Abstract
Background
Alzheimer's disease (AD) is the most common form of dementia, marked by progressive cognitive decline. Low-density lipoprotein cholesterol (LDL-C) has been implicated in AD pathology, but findings remain inconsistent. Apolipoprotein E4 (APOE4) status and sex may contribute to this variability.
Objective
To examine how LDL-C association with neurodegeneration in AD patients, differ according to APOE4 status and gender.
Methods
We stratified 106 AD patients by APOE4 status and sex into four subgroups: male APOE4+, female APOE4+, male APOE4−, and female APOE4−. Longitudinal cortical thickness changes were assessed using magnetic resonance imaging (MRI). We examined the association between LDL-C levels and cortical thinning within each subgroup.
Results
In APOE4-positive females, higher LDL-C levels were significantly associated with accelerated cortical thinning in several regions, including the parahippocampal (β = −0.0075, p = 0.017), medial orbitofrontal (β = −0.0025, p = 0.028), fusiform (β = −0.0047, p = 0.034), posterior cingulate (β = −0.0097, p = 0.006), and inferior temporal cortices (β = −0.0085, p = 0.019). This subgroup also showed a significant association between LDL-C and MMSE decline (β = −1.409, p = 0.014) as well as longitudinal increases in cerebrospinal fluid phosphorylated tau181 (β = 0.014, p = 0.039). These effects were not observed in other subgroups.
Conclusions
Elevated LDL-C is associated with increased neurodegeneration and cognitive decline in female AD patients carrying the APOE4 allele. These exploratory findings highlight a subgroup-specific vulnerability to lipid-related neurodegeneration in AD and underscore the importance of considering both sex and genetic background in future studies.
Keywords
Introduction
About two-thirds of dementia cases are caused by Alzheimer's disease (AD), a progressive neurological disease. The onset and course of AD are influenced by a combination of environmental and genetic variables, making the disease complex. This causes variations in the distribution of neurofibrillary tangles and atrophy patterns that are detected by magnetic resonance imaging (MRI). Researching the illness is made more difficult by this heterogeneity.1–3
Both elevated levels of the low-density lipoprotein cholesterol (LDL-C) and an increased risk of AD have been linked to the Apolipoprotein E4 (APOE4) allele.4,5 An increased risk of AD has also been independently associated with higher LDL-C levels. APOE4 positivity does not fully explain the connection between LDL-C levels and AD, which seems to be stronger when utilizing earlier measures of LDL-C.6,7 Some studies showed higher LDL-C levels are more strongly linked to an AD diagnosis in APOE4 carriers than in non-carriers, suggesting that APOE4 modifies the effect of LDL-C on AD risk.8,9
The results of earlier research, however, seem to be contradictory. While some demonstrated that LDL-C has a negligible impact on the risk of AD, others demonstrated that greater levels of LDL-C in midlife are linked to larger brain volumes in later life in areas linked to AD. Even after controlling for interactions with APOE4 status, these differences still exist; some studies indicate that higher LDL-C is linked to an increased risk of AD in APOE4 carriers, while other studies report that LDL-C is linked to an increased risk in non-carriers, and still others find no interactions between the two.10–16
The confounding effect of lipid-lowering medications or reverse causality, such as decreased body mass index (BMI) and LDL-C brought on by the course of the disease, could be the cause of these discrepancies.6,7 Sex-related differences may also be the source of discrepancy; multiple studies indicate that the impact of LDL-C on AD risk is greater in APOE4 positive females. Additionally, brain areas that overlap with those previously identified as susceptible to APOE4-related neurodegeneration showed more severe atrophy in female AD patients.17–20
Longitudinal studies investigating the impact of LDL-C on regional cortical thickness are scarce, particularly in areas susceptible to neurodegeneration linked to APOE4. To learn more about how APOE4, LDL-C, and sex interact to affect neurodegeneration in AD, more research is required.
In this work, we investigated the possible contributions of sex, ApoE4, and LDL-C to the occurrence and development of AD. Examining the relationship between their interaction and the chance of an AD diagnosis and evaluating subgroup variations in the regional cortical atrophy patterns linked to these factors over time were our two main goals.
