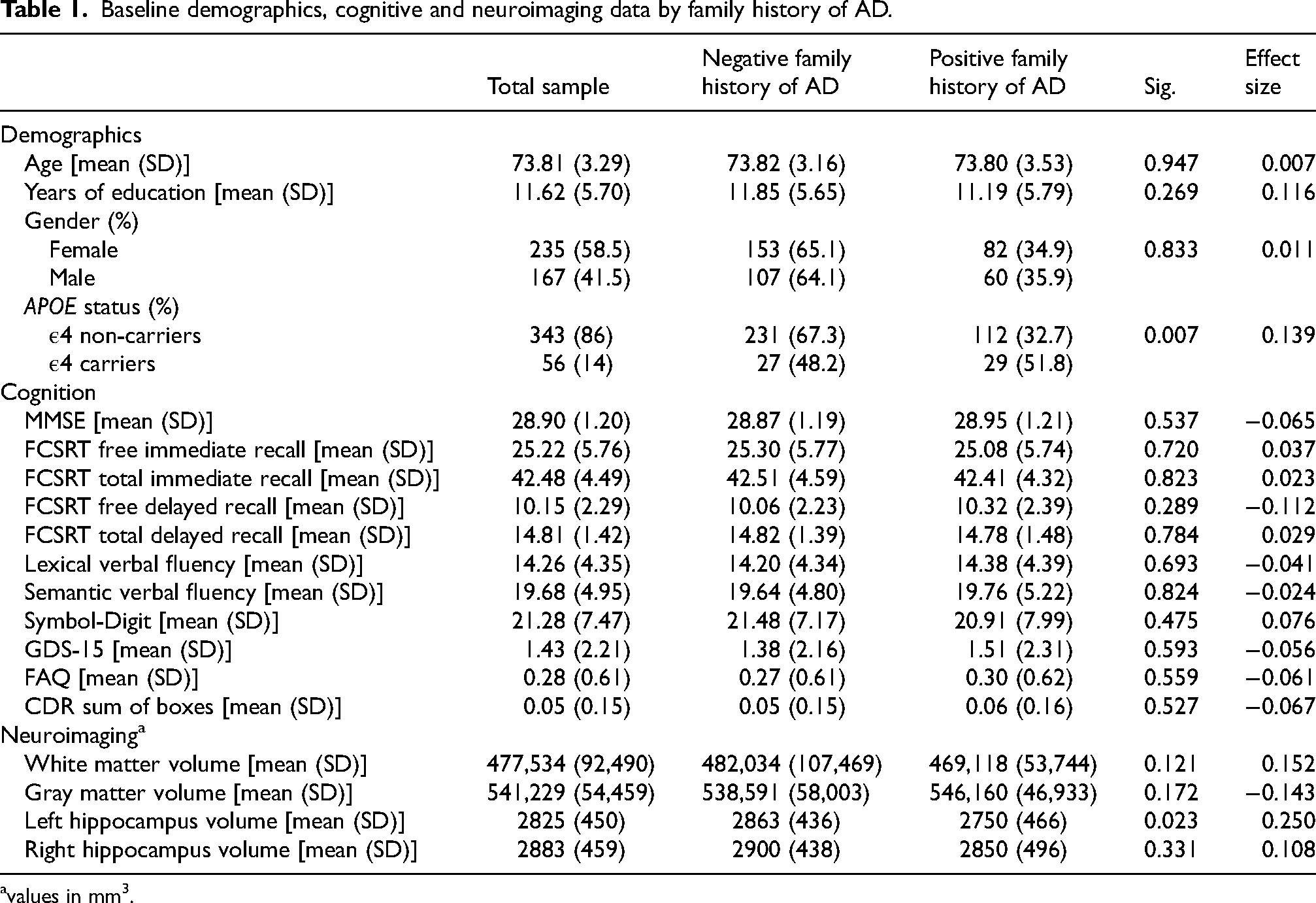
avalues in mm3.
Baseline demographics, cognitive and neuroimaging data by APOE status.
