Abstract
Background:
Physicians may soon be able to diagnose Alzheimer’s disease (AD) in its early stages using fluid biomarkers like amyloid. However, it is acknowledged that additional biomarkers need to be characterized which would facilitate earlier monitoring of AD pathogenesis.
Objective:
To determine if a potential novel inflammation biomarker for AD, symmetric dimethylarginine, has utility as a baseline serum biomarker for discriminating prodromal AD from cognitively unimpaired controls in comparison to cerebrospinal fluid amyloid-β42 (Aβ42).
Methods:
Data including demographics, magnetic resonance imaging and fluorodeoxyglucose-positron emission tomography scans, Mini-Mental State Examination and Functional Activities Questionnaire scores, and biomarker concentrations were obtained from the Alzheimer’s Disease Neuroimaging Initiative for a total of 146 prodromal AD participants and 108 cognitively unimpaired controls.
Results:
Aβ42 (
Conclusions:
Correlations were smaller between Aβ42 and neuropathological biomarkers over time and were absent in prodromal AD participants, suggesting a plateau effect dependent on age and disease stage. Evidence supporting symmetric dimethylarginine as a novel biomarker for AD as a single measurement was not found.
INTRODUCTION
Alzheimer’s disease (AD) dementia is an incurable neurodegenerative disease of the brain most often characterized by relentless memory loss, cognitive decline, and hypometabolism and neurodegeneration. A current challenge faced by health care providers is the early diagnosis of AD, particularly when individuals are in the mild cognitive impairment (MCI) stage or even earlier during the asymptomatic phase of the disease [1]. To that end, substantial efforts are centered on developing and identifying fluid biomarkers that are indicative of future cognitive decline. Given its importance in AD dementia and in particular, in the context of anti-amyloid therapies, several studies have centered on determining the utility of fluid biomarkers of amyloid, including biomarkers for amyloid-β42 (Aβ42), with the closely related Aβ40 first being sequenced in AD dementia and Down’s syndrome participants approximately twenty years ago and was later discovered to be the primary component in AD plaques [2].
In recent years, cerebrospinal fluid (CSF) Aβ42 or Aβ42/40 have emerged as promising biomarkers for detection of AD dementia pathology for enrichment of clinical trials and potentially for clinical practice given that Aβ42 is a surrogate marker for Aβ aggregation into senile plaques and that these CSF biomarkers have a relatively close association with amyloid pathology as measured with amyloid positron emission tomography (PET) [3, 4]. While some studies suggest that the Aβ42/40 index does not improve diagnostic accuracy compared to Aβ42 alone [5, 6], the ratio accounts for both production of amyloid as well as deposition, and the ratio is more closely associated with amyloid PET assessments [7]. Identifying the best biomarkers for clinical trial selection is critical, as improved treatments for AD dementia are expected in the coming years. The recently approved drug Aducanumab was found to reduce amyloid plaques in two international phase III randomized controlled trials known as ENGAGE and EMERGE, although ENGAGE found no cognitive improvement and the clinical improvement found in EMERGE was only modest at best [8]. While the United State Food and Drug Administration’s (FDA) Peripheral and Central Nervous System Drugs Advisory Committee voted there was insufficient evidence of efficacy, the FDA accelerated its approval even as roughly 35% of participants suffered amyloid-related imaging abnormalities which include edema and brain hemorrhages [8]. More promise has been observed with recent monoclonal antibody treatments. Lecanemab, for example, has been shown to reduce markers of amyloid in early AD with patients having less cognitive and functional decline, though this treatment also suffered a high prevalence of adverse effects [9]. The FDA has at this time fully approved Lecanemab for the mild dementia stage of AD [10].
In addition to amyloid pathology, developments continue for additional biomarkers that relate to other aspects of AD pathophysiology and that could be used to improve early detection of AD dementia, as well as serve as outcomes in future clinical trials. Innate immune activation from genome-wide association studies have found associations between immune receptor genes and AD pathogenesis [11, 12]. Aging is also associated with decline in adaptive immunity and anti-inflammatory marker production, hence a potential reason why older age is a risk factor for AD [13]. Bacterial and viral infections, especially among periodontal infections and H. pylori, have been associated with AD due to the release of neuroinflammatory interleukins and inflammasomes [14], with Aβ42 even having antimicrobial properties against periodontal bacteria [15]. Glial activation and associated neuroinflammation has long been recognized to play a role in AD. The activation of microglia and other immune cells has been implicated in the exacerbation of both amyloid and tau pathologies, while at the same time, a more systematic proinflammatory mechanism has been proposed as the common thread across risk factors of AD like diabetes mellitus, cardiovascular disease and atherosclerosis, among others [16]. In chronic kidney disease for example, worsening stages of the disease were associated with higher AD and vascular dementia risk [17]. Reasons proposed for an association between chronic kidney disease and AD include the kidneys being unable to excrete uremic toxins, which are inflammatory, as well as the kidney’s involvement with blood pressure, erythropoietin and vitamin D production, two factors that are both neuroprotective and anti-inflammatory [18].
Given these observations, it is important to test novel biomarkers that act as predictors of non-amyloid processes linked to the natural history of AD; this would include, among others, neuroinflammation and neurodegeneration. One of such potentially useful biomarkers is symmetric dimethylarginine (SDMA), an enantiomer of asymmetric dimethylarginine (ADMA) which has been heavily studied in AD in addition to chronic kidney disease and cardiovascular disease. SDMA is a biomarker for and a proinflammatory agent in chronic kidney disease but has also been correlated with neuropathological progression and cognitive decline in prodromal AD dementia participants [19–21]. It is significantly altered in participants with early and progressive stages of AD dementia when compared to controls [21, 22], and was also correlated with neurofilament light chain, a marker for neurodegeneration, in both Aβ– and Aβ+ groups [23]. SDMA was also increased in AD frontal cortex tissue samples [24]. As McEvoy et al. stated, SDMA may be associated with memory impairment through inflammation and an inflammatory state [25]. SDMA has also been linked to the proinflammatory state that takes place after an acute stroke [26]. It is our intention therefore to compare this novel blood constituent and its relationship to prodromal AD dementia against CSF Aβ42. Aβ42 is one of the core biochemical markers for the amyloidogenic process in AD as supported by the 2018 National Institute of Aging and Alzheimer’s Association Research Framework [27]. Additionally, notwithstanding the probable limitations of CSF Aβ42 as a lone measure in preclinical stages of AD dementia and MCI, we found it a suitable initial candidate to compare SDMA to, as it would allow us to revisit any lingering questions around the use of Aβ42 use in the earliest stages of the disease. We hypothesized that SDMA would be more effective than Aβ42 at discriminating between cognitively unimpaired controls and prodromal AD dementia and that either biomarker would be associated with brain hypometabolism and atrophy as well as cognition and function.
