Abstract
Background:
The Montreal Cognitive Assessment (MoCA) is recommended by the Movement Disorder Society for cognitive testing in movement disorders including Parkinson’s disease (PD) and lewy body dementia. Few studies have compared cognitive screening instruments in these diseases, which overlap clinically.
Objective:
To compare the MoCA and Quick Mild Cognitive Impairment (Q
Methods:
Patients attending memory and movement disorder clinics associated with a university hospital had the MoCA and Q
Results:
In total, 133 assessments were available, median age 74±5. Median education was 11±4 years and 65% were male. Median total UPDRS score was 37±26. Median Q
Conclusions:
Both the MoCA and Q
Keywords
INTRODUCTION
The prevalence of movement disorders is increasing in tandem with population aging [1]. Approximately 28% of adults aged over 50 years can be classified as having a movement disorder, most commonly a tremor [2]. One of the most prevalent movement disorders worldwide is Parkinson’s disease (PD); the number of persons with PD is expected to double by 2030 [3] with an increasing incidence and burden associated with this condition in most regions and countries globally [4]. As cognitive impairment (CI) is common in PD, particularly as the disease advances, this will result in an overall increase in the number of patients reporting cognitive symptoms associated with PD. Many of these will be older patients. Prevalence rates of PD-related CI approach 80% [5], while the rate of progression to Parkinson’s disease dementia (PDD) is approximately 10% per year [6]. Mean duration from onset of PD symptoms to dementia is estimated at 10 years [7]. Many patients with PD have CI from the time of diagnosis (approximately 15–20%), though the prevalence in prodromal disease is less clear [8]. Lewy body dementia (LBD) is one of the most common dementia subtypes and is also associated with the development of a movement disorder [9]. Similar to PD, LBD is a dementia associated with parkinsonism and Lewy body formation and it likewise increases in prevalence with age, representing approximately 5% of dementia cases in older populations [9, 10]. As with CI in PD, it is likely under-reported [10].
Mild cognitive impairment (MCI) affects at least one-third (27%–42.5%) of people with PD [11–13] and is associated with a greater chance of developing dementia, especially if present when patients are first diagnosed [14]. Single domain, non-amnestic is the most common subtype [12]. The diagnosis of PD-MCI is challenging with uncertainty surrounding the duration of follow-up and number of repeated cognitive tests required to make the diagnosis [15]. Reflecting this, some guidelines including the Movement Disorder Society Task Force Guidelines [11], suggest that up to a minimum of 10 repeat assessments are required to diagnose and subtype PD-MCI [15]. PD-MCI features exaggerated attention and executive function deficits [16]. LBD is also associated with an MCI syndrome referred to as LBD-MCI, which, similar to PD-MCI, is characterized by executive, visuospatial, and attentional deficits [17, 18]. CI in persons with movement disorders impacts upon life expectancy [19], quality of life [20], healthcare costs [21] and activities of daily living (ADLs), even among those without dementia, i.e., at prodromal stages [20].
Early diagnosis of CI is important to facilitate prompt treatment, identify reversible or compounding factors and plan for the future [22]. Monitoring change over time is important in this context and relies on the use of accurate short cognitive screening instruments (CSIs) [23]. Repeated neuropsychological testing [14] is recommended to increase prognostic accuracy, particularly conversion from MCI syndromes to clinical dementia. The progression from PD-MCI and LBD-MCI to dementia is particularly challenging to diagnose. An ideal short CSI in this setting would be reliable, brief, sensitive to early change, have normative data available, cover core cognitive domains relevant to these conditions (i.e., attention-working memory, memory, executive functioning, visuospatial skills, and language) and be largely immune to the effects of motor limitations [23, 24]. Such CSIs would also ideally correlate well with functional measures and emerging biomarkers [23].
At present, the Movement Disorder Society recommends the use of the Montreal Cognitive Assessment (MoCA) [25] for cognitive testing in movement disorders including PD and LBD [26–28]. The reason why the MoCA is well suited for use in this population is because it lacks ceiling effects and is weighted towards domains such as executive and visuospatial functioning and less towards orientation and language, which are relatively well preserved in PD [29]. Other widely-used CSIs may be less suitable. For example, the Mini-Mental State Examination (MMSE), one of the most established and widely-used CSIs, is still commonly used for these patients [28, 30]. However, the MMSE, because of its low ceiling effects and selected cognitive domains, may miss early cognitive deficits in these conditions [29] and is recommended for use only with this caveat [28]. This may result in failed opportunities to initiate early appropriate treatment and discontinue inappropriate medications like anti-cholinergics (medications that bind to muscarinic receptors and block acetylcholine neurotransmission), which is particularly important in diseases associated with a cholinergic deficit such as Alzheimer’s disease, PDD and LBD [31]. Despite the increasing use of the MoCA, questions remain about its accuracy; studies suggest that up to one-quarter of patients with PD with normal MoCA scores report functional cognitive difficulties, while three-quarters with low MoCA scores report none [32]. On this basis, other tests with a greater emphasis on visio-cognitive impairment, may be preferable[33].