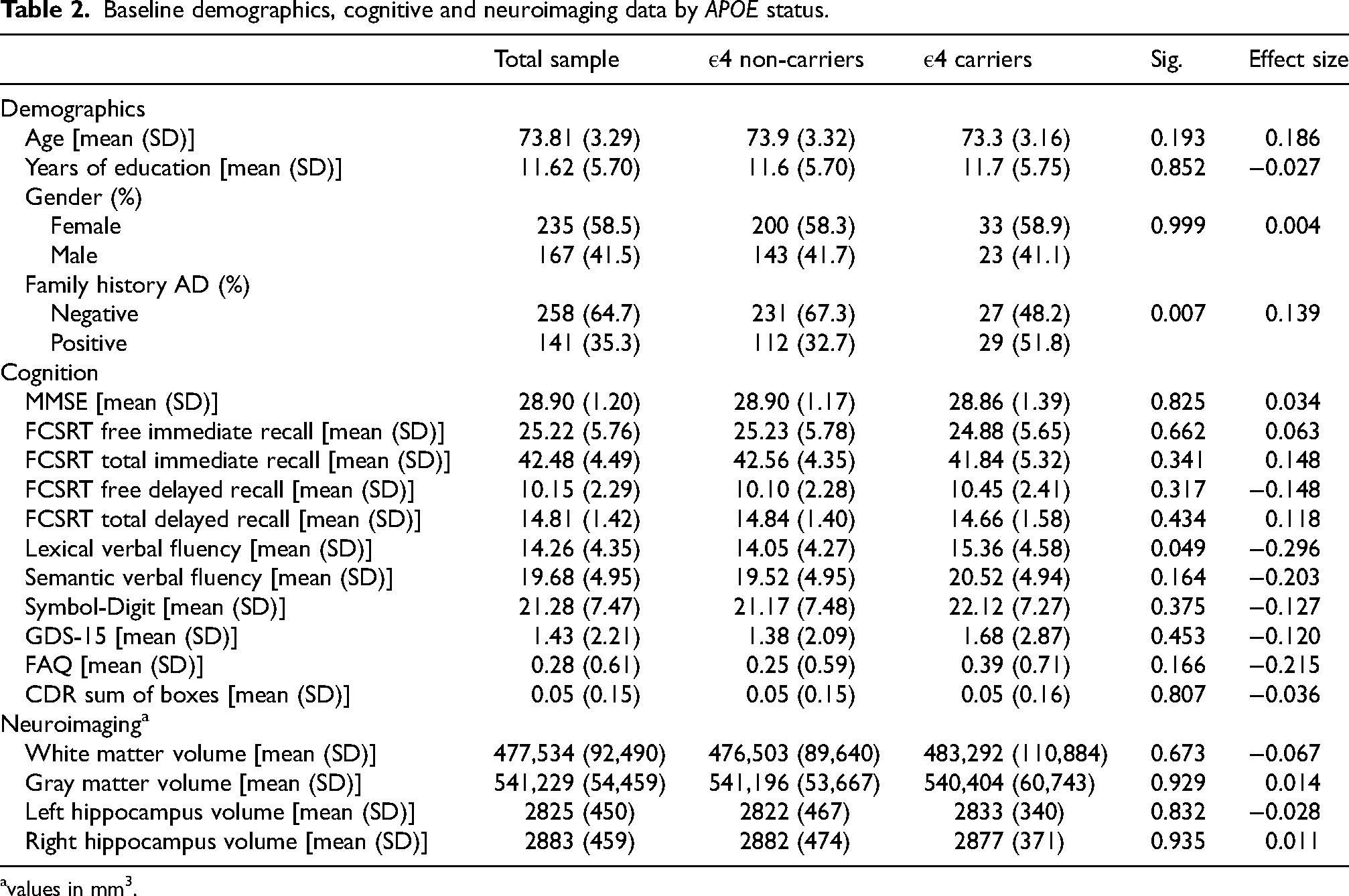
values in mm3.
Finally, to determine the effect of familial AD history on cognitive and neuroanatomical changes over time, we run generalized additive mixed models (GAMMs). These models are particularly suited for longitudinal data analysis, allowing us to account for both fixed effects, such as familial AD history as a linear term, and random effects, such as individual variability. As covariates to isolate their effects on the outcomes, age and years of education were both included as smooth terms using a penalized cubic regression spline, whereas gender was added as a linear term. To measure the models fit, special consideration was given to tests of signification for the estimators and the square deviance. Analysis of residuals was also performed to test the adjustment of the models to the data.