MATERIALS AND METHODS
Data source
Data for this study was obtained from the Alzheimer’s Disease Neuroimaging Initiative (ADNI). ADNI is an ongoing, longitudinal study launched in 2003 as a public–private partnership, led by principal investigator Michael W. Weiner, MD. The primary goal of ADNI has been to test the extent to which serial magnetic resonance imaging (MRI), PET, other biological markers, and clinical and neuropsychological assessments provide utility for measuring the progression of MCI and early AD dementia, and to validate brain imaging, blood tests, and other diagnostics, as well as share data with the scientific community. For up-to-date information, see https://adni.loni.usc.edu/. Details on study design, participant recruitment, study approval, and informed consent procedures have been published previously [28]. The study was approved by the institutional review boards of all of the participating institutions/study sites. Informed written consent was obtained from all participants at each site. Metabolomics data for ADNI samples were generated by the Alzheimer Disease Metabolomics Consortium (ADMC) and deposited to the Laboratory of Neuroimaging (LONI). The mission of the ADMC is to create a comprehensive metabolomics database for AD. ADNI data used in the preparation of this article were also obtained from the ADNI-1 database (https://adni.loni.usc.edu) and included baseline blood serum metabolite concentrations (with concurrent structural MRI data) on 767 participants and concurrent CSF AD biomarker data on 403 participants. All ADNI studies are conducted according to the Good Clinical Practice guidelines, the Declaration of Helsinki, and U.S. 21 CFR Part 50 (Protection of Human Subjects), and Part 56 (Institutional Review Boards). Written informed consent was obtained from all participants before protocol-specific procedures were performed. ADNI was enriched with participants with MCI, who represent “prodromal AD”. In this study, participants with MCI at baseline who subsequently converted back to normal cognition were excluded.
Participant cohort and assessments