Methods
Data source
The Alzheimer's Disease Neuroimaging Initiative (ADNI) database provided the data for this investigation. To better define the course of AD, ADNI, launched in 2003, provides researchers with data, such as imaging data, genetics, cognitive tests, CSF, and blood biomarkers that are gathered, verified, and used by researchers as disease predictors. Complete guidelines and documentation are accessible on the ADNI website (https://adni.loni.usc.edu/).
Data selection and description
Deidentified data from ADNI database was used to select 313 subjects, ages 65 to 91, who were either diagnosed with AD (114 subjects) or cognitively normal (CN) (199 subjects). To eliminate cases of early-onset AD, a minimum age of 65 years was established as a selection criterion. Exclusion criteria included patients with a history of substance misuse, other forms of dementia, and confounding neurological diseases. 20
The study used baseline demographic information (age, race, gender, and education) along with LDL-C levels, brain regional cortical thicknesses, and APOE4 genotype. Nuclear magnetic resonance was used by Nightingale Health to determine LDL-C levels (Supplemental Material 1). We included a history of smoking and hypertension as cardiovascular risk factors; we did not include a history of diabetes or hypolipidemia since these variables were either absent or unreliable. Cognitive function was assessed using Mini-Mental State Exam (MMSE).
MRI data
T1 weighted 1.5 Tesla MRI scans were pre-processed by ADNI using gradient wrapping, B1 field correction and N3 inhomogeneity correction at Mayo Clinic. T1 MPRAGE/IR-SPGR images were then processed by UCSF using FreeSurfer version 4.3 (cross-sectional pipeline) and version 4.4 (longitudinal pipeline), with cortical parcellation on Desikan-Killiany atlas. We used UCSF-generated cortical thicknesses files available on ADNI for our analysis.
Images that did not pass UCSF quality control were not included. Only the impacted regions’ cortical thickness values are excluded in cases of partial quality control failures. Baseline cortical thicknesses were evaluated using cross-sectional files, and changes over time were evaluated using longitudinal files that used within-subject templates. Additional information is available in the UCSF image processing file (Supplemental Material 2) that is accessible through the ADNI database.
Cerebrospinal fluid biomarkers
Cerebrospinal fluid (CSF) Aβ42, total tau (t-tau), and phosphorylated tau (p-tau181) were measured at the ADNI Biomarker Laboratory at the University of Pennsylvania using the Roche Elecsys® automated electrochemiluminescence immunoassays, following the manufacturer's protocol. Full assay procedures are provided in Supplemental Material 3.
Statistical analysis
Stata version 17 (StataCorp, College Station, TX) was used for statistical analyses. Clinical, radiological, and baseline demographic data were compiled and compared between the AD and CN diagnosis groups.
We used multivariate logistic regression to evaluate the baseline relationship between LDL-C levels and AD risk. Next, we used multiple linear regression to examine how LDL affected AD patients’ regional cortical thicknesses, MMSE scores, and levels of CSF biomarkers.
To investigate longitudinal impacts, we used linear mixed effects models to create best linear unbiased predictor slopes, that represent annualized rates of change in regional cortical thickness, MMSE scores and CSF biomarkers. To take into consideration correlations between baseline values and change rates, we employed unstructured covariance.
Our main hypothesis was that APOE4+ females are more affected by LDL-C in terms of cortical atrophy and AD risk. As a result, we divided our analysis into four subgroups based on APOE4 status and sex: male APOE4+, female APOE4+, female APOE4-, and female APOE4+. The differences in baseline characteristics between the groups were then compared.
Age, sex, race, education, BMI, use of lipid-lowering drugs, diagnosis of hypertension, history of smoking, and total intracranial volume (for cortical thickness models) were all considered in all models. The entorhinal, parahippocampal, posterior cingulate, fusiform, middle temporal, inferior temporal, precuneus, medial orbito-frontal, and inferior parietal cortices were among the regions of interest that were chosen based on previous research on APOE4-related cortical atrophy.18–25
Results
Dataset characteristics
The study involved 313 participants in total, 114 of whom had AD and 199 of whom were cognitively normal (CN). 91% (N = 104) of the AD group where of mild severity while the rest where of moderate severity. The baseline characteristics are compiled in Table 1. Significant differences were observed in several key variables. The AD group had significantly lower education years (median 14 versus 16, p < 0.001) and MMSE scores (median 23 versus 29, p < 0.001) compared to the CN group. A higher proportion of AD patients were APOE4 positive (70.18% versus 26.62%, p < 0.001). The AD group also had a significantly lower mean BMI (25.46 versus 26.82, p = 0.01) and a marginally higher LDL-C level (2.98 versus 2.81, p = 0.05). Cortical thickness measurements were significantly lower in the AD group compared to controls (p < 0.001). Total and phosphorylated tau181 were higher in CSF of AD patients while Aβ42 was lower compared to healthy controls (p < 0.001).