Despite the advantages of the MoCA and its extensive validation [34], some challenges remain with its use in persons with movement disorders and other populations such as in primary care and community-based memory clinics (where access to neuropsychology may be limited) including elements of redundancy, a relatively long administration time for a CSI (10–12 min), its known floor effects, its low specificity and a high false positive rate in older adults, particularly at its recommended cut-off score (≥26) [35–39]. Given this, we sought to investigate if the Quick Mild Cognitive Impairment (Q
MATERIALS AND METHODS
Patients
This is a secondary analysis of a cross-sectional study that was conducted parallel to a larger study evaluating the Q
Data collection
Patients underwent comprehensive clinical assessment and were screened for cognitive impairment. This assessment included a full history, physical examination, laboratory testing and neuroimaging. A short neuropsychological battery including the Standardized MMSE [47] and two informant-rated assessments, the AD8 questionnaire [48] and IQCODE Short Form [49] were conducted by a consultant geriatrician, blind to the results of the CSIs, to inform the clinical diagnosis. The MoCA and Q
Measures
Montreal cognitive assessment
The MoCA has seven subtests covering five cognitive domains; specifically, it includes visuospatial, attention, processing speed, language, memory, and cognitive control scored out of 30 points with lower points indicating cognitive impairment. A cut-off of < 26/30 is suggested for use in routine practice [25], although lower cut-offs have been suggested. Lower scores imply more impaired cognition.
Quick mild cognitive impairment screen
The Q
Unified Parkinson’s disease rating scale
The severity of PD was determined using the UPDRS [51]. The UPDRS total score consists of the sum of parts I (mentation, behavior, and mood), II (ADLs) and III (motor examination), with scores ranging from 0 (not affected) to 176 (most severely affected). A score of one or more on item one (intellectual impairment) of part I was taken as supportive of CI (PD-MCI or PDD).
Analysis
Data were analyzed using R version 4.2.2 (2022-10-31) -“
RESULTS
In all, 133 patient assessments were available and included. In addition to those excluded with other movement disorders, one duplicate was removed. The characteristics of patients according to their diagnostic category are presented in Table 1. The median age of the sample was 74 years, interquartile range (IQR)±5. Most, 65%, were male. The median number of years in education was 11 IQR±4 years. The majority of patients were diagnosed with parkinsonism with 109 (82%) having idiopathic PD and 11 (8%) having vascular-type parkinsonism. The remaining 10% (