The primary objective of this retrospective study is to determine if early baseline serum SDMA and CSF Aβ42 levels could be used as reliable predictors for future AD dementia development. The reason for not conducting serum to serum comparisons is that the sample size of participants in ADNI that have both serum constituents is low and would have drastically reduced the power of our analysis. Additionally, CSF Aβ42 remains highlighted as one of the most signature diagnostic markers according to the National Institute of Aging - Alzheimer’s Association [27]. To obtain this data, research staff collected baseline subject level demographic information including age, sex, apolipoprotein E (
Statistical analysis
Descriptive statistics were used to report baseline subject characteristics, and chi-square or student
To study adjusted associations in neurocognitive function
For partial correlations, tests which showed statistical significance with Bonferroni correction for multiple comparisons of FDG, MRI, and cognition (3 FDG regions×2 visits, 4 MRI regions×2 visits, 2 cognitive scores×2 visits) were defined as
RESULTS
Subject characteristics are outlined in Table 1. Of the following demographic predictors—age, sex, subject education level, and
Group characteristics
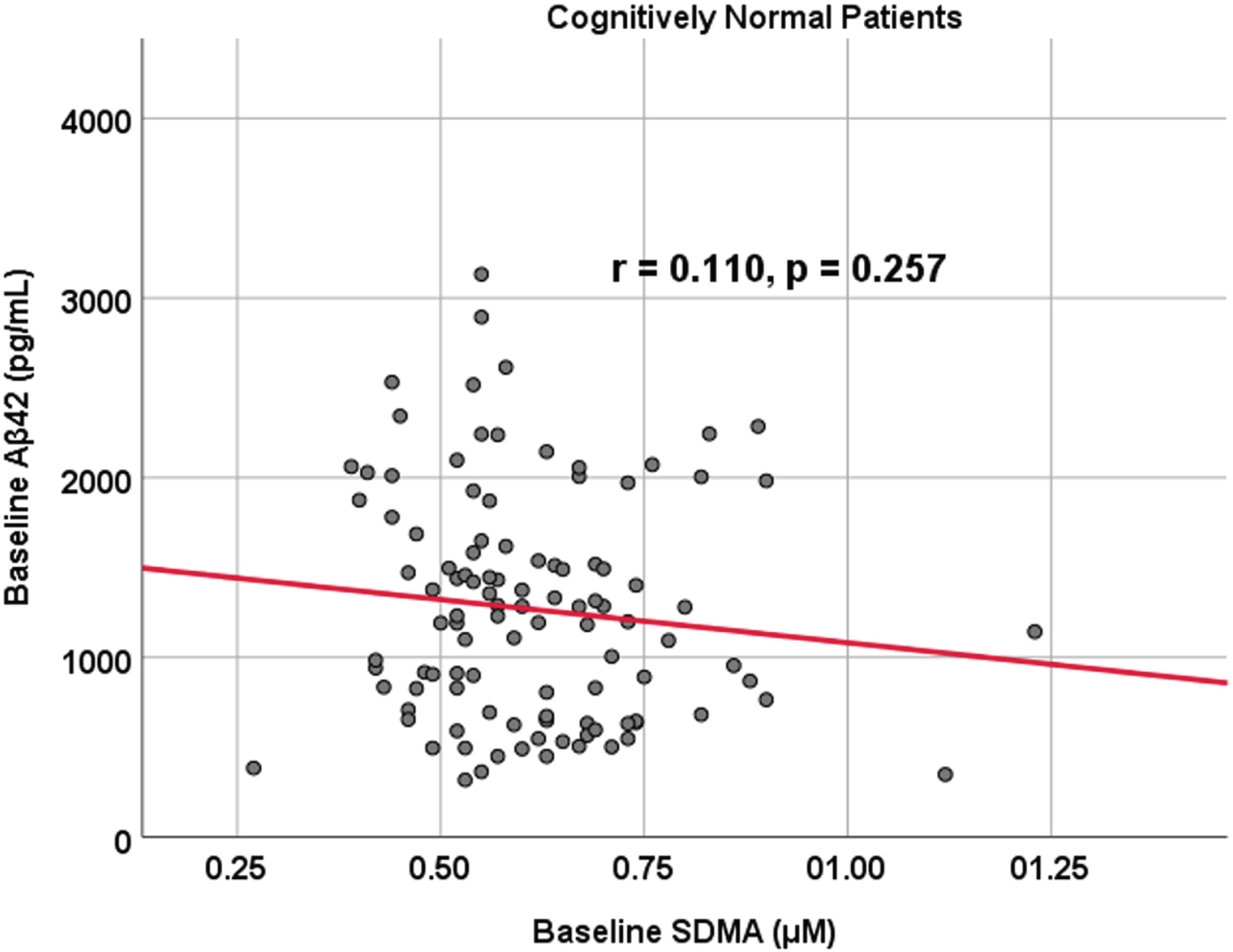
Associations between baseline Aβ42 and SDMA in cognitively unimpaired control participants. Scatterplots above are uncorrected for age, sex, education, and
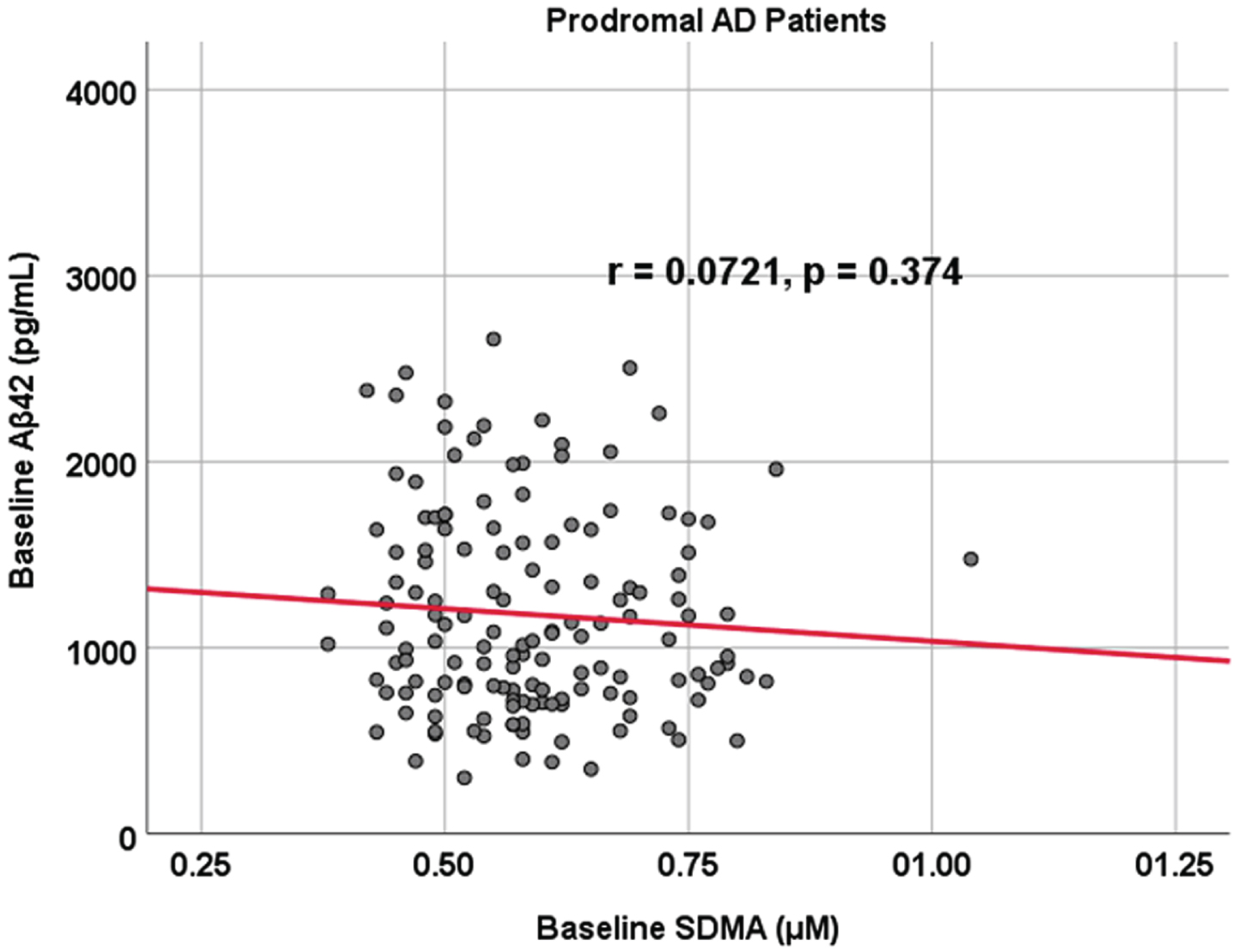
Associations between baseline Aβ42 and SDMA in prodromal AD dementia participants. Scatterplots above are uncorrected for age, sex, education, and
Because AD dementia is an age-dependent disease, we analyzed the relationship between age and biomarker levels in cognitively unimpaired control and prodromal AD dementia subject cohorts (Figs. 3 and 4). We observed a positive correlation between age and SDMA levels in both cognitively unimpaired control (
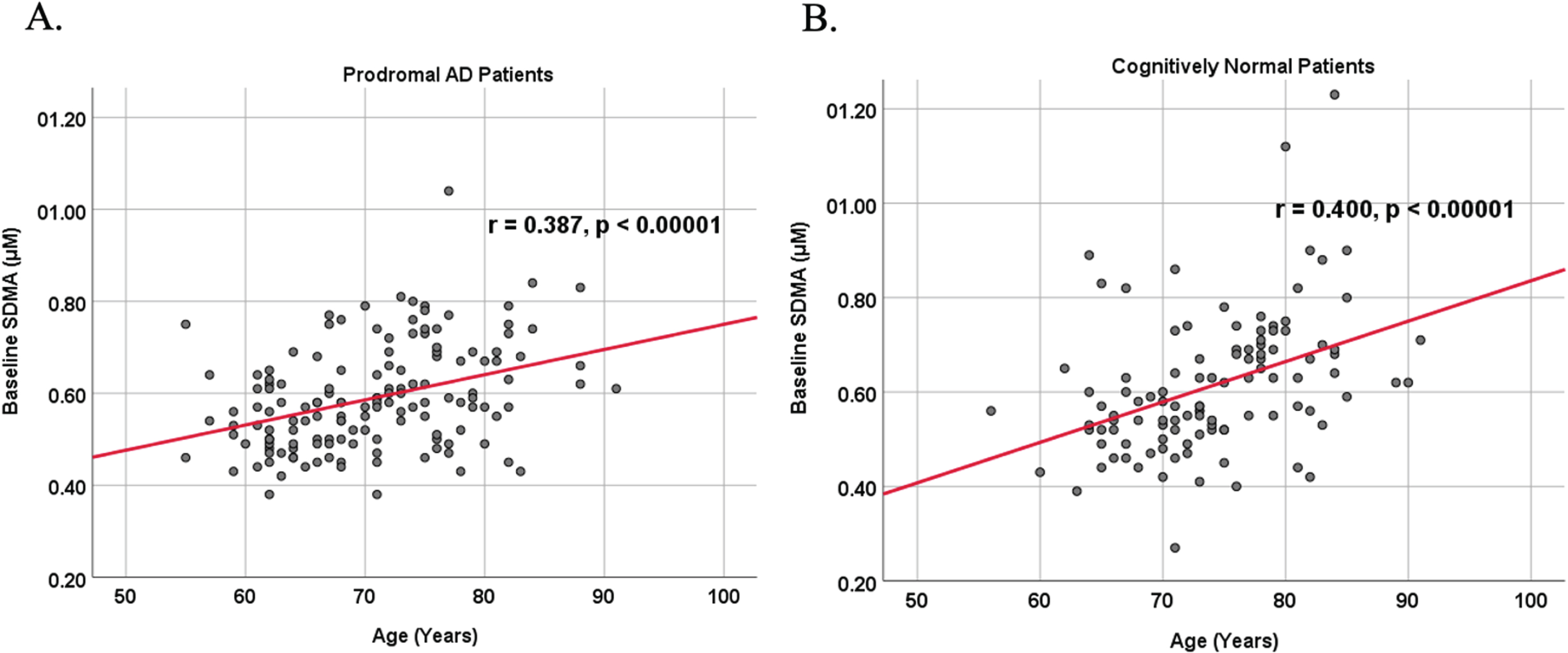
Associations between baseline SDMA and age in cognitively unimpaired control and prodromal participants. Scatterplots above are uncorrected for sex, education, and APOE. A) SDMA versus Age in prodromal AD dementia participants (
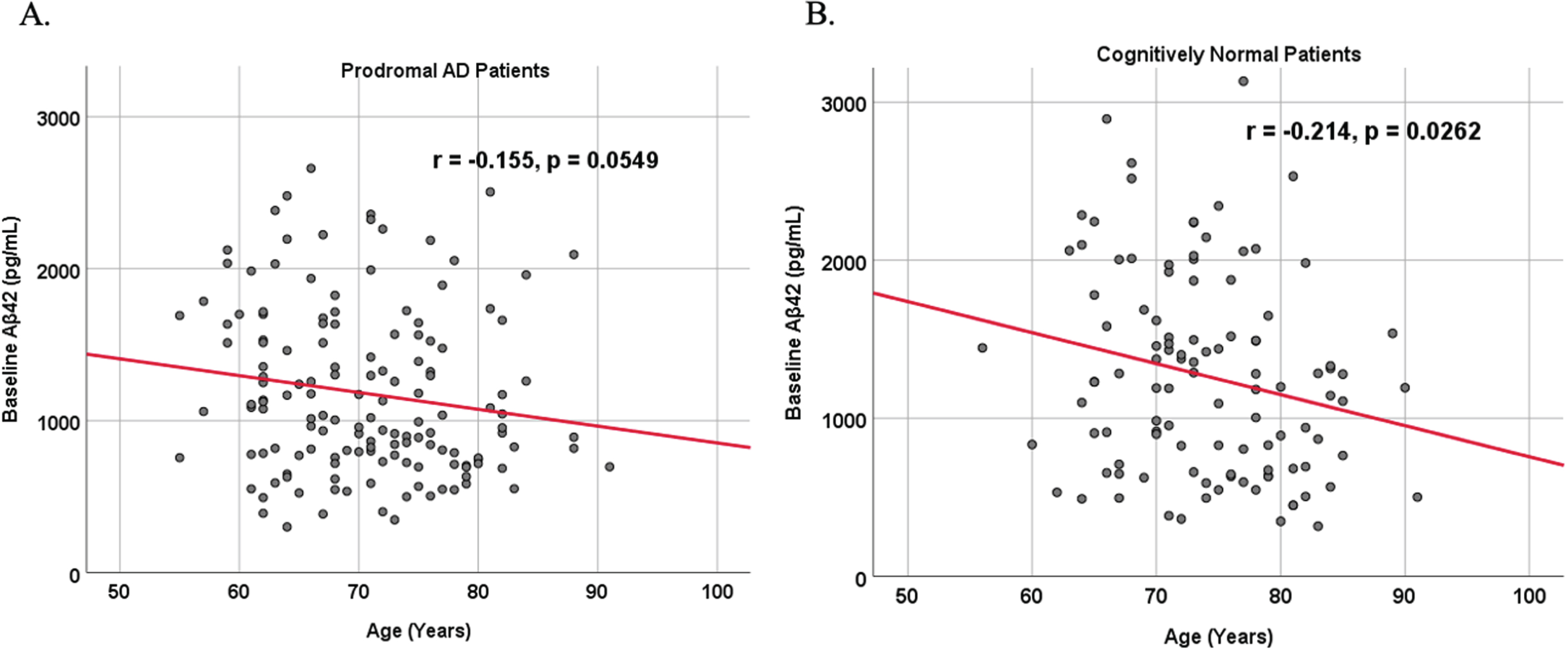
Associations between baseline Aβ42 and age in cognitively unimpaired control and prodromal participants. Scatterplots above are uncorrected for sex, education, and APOE. A) Aβ42 versus Age in prodromal AD dementia participants (