Baseline descriptives of AD and CN groups.

Independent sample t-test.
Wilcoxon-rank-sum test.
Pearson Chi-Square test.
Fisher's exact test.
*Number of patients with data on CSF biomarkers was 80 in the AD group and 106 in the CN group.
AD: Alzheimer's disease group; CN: Cognitively normal group; SD: standard deviation; IQR: interquartile range; LDL-C: low density lipoprotein cholesterol; BMI: body mass index; MMSE: Mini-Mental State Exam.
Baseline cross-sectional analysis
LDL-C was not linked to an increased risk of being diagnosed with AD in multiple logistic regression models that included age, sex, race, education, APOE4 status, BMI, use of lipid-lowering medications, hypertension, smoking history, and baseline LDL-C levels (odds ratio = 1.43, SE = 0.32, p = 0.11). Higher BMI was linked to a lower risk of AD (odds ratio = 0.92, SE = 0.04, p = 0.03), and the presence of APOE4 was significantly linked to an elevated risk of AD (odds ratio = 6.16, SE = 1.74, p < 0.001).
In multiple regression models within the AD group, adjusting for age, sex, race, education, APOE4 status, BMI, lipid lowering drugs, hypertension, smoking history and intracranial volume, higher LDL-C levels were associated with reduced cortical thickness only in the precuneus region (β = −0.05, SE = 0.02, p = 0.045).
APOE4 positivity was not associated with cortical thickness of any region at baseline. Notably, hypertension was associated with reduced cortical thickness in the entorhinal, fusiform, posterior cingulate, precuneus, inferior parietal, middle temporal and inferior temporal cortices (p < 0.05). No significant LDL-C effect was observed in APOE4 positive females when stratified by APOE4 status and sex. Baseline MMSE scores and CSF tau levels were not influenced by neither LDL-C nor APOE4 status. However, APOE4 positivity was associated with decreased CSF Aβ42 (β = −347.88, SE = 84.96, p < 0.001).
Longitudinal analysis
Follow-up MRIs were performed on 106 AD patients, with a median (IQR) follow-up period of 2.03 years (1.08–2.10). There were significant differences in age, LDL-C, ICV, and the rates of change in parahippocampal cortical thickness and CSF Aβ42 across subgroups based on APOE4 status and sex (Table 2).
Demographic, clinical, and imaging characteristics of AD patients (N = 106) stratified by APOE4 status and sex.