Comparison of characteristics of patients with normal cognition, mild cognitive impairment (MCI) and dementia
IQR, Interquartile range. *Statistically significant. UPDRS, Unified Parkinson’s Disease Rating Scale; SMMSE, Standardized Mini-Mental State Examination; Q
A strong, positive correlation was seen between both CSIs,
Table 2 shows the optimal cut-off scores with sensitivity and specificity values according to Youden’s Index for both CSIs for different comparisons. The diagnostic accuracy based on AUC are also provided and presented visually as ROC curves in Fig. 1. The Q
Sensitivity, Specificity, and diagnostic accuracy for each cognitive screening instrument based upon area under the receiver operating characteristic curve (AUC) with 95% confidence intervals, for the optimal cut-off scores for cognitive impairment
Q
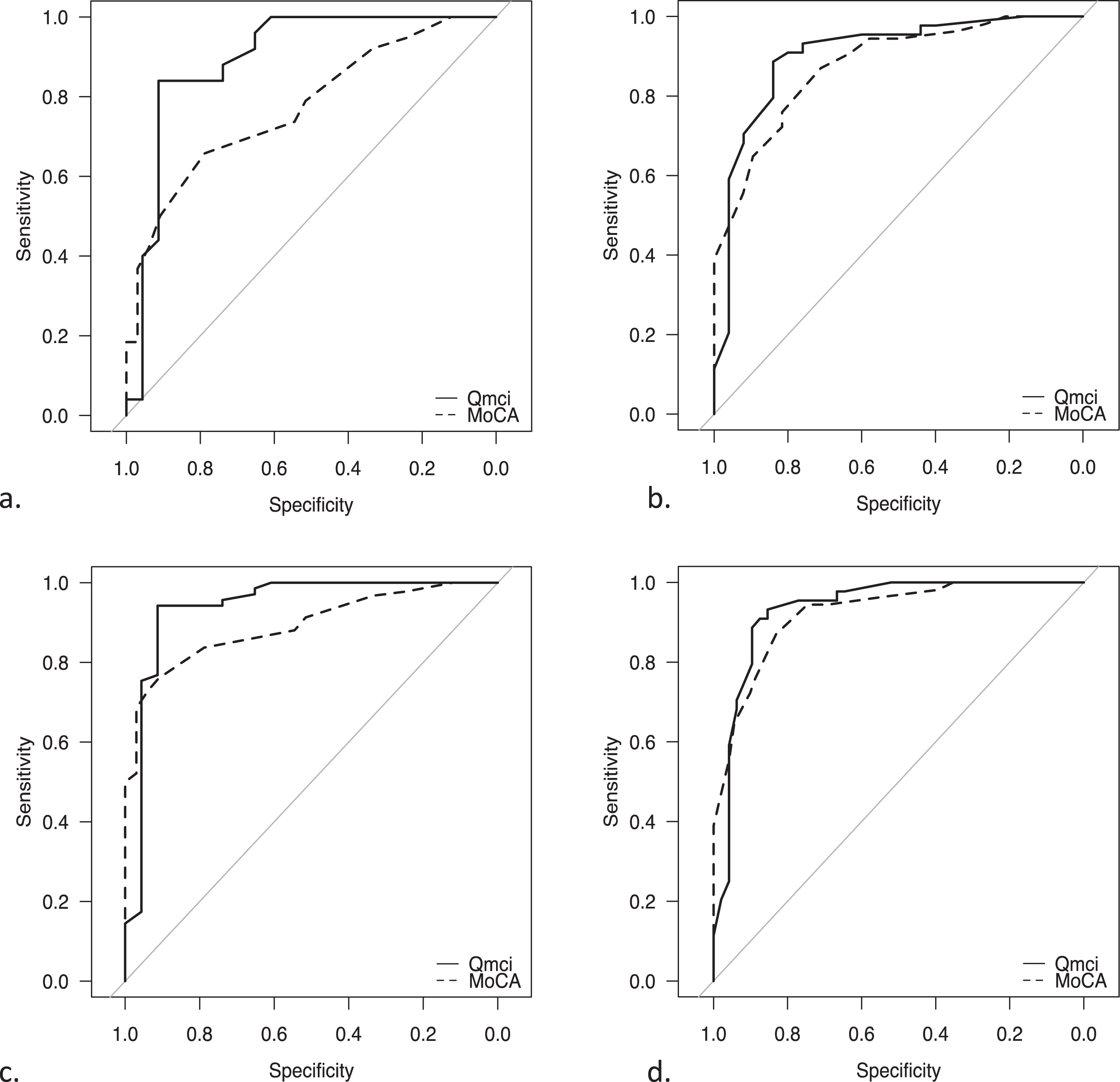
Receiver Operating Characteristic curves demonstrating the accuracy of the Quick Mild Cognitive Impairment (Q
DISCUSSION
This study compares the diagnostic accuracy of two short CSIs, the MoCA and the Q
This middle-aged and older sample was predominantly comprised of patients with idiopathic PD and despite a moderate median UPDRS (total) scores, a large proportion (75%) of this sample had CI. Given that the median time since diagnosis in this study to the date of the assessment was seven years and this is likely an under-estimation, this likely explains the high prevalence of CI found in this sample. Evidence suggests that the mean time from PD onset to PDD is approximately 10 years [54]. While this study included middle-aged adults > 40 years, the median age of the sample was much older (median age 74 years±5), likely reflecting the service (geriatrician-led clinics). Most patients, 65%, were male, consistent with the higher prevalence of movement disorders and in particular PD, among male patients [55].
The optimal cut-off for the Q
Limitations
This study has a number of limitations. As this study was a secondary analysis of an existing database (gathered approximately ten years ago), rather than a primary data collection and thus lacks
Further research comparing the Q
In conclusion, the Q
AUTHOR CONTRIBUTIONS
Rónán O’Caoimh (Conceptualization; Data curation; Formal analysis; Writing – original draft); Mary J. Foley (Investigation); Suzanne Timmons (Supervision); D. William Molloy (Supervision).
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
The authors have no funding to report.
CONFLICT OF INTEREST
Dr. Rónán O’Caoimh and Prof. D. William Molloy are co-copyright holders of the Quick Mild Cognitive Impairment Screen. The authors report no other conflict of interest.
DATA AVAILABILITY
Available on request.