We used 2-sided significance tests for all analyses, with statistical significance set at p-value <0.05. All statistical analyses were performed using R version 4.2.1. 45 GAMMs were implemented with the mgcv package (version 1.8-42).
Results
Tables 1 and 2 provide a detailed overview of the demographic, cognitive, and neuroanatomical characteristics at the study's baseline for all participants. Additionally, they offer a comparison between positive and negative levels of family history in Table 1, and between carriers and non-carriers of the APOE variable in Table 2, displaying descriptive statistics for each group.
The average age of participants was 73.81 years, with an average of 11.62 years of education. There were 235 female participants and 167 male participants. Regarding the APOE genotype, 56 carriers of the ε4 allele and 343 non-carriers were identified. Additionally, 258 individuals had a family history of AD, while 141 did not have a family history of the disease.
The results presented in Table 1 and Table 2 indicate that no statistically significant differences were found in demographic or cognitive variables for either of the two independent groups (family history of AD and APOE). Additionally, no statistically significant differences were detected in the neuroanatomical characteristics associated with APOE. However, significant differences were observed in the volume of the left hippocampus between individuals with and without family history, with the latter group being favored.
When analyzing cognitive and neuroanatomical data at the last follow-up visit, a similar trend to baseline data is observed: APOE does not show significant differences in any cognitive or neuroanatomical parameter, while family history maintains a significant difference (Welch's t = 2.339; p = 0.020; Cohen's d = 0.279) in the size of