To understand if SDMA or Aβ42 as well as its interactions are associated with prodromal subject status, three multivariable logistic analyses were conducted adjusting for age: one looking at SDMA levels, another at Aβ42 levels, and one at both together (Tables 3–5). Of note is that univariable analysis found baseline FAQ score to be able to significantly predict prodromal AD dementia participants and healthy controls (
Univariable logistic regression
FAQ, Functional Activities Questionnaire; BL, Baseline Level; 24M, 24 months; MMSE, Mini-Mental State Examination;
Multivariable logistic regression including SDMA
SDMA, symmetric dimethylarginine; BL, Baseline Level; 24M, 24 months.
Multivariable logistic regression including Aβ42
BL, Baseline Level; 24M, 24 months.
Multivariable logistic regression including SDMA×Aβ42
FAQ, Functional Activities Questionnaire; BL, Baseline Level; 24M, 24 months.
SDMA, symmetric dimethylarginine; BL, Baseline Level; FAQ, Functional Activities Questionnaire.
To analyze for relationships between biomarker levels and cognitive function, scatterplots and associations were computed (Figs. 5–7). Note that a lower MMSE score indicates cognitive impairment, whereas a higher FAQ score may indicate cognitive impairment. A positive relationship was found in cognitively unimpaired participants between baseline Aβ42 and baseline MMSE (
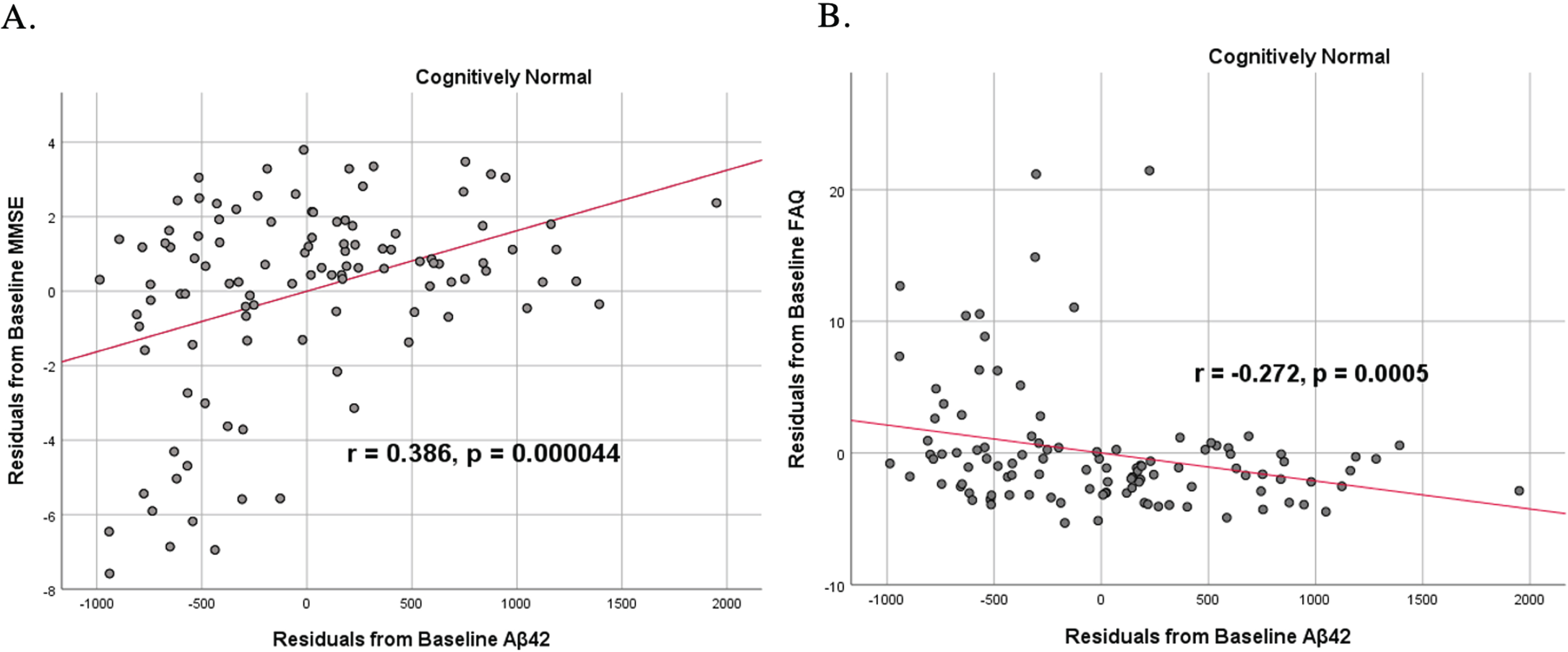
Partial correlations between baseline Aβ42 concentration and baseline neurocognitive function in normal controls, corrected for age and education. Scatterplots above are corrected for age and education. A) Aβ42 versus MMSE (
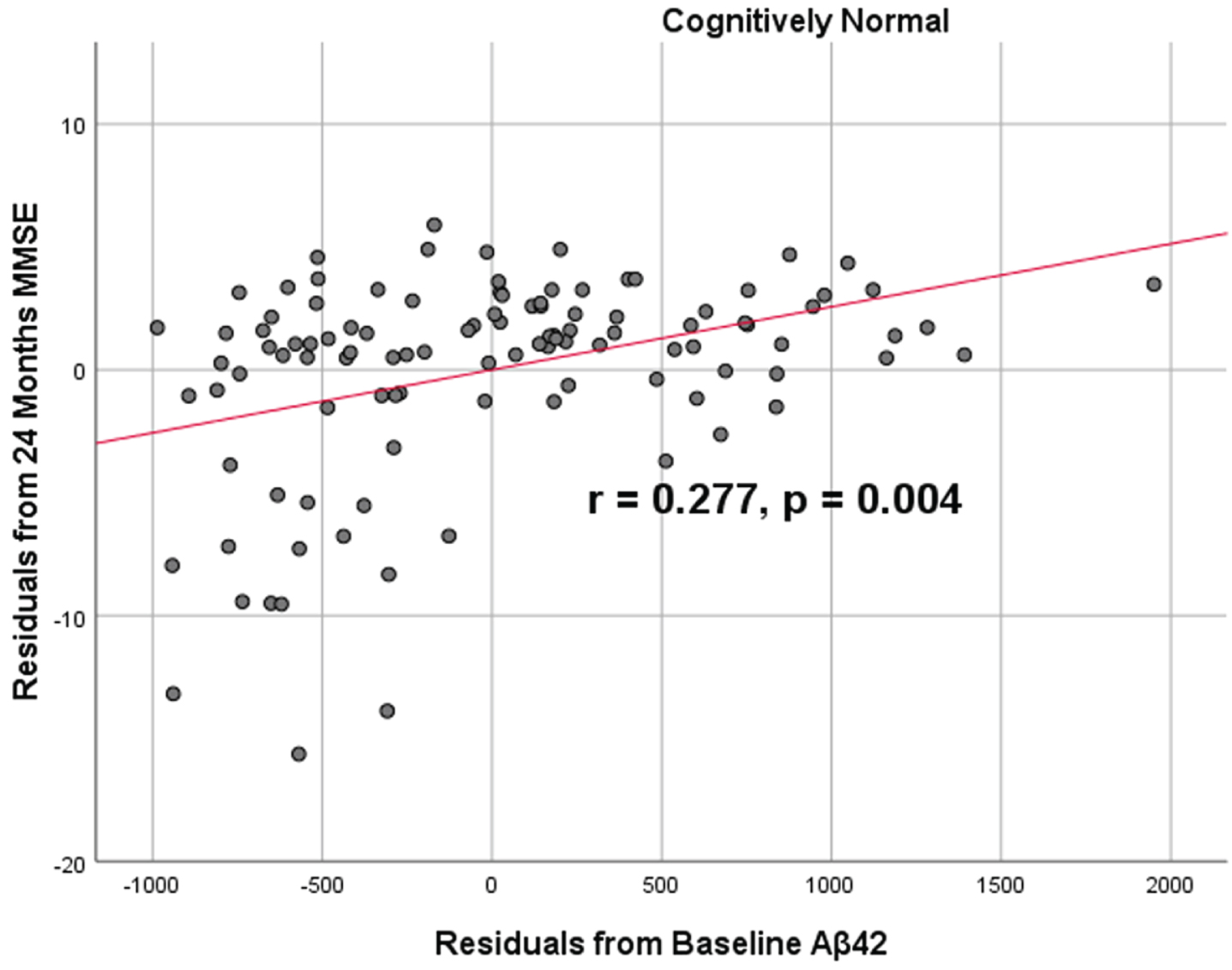
Partial correlations between baseline Aβ42 concentration and 24 months neurocognitive function in normal controls, corrected for age and education. Scatterplots above are corrected for age and education. Aβ42 versus MMSE (
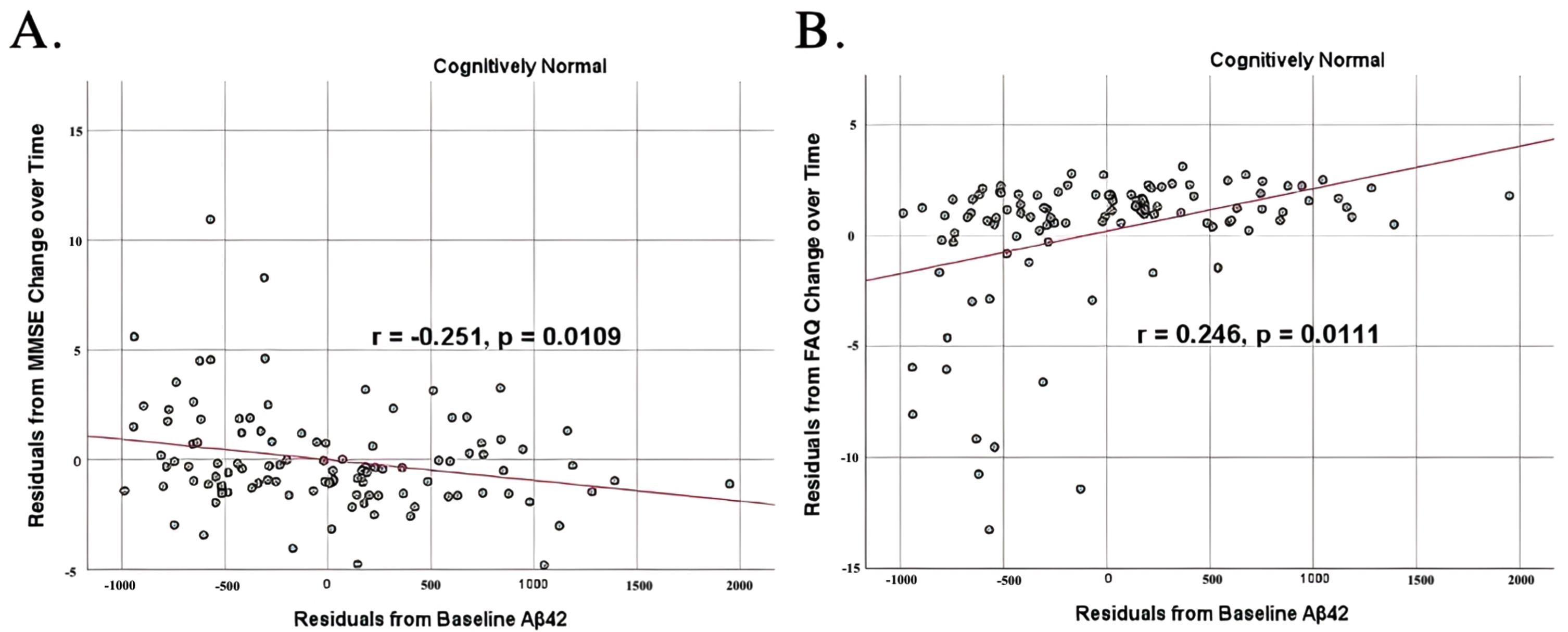
Partial correlations between baseline Aβ42 concentration and neurocognitive function decline in normal controls, corrected for age and education. Scatterplots above are corrected for age and education. A) Aβ42 versus MMSE (