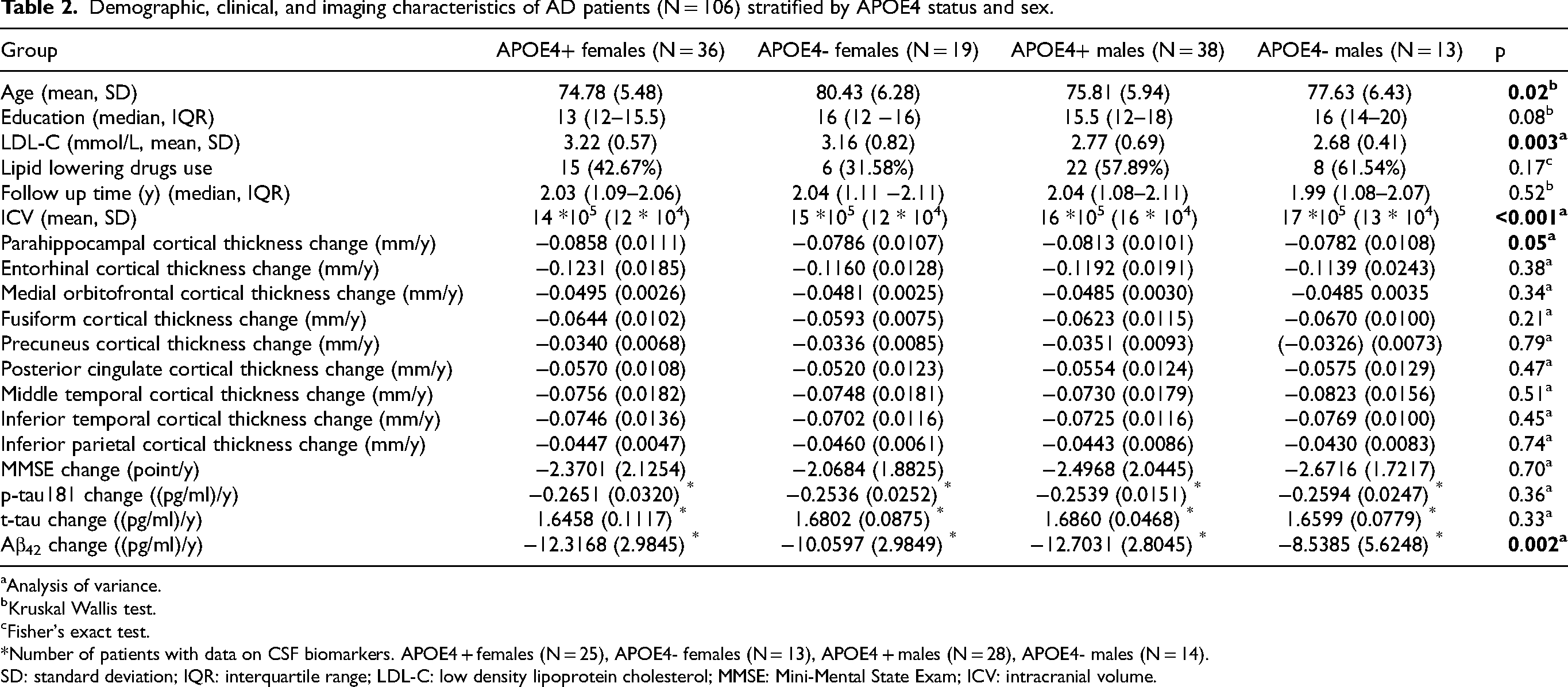
Analysis of variance.
Kruskal Wallis test.
Fisher's exact test.
*Number of patients with data on CSF biomarkers. APOE4 + females (N = 25), APOE4- females (N = 13), APOE4 + males (N = 28), APOE4- males (N = 14).
SD: standard deviation; IQR: interquartile range; LDL-C: low density lipoprotein cholesterol; MMSE: Mini-Mental State Exam; ICV: intracranial volume.
Pairwise comparisons revealed no significant difference in the rate of change in parahippocampal thickness among the subgroups. APOE4-positive females had significantly higher LDL-C compared to both APOE4-positive males (p = 0.001) and APOE4-negative males (p = 0.001). LDL-C values were overall greater in females than in males (p < 0.001). On pairwise comparisons, the difference between subgroups in Aβ42 rate of change was revealed to be due to APOE4 genotype, without a specific effect of female sex. APOE4 positive patients had more per year decline in CSF Aβ42 compared to APOE negative patients (p < 0.001).
Pairwise comparisons showed that APOE4 + positive females were significantly younger than APOE4- females (p = 0.002), and that APOE4 + positive males were significantly younger than APOE4- females (p = 0.02). Nevertheless, overall, APOE4+ AD patients were younger than APOE4-AD patients (p = 0.003). Further subgroups characteristics are summarized in Table 2.
Excluding interaction terms, we used multivariable regression to investigate the relationship between regional cortical thickness change annually, APOE4 status, and bassline LDL-C. APOE4 positivity was associated with the more rapid annual parahippocampal cortical thinning (β = −0.0052, SE = 0.0023, p = 0.022). The fusiform and inferior temporal cortices showed greater thinning in response to higher LDL-C levels (β = −0.0032, SE = 0.0016, p = 0.043 and β = −0.0047, SE = 0.0019, p = 0.015, respectively).
When stratifying by APOE4 status only, LDL-C levels among APOE4 positive patients (n = 68) were significantly associated with thinning in the fusiform and inferior temporal cortices (β = −0.0055, SE = 0.0020, p = 0.006 and β = −0.0055, SE = 0.0023, p = 0.021, respectively). No region showed significant effects of LDL-C levels among APOE4- patients (n = 25–32 depending on regions passing quality control).