the left hippocampus in favor of the group without a family history of AD (2729 ± 538 versus 2585 ± 495). Additionally, significant differences are observed at this visit in the total volume of white matter (Welch's t = 2.512; p = 0.013; Cohen's d = 0.271) also in favor of the group without a family history of AD (474,565 ± 120,079 versus 449,457 ± 51,882). In any case, it is noted that family history and APOE are strongly associated with each other (Fisher's exact test = 7.71; p = 0.007; Cramer's V = 0.139).
In the generalized additive mixed models (Table 3), the results obtained from the univariate analysis with baseline data are confirmed. Additionally, longitudinally, it is observed that family history of AD is significantly associated with white matter volume (B = −20,869; 95% CI, −36,664.16 to −5074.089; p-value = 0.009; standardized β = 0.80; 95% CI, 0.67 to 0.95) and left hippocampal volume (B = −119.92; 95% CI, −221.49 to −18.34; p-value = 0.021; standardized β = 0.78; 95% CI, 0.64 to 0.96). Figure 1 shows how neuroimaging parameters change over time depending on family history of AD.
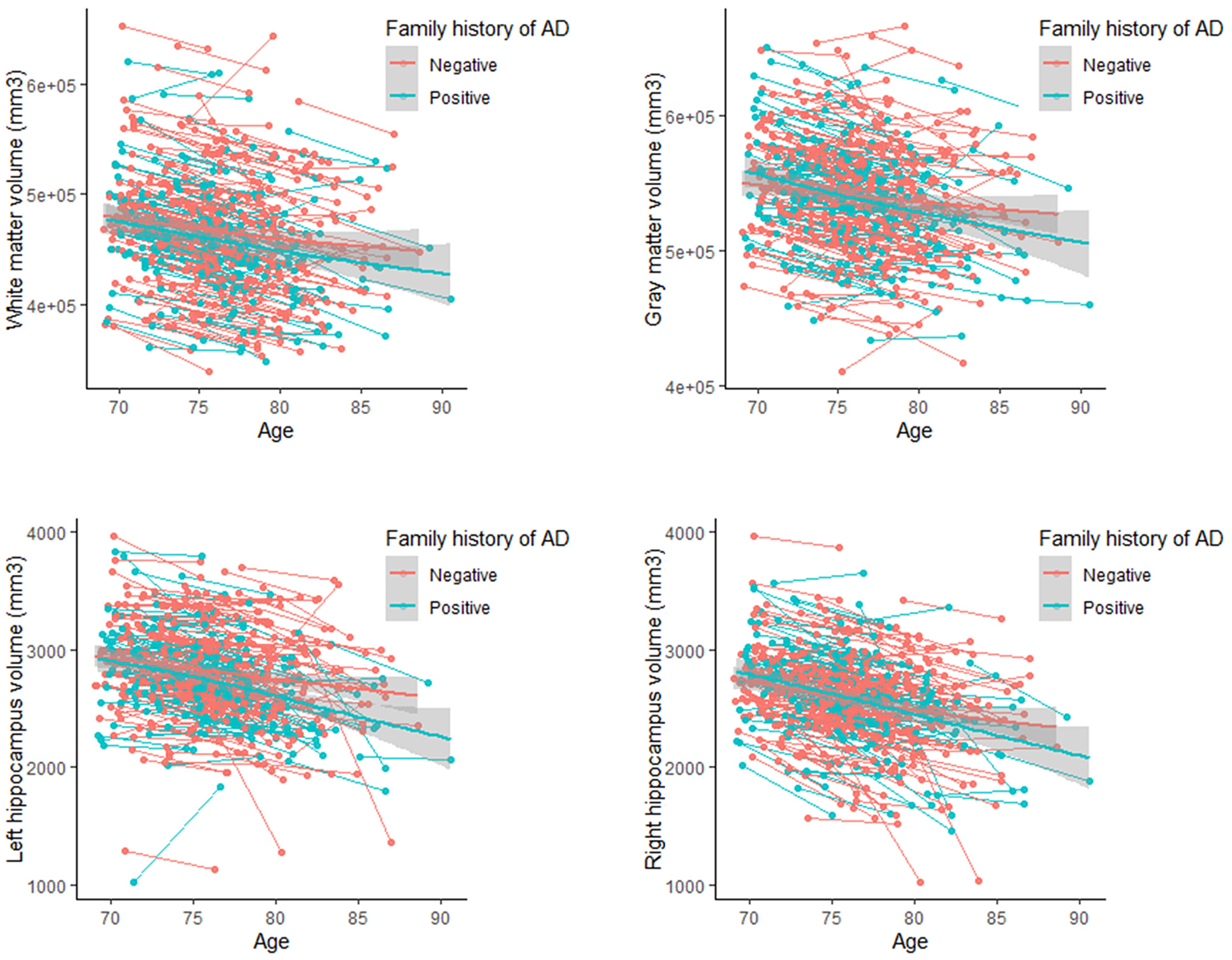
Evolution of brain volume according to the presence of a family history of AD.
Generalized additive mixed models of cognitive and neuroimaging variables with additional adjustment for potential mediators.
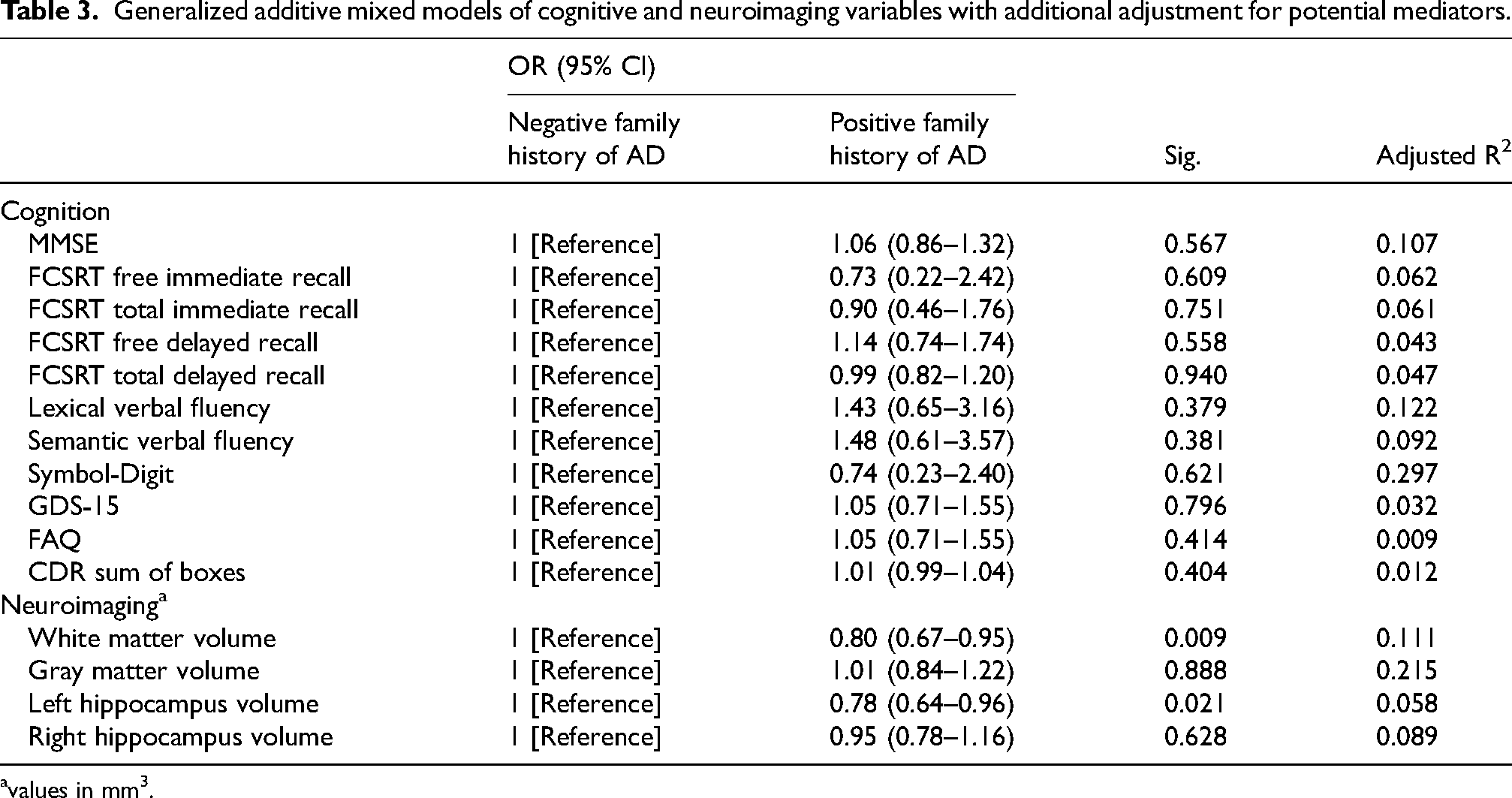
values in mm3.
Discussion
The aim of the present study was to investigate, in a longitudinal design, the possible association between family history of AD, APOE genotype, cognitive performance and brain volume. For this purpose, a sample of cognitively healthy people over 70 years of age was followed for 5 years and both baseline values and changes over time were analyzed. Our main results demonstrate that a family history of AD is associated with