Increased LDL-C was associated with annual worsening of MMSE score (β = −0.8037, SE = 0.2906, p = 0.007). This association was only significant in APOE4 positive patients when further stratifying by APOE4 status (β = −1.0360, SE = 0.3525, p = 0.004). There was no significant effect of APOE4 status or LDL-C levels on longitudinal changes of CSF tau levels when using models not examining interaction between sex and APOE4 status.
On further stratification by APOE4 status and sex, we compared APOE4 + females to all other subgroups (Table 3), as well as to a pooled comparator group of non (APOE4 positive females) (Table 4). According to Table 3, only in APOE4 + females, higher LDL-C levels were significantly associated with faster cortical thinning in the parahippocampal (β = −0.0075, p = 0.019), medial orbitofrontal (β = −0.0025, p = 0.032), fusiform (β = −0.0066, p = 0.031), posterior cingulate (β = −0.0097, p = 0.007), and inferior temporal regions (β = −0.0085, p = 0.019). A significant association was also observed with MMSE decline (β = −1.409, p = 0.014) as well as increase in CSF t-tau (β = 0.0517, p = 0.036) and p-tau181 (β = 0.0148, p = 0.042). These significant effects were further supported by comparing the LDL-C effect in APOE4-positive females to the pooled non-APOE4-positive female group. Significant interaction effects (i.e., differences between APOE4 + females and the pooled group) were observed in parahippocampal cortex (β = 0.0079, p = 0.028), medial orbitofrontal (β = 0.0027, p = 0.037), and posterior cingulate (β = 0.0099, p = 0.015), as well as CSF t-tau (β = −0.0642, p = 0.026) and p-tau181 (β = −0.0182, p = 0.032). These interactions suggest increased susceptibility of APOE4- positive females to LDL-C effects compared to other groups, as other groups showed less cortical thinning and less increase in CSF tau over time.
Comparison of LDL-C association with cortical atrophy rate, MMSE decline and CSF biomarker changes between APOE4 + females and other subgroups. First column shows LDL-C effect within APOE4 positive females. All other columns reflect interaction terms (differences in LDL-C effects relative to APOE4 positive females).

SE: standard error; LDL-C: low density lipoprotein cholesterol; MMSE: Mini-Mental State Exam; CSF: cerebrospinal fluid.
Comparison of LDL-C association with cortical atrophy rate, MMSE decline and CSF biomarker changes between APOE4 + females and pooled non (APOE4 + females) group. First column shows LDL-C effect within APOE4 positive females. Second column reflect interaction term indicating difference in LDL-C effects between non (APOE4 + females) and APOE4 positive females.

SE: standard error; LDL-C: low density lipoprotein cholesterol; MMSE: Mini-Mental State Exam; CSF: cerebrospinal fluid.
None of the regions survived Bonferroni or false discovery rate (FDR) corrections for multiple comparisons, results are therefore considered exploratory.
On sensitivity analysis stratifying AD patients by statin intake, we found that LDL-C associations with cortical atrophy, MMSE decline and CSF tau in APOE4 positive females, remained significant only in patients not taking statins (N = 56) (Table 5). We found no significant interactions between APOE4 status, female sex and LDL-C in patients taking statins (N = 50). 86% (N = 43) of AD patients taking stating were using statins that were lipophilic and can cross the blood brain barrier.
Comparison of LDL-C association with cortical atrophy rate, MMSE decline and CSF biomarker changes between APOE4 + females and pooled non (APOE4 + females) group among patients not taking statins. First column shows LDL-C effect within APOE4 positive females. Second column reflect interaction term indicating difference in LDL-C effects between non (APOE4 + females) and APOE4 positive females.

SE: standard error; LDL-C: low density lipoprotein cholesterol; MMSE: Mini-Mental State Exam; CSF: cerebrospinal fluid.
Discussion
The results of this study highlight a significant and region-specific association of higher LDL-C levels with neurodegeneration and global cognitive function (MMSE) in APOE4-positive females. The findings demonstrate that within this specific subgroup, elevated LDL-C is consistently associated with accelerated thinning in brain regions such as the parahippocampal, medial orbitofrontal, fusiform, posterior cingulate, and inferior temporal cortices, all of which are known to be vulnerable in neurodegenerative processes in AD.18–25 Furthermore, an association between higher LDL-C and a faster rate of MMSE decline as well as increased CSF tau was observed in APOE4-positive females.
Our study showed that LDL-C was generally higher in females than males, and that its correlation with cortical thinning was significantly stronger in females who were APOE4-positive. This finding indicates the possibility that female sex is a substrate for elevated LDL-C, with APOE4 positivity being the factor that drives its effect on cortical thinning. The biological mechanisms underlying these findings are likely complex. APOE4 is known to impair lipid metabolism, increase amyloid-beta aggregation, and promote neurodegeneration, all of which could be exacerbated by high LDL-C levels in a sex-specific manner. Hormonal factors, particularly estrogen decline in post-menopausal women, could further modulate the brain's response to LDL-C and APOE4, contributing to the heightened vulnerability observed in APOE4-positive females. Although men are generally known to have higher LDL-C levels than women, this difference diminishes—and can even reverse—after menopause due to decreased LDL-C clearance driven by low estrogen levels. Previous studies have shown that postmenopausal status and increasing age in women are key factors contributing to higher LDL-C levels compared to both men and premenopausal women.26–28 This aligns with our finding that women in the AD group exhibited higher LDL-C levels than men. To further investigate this finding considering previous research, we examined the potential interaction between the effects of age and female sex on LDL-C levels in the AD cohort. In line with previous findings of more negative lipid profiles in aging females, there was a trend for females to have higher LDL-C at older ages (β for Female × Age interaction = −0.04, p = 0.03; model adjusted for APOE4 status, lipid-lowering therapy, and BMI). No such interaction was seen in the CN group, which might indicate that female AD patients are more vulnerable to deteriorating lipid profiles.
APOE is essential for the removal of LDL-C and Aβ amyloid. It has been repeatedly demonstrated that the APOE4 variant is less effective in mediating the clearance of both Aβ and LDL-C, which results in their buildup, elevated oxidative stress, cellular damage, and neurodegeneration. According to some research, elevated LDL-C levels may compete with Aβ for clearance by APOE, particularly when APOE4 is already not operating optimally. Because both conditions rely on APOE-mediated receptor pathways, increased LDL-C may worsen Aβ clearance in these situations.29,30
Estrogen has been demonstrated to protect against AD by decreasing the formation of Aβ and improving the brain's lipid metabolism and LDL-C clearance.31–34 Given that female APOE4 carriers may already have compromised Aβ clearance pathways due to APOE4 allele, postmenopausal increases in LDL-C due to decreased estrogen could further exacerbate the effects of APOE4 allele on neurodegeneration. These biological processes could help explain our results and are consistent with earlier studies that found female APOE4 carriers were more susceptible to AD.17–20
This study demonstrated the longitudinal association of LDL-C withcortical thinning in AD patients, while providing more information about the interactions between APOE4 allele, LDL-C levels, and sex in AD, which aids in explaining the varied relationships between LDL-C, APOE4, and AD. Our study showed that the interaction between APOE4 status, female sex and LDL-C is only significant in patients who do not take statins which may highlight a possible protective effect of these medications. However, further studies investigating causative effects on larger samples are needed. As prior research has shown that statins have a larger protective effect on females than on males, these findings may help explain the inconsistent effects of statins on AD risk by emphasizing the significance of stratifying by gender in addition to APOE4 status.35–39 Furthermore, by taking into consideration the interactions between APOE4, LDL-C, and sex, these findings may have additional implications for comprehending the diverse patterns of AD on MRI and clinical outcomes.40,41 The link between hypertension and decreased baseline cortical thickness was another noteworthy finding that was not included in our main hypothesis. This finding emphasizes the significance of general cardiovascular risk factors, not just LDL-C. This supports the idea that managing cardiovascular risk factors could be a dementia prevention strategy and is consistent with earlier research showing cross-sectional and longitudinal relationships between cardiovascular risk and dementia.42,43
One of the study's limitations is the small sample size used to construct stratified models across the four subgroups that were discovered. This reduced the statistical power to detect statistically signficant associations and could account for the fact that none of the regions with significant relationships survived multiple comparison adjustments. Another limitation of this study was that all our AD patients were of mild to moderate severity, limiting generalizability to patients with later disease stage and our ability to detect effects on white matter atrophy. The lack of data on estrogen levels further limited our ability to further test our hypothesis on postmenopausal estrogen decline on LDL metabolism. Future research could benefit from larger, more granular subgroup analyses Longitudinal studies with repeated measures of LDL-C and detailed assessments of other cardiovascular risk factors would provide a more complete picture of the dynamic interplay between lipids, genetics, and brain health. These limitations highlight the exploratory nature of our study and highlight the importance of cautious interpretation of its results.