neuroanatomical variations, particularly in left hippocampal volume in both cross-sectional and longitudinal analyses, along with white matter measurements in the longitudinal analysis. Interestingly, APOE genotype shows no association with neuroanatomical parameters. Regarding cognitive performance, no association was found with family history of AD or APOE.
Considering the above results, we can conclude that, contrary to the conclusions of Haussmann et al., 27 no statistically significant differences were observed in cognitive variables associated with family history of AD. Furthermore, our results also differ from those of Veréb et al., 28 who claimed that individuals with a family history of AD had poorer memory test scores. These discrepancies could be attributed to the fact that the sample in our study consisted exclusively of cognitively healthy individuals. In this context, the lack of significant differences in cognitive tests between groups with and without a family history of AD could reflect a preclinical stage of the disease in which clinical manifestations are not yet apparent.
Likewise, our analyzes revealed no significant differences in cognitive variables associated with the presence of the ε4 allele in the APOE genotype in contrast to previous results that found that carriers of the ε4 allele of the APOE genotype have lower cognitive performance.20–22 This could be because, APOE ε4 is typically associated with increased amyloid deposition. However, amyloid is not directly associated with cognitive impairment. 46 The number of scholar years is relatively high in this sample, in comparison with other studies, and this factor could be contributing to delay cognitive decline avoiding the penetrance of the two risk factors considered in this study: being relative of AD and carrier of the ε4 allele of the APOE.