In conclusion, this study shows that female AD patients carrying the APOE4 allele exhibit increased vulnerability to LDL-C effects on neurodegeneration over time, particularly in regions previously identified as sensitive to APOE4-related neurodegeneration. This structural decline was accompanied by greater cognitive deterioration, suggesting that elevated LDL-C may accelerate disease progression in this high-risk subgroup. While exploratory in nature, this study shows the longitudinal association between LDL-C and regional brain atrophy on MRI and supports the need for sex- and genotype-stratified analyses in AD research. These results may help explain prior inconsistencies in the literature and inform more personalized prevention and therapeutic strategies targeting lipid metabolism in AD.
Supplemental Material
sj-pdf-1-alz-10.1177_13872877261422449 - Supplemental material for Increased vulnerability to low-density lipoprotein cholesterol in apolipoprotein E4-positive females: Longitudinal effects on neurodegeneration and cognition
Supplemental material, sj-pdf-1-alz-10.1177_13872877261422449 for Increased vulnerability to low-density lipoprotein cholesterol in apolipoprotein E4-positive females: Longitudinal effects on neurodegeneration and cognition by Abdul Jaber Tayem, Ahmad Bani Jaber, Ahmad M. Altantawi and Noor M. Masadeh, Laith Al Quran, Omar Al-Jammal, in Journal of Alzheimer's Disease
Supplemental Material
sj-pdf-2-alz-10.1177_13872877261422449 - Supplemental material for Increased vulnerability to low-density lipoprotein cholesterol in apolipoprotein E4-positive females: Longitudinal effects on neurodegeneration and cognition
Supplemental material, sj-pdf-2-alz-10.1177_13872877261422449 for Increased vulnerability to low-density lipoprotein cholesterol in apolipoprotein E4-positive females: Longitudinal effects on neurodegeneration and cognition by Abdul Jaber Tayem, Ahmad Bani Jaber, Ahmad M. Altantawi and Noor M. Masadeh, Laith Al Quran, Omar Al-Jammal, in Journal of Alzheimer's Disease
Supplemental Material
sj-pdf-3-alz-10.1177_13872877261422449 - Supplemental material for Increased vulnerability to low-density lipoprotein cholesterol in apolipoprotein E4-positive females: Longitudinal effects on neurodegeneration and cognition
Supplemental material, sj-pdf-3-alz-10.1177_13872877261422449 for Increased vulnerability to low-density lipoprotein cholesterol in apolipoprotein E4-positive females: Longitudinal effects on neurodegeneration and cognition by Abdul Jaber Tayem, Ahmad Bani Jaber, Ahmad M. Altantawi and Noor M. Masadeh, Laith Al Quran, Omar Al-Jammal, in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
Data collection and sharing for this project was funded by the Alzheimer's Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U19 AG024904 and NIH Grant U01 AG024904; Department of Defense award number W81XWH-12-2-0012). ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through contributions from: AbbVie; Alzheimer's Association; Alzheimer's Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb; CereSpir, Inc.; Cogstate; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd and affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics. The Canadian Institutes of Health Research additionally supports ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health. The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer's Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
ORCID iDs
Ethical considerations
This study was conducted using de-identified data from the Alzheimer's Disease Neuroimaging Initiative (ADNI) database. As such, it was not considered human subjects research and did not require institutional review board (IRB) approval.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Data collection and sharing for this project was funded by the Alzheimer's Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12-2-0012).
The authors received no specific grant from any funding agency for the analysis and writing of this manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare no conflicts of interest. Data used in this study were obtained from the Alzheimer's Disease Neuroimaging Initiative (ADNI), and the authors received no direct funding for data analysis or manuscript preparation.
Data availability statement
The data used in this study were obtained from the Alzheimer's Disease Neuroimaging Initiative (ADNI) database (http://adni.loni.usc.edu). Access to the data is available to researchers upon application and approval by the ADNI Data Sharing and Publications Committee. Further details on data access procedures can be found at: ![]() .
.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