Our study found no differences in the neuroanatomical features associated with the presence of the ε4 allele in the APOE genotype, in contrast to the available evidence which suggests an association between carrying the ε4 allele and notable structural alterations, particularly in the hippocampus.18,27,47–49 However, significant differences in left hippocampal volume were observed depending on family history of AD, favoring the group with no history of AD. These results are consistent with previous evidence that associates the presence of a family history of AD with volume reductions in regions vulnerable to the disease, particularly in the hippocampus.26,29 Thus, according to this association, it could be hypothesized that being a relative of an AD patient is a risk factor for a more rapid reduction in medial temporal lobe volume over time compared to those without a family history of AD. This subclinical factor, at the time-point of our study, could be associated with an earlier onset of clinical symptoms compared to participants without a family history of AD. This hypothesis would be assessed in future cognitive evaluations of this sample.
Regarding the longitudinal analysis, we identified no significant cognitive or neuroanatomical differences in relation to APOE genotype. This contrasts with previous findings that link the presence of the APOE ε4 genotype with cognitive decline.20–22 Additionally, it differs from what was found by Debette et al., 31 who observed greater global cerebral atrophy in APOE ε4 carriers. Nevertheless, we did find longitudinal significant differences both in the left hippocampal volume and white matter measurements associated with family history of AD, favoring individuals with no family history. These results align with previous evidence which indicates that individuals with normal cognitive function and a family history of AD exhibit anomalies in white matter.26,30 According to the recent study by Zhang et al., 50 it is likely that white matter hyperintensity is associated with intrinsic degenerative processes of AD.
Based on the above, our findings suggest the feasibility of considering family history as a variable or risk factor in the assessment of Alzheimer's disease. This variable presents significant advantages compared to the APOE genotype, as its acquisition is straightforward, immediate, and cost-effective, making it particularly useful for population studies. As we have demonstrated, family history correlates with expected changes in the pathogenesis of AD. Additionally, the inclusion of family history would enable the recruitment of older individuals with an increased risk of progressing to dementia, which would be beneficial for conducting clinical trials and implementing community interventions aimed at dementia prevention. Furthermore, this measure can be employed for monitoring older relatives of AD patients, who are at a higher risk of developing the disease.
The study has certain limitations, among which the high educational level of the sample stands out. Specifically, the sample has an average of 11.62 years of formal education, which is considerably higher than the average in the general Spanish population. In this population, 47.4% of individuals over 55 years old have only secondary education or less. 51 This disparity in educational levels could influence the study's results, as higher education levels have been previously associated with greater cognitive reserve, potentially biasing the detection and characterization of cognitive impairment. Therefore, it is necessary to consider this variable when interpreting the findings and generalizing the conclusions to a broader population.
In summary, the presence of a family history of AD appears to correlate with a reduction in the volume of the left hippocampus and white matter, both basally and longitudinally, without a significant association with cognitive performance. Additionally, analyses of the APOE genotype did not reveal significant differences in neuroanatomical parameters or cognitive performance. This lack of correlations could be due to the nature of our sample, consisting of individuals who have undergone exhaustive annual evaluations, ensuring the absence of cognitive impairment. However, only family history showed an association with the reduced size of the left hippocampus and white matter, unlike the APOE genotype. These findings underscore the importance of considering family history in the study of neuroanatomical biomarkers of AD and in the selection of participants for clinical studies, which could enhance the understanding and prevention of the disease. Furthermore, the fact that the longitudinal studies showed a progressive decrease of hippocampal and white matter volume, highlight the need of a neuroanatomical monitoring in subjects with a first grade relative of AD patients. More data points are needed to assess time of cognitive decline in association with neuroanatomical variables.
Footnotes
Acknowledgments
The authors have no acknowledgments to report.
Author contributions
Patricia De María-Ureta (Investigation; Methodology; Writing – original draft; Writing – review & editing); Marina Ávila-Villanueva (Investigation; Methodology; Writing – review & editing); Miguel Ángel Fernández-Blázquez (Data curation; Formal analysis; Methodology; Supervision; Writing – review & editing); Fernando Maestú (Conceptualization; Supervision; Writing – review & editing).
Funding
Financial support came from the project CONNECT-AD (RTI2018-098762-A-C31 and RTI2018-098762-A-C32) funded by the Spanish Ministry of Science, Innovation, and Universities—State Research Agency—through the European Regional Development Fund.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
All data generated during this study are included in this article or are available from the corresponding author on reasonable request.