To analyze for relationships between biomarker levels and regional neuroimaging assessments, scatterplots and associations were computed (Figs. 8–12). See Supplementary Figures for scatterplots and associations across all participants. Positive relationships were found in cognitively unimpaired participants between baseline Aβ42 and posterior cingulum metabolism (
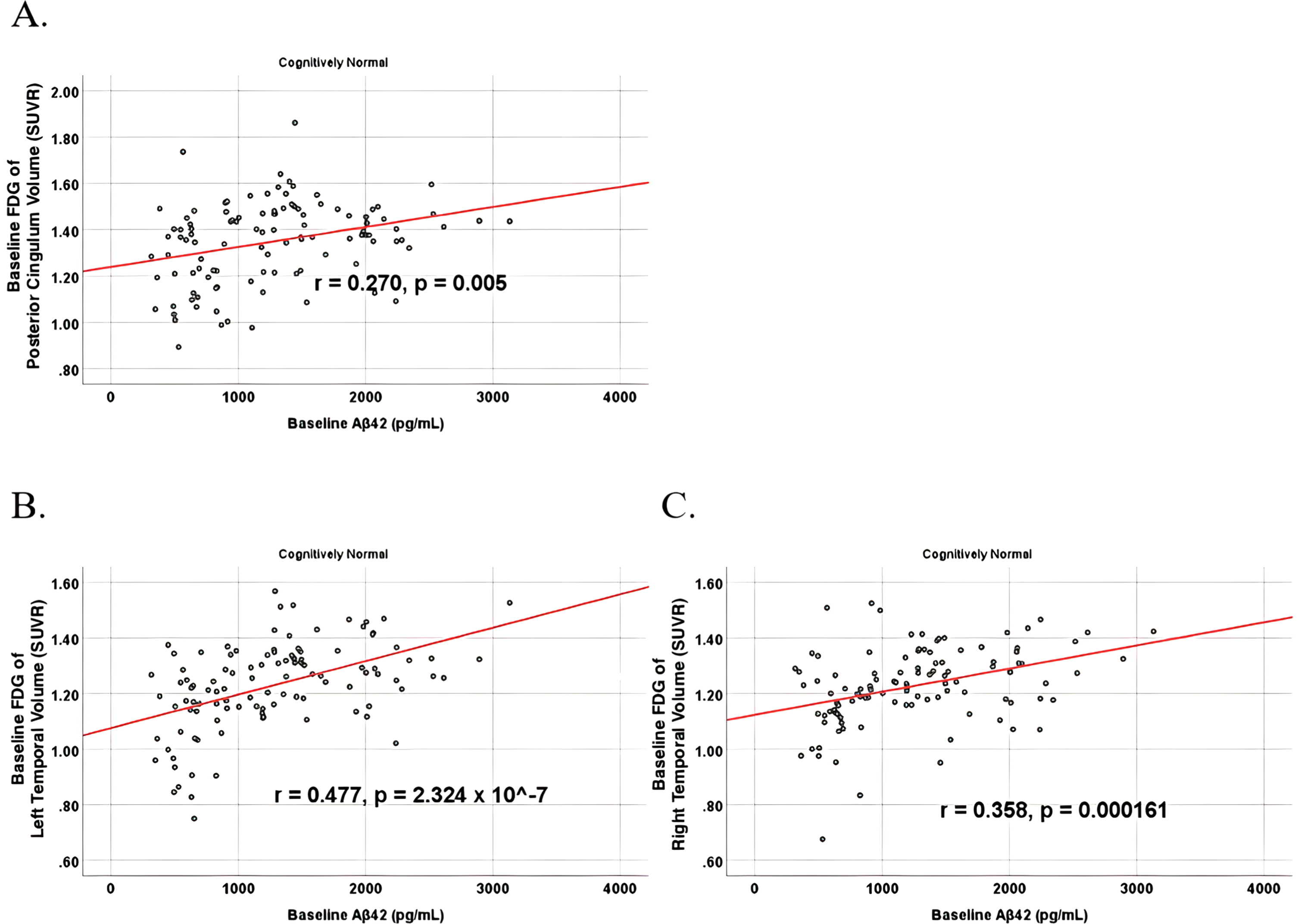
Partial correlations between baseline Aβ42 and baseline FDG brain volume regions in cognitively unimpaired control participants, corrected for age and education. Scatterplots above are corrected for age and education. A) Aβ42 versus Posterior Cingulum (
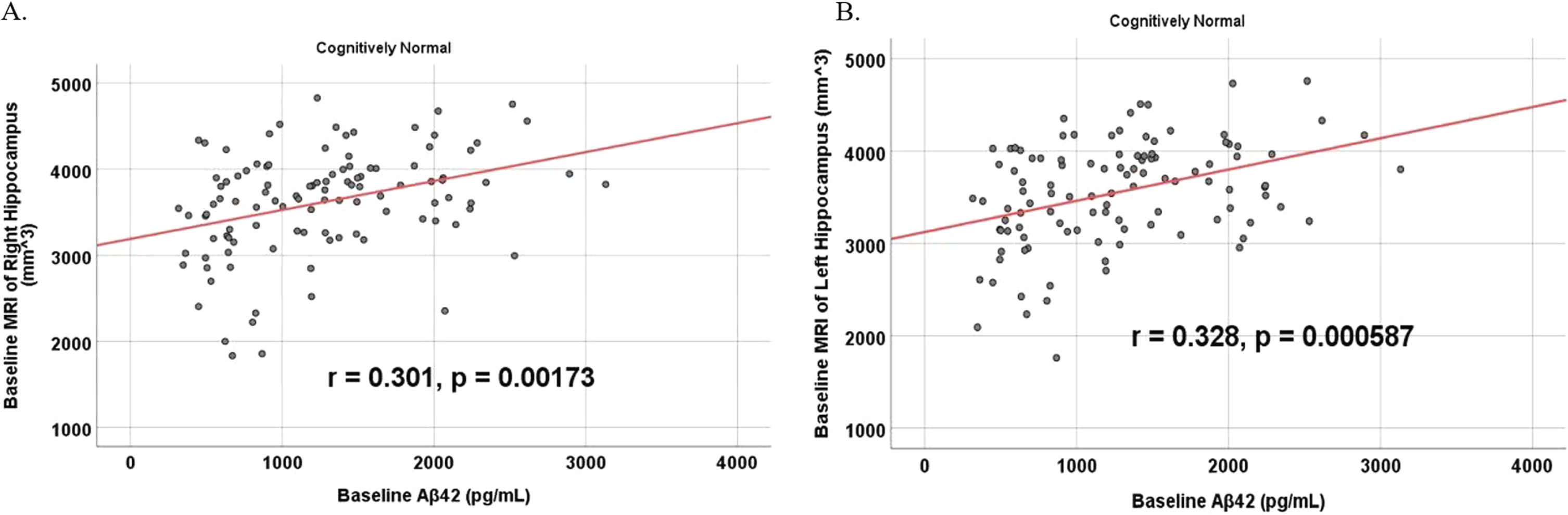
Partial correlations between baseline Aβ42 and baseline MRI brain volume regions in cognitively unimpaired control participants, corrected for age and education. Scatterplots above are corrected for age and education. A) Aβ42 versus Right Hippocampus (
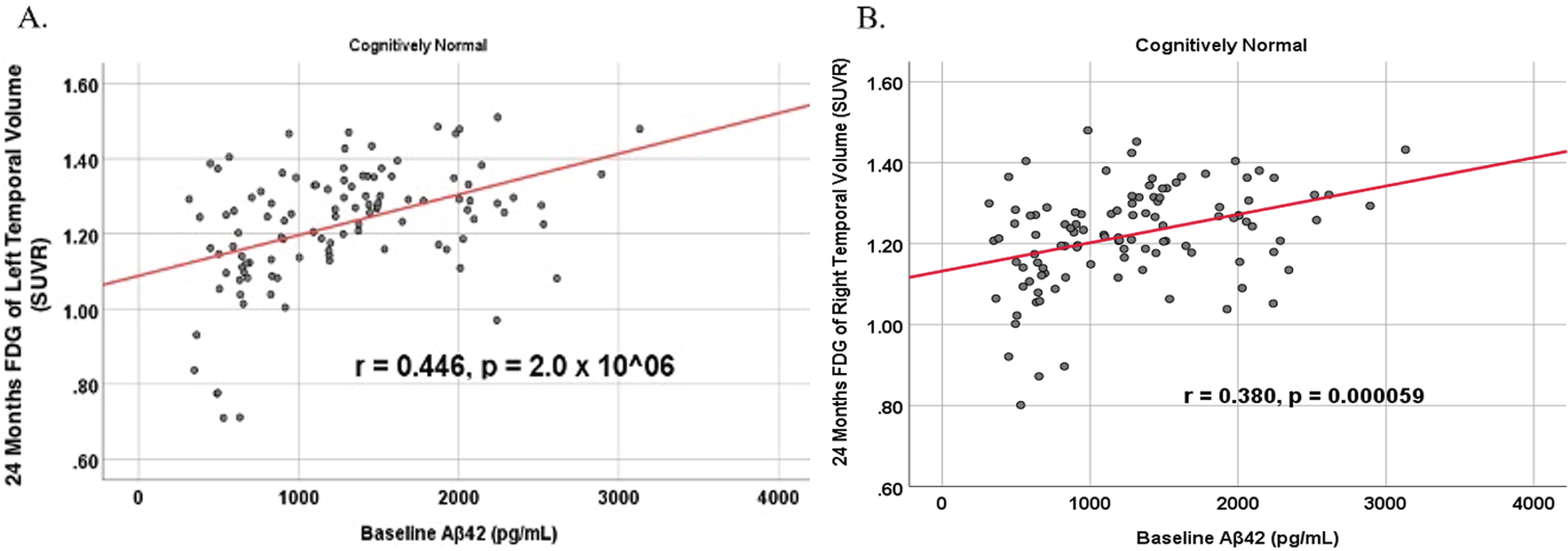
Partial correlations between baseline Aβ42 and 24 Month FDG brain volume regions in cognitively unimpaired control participants, corrected for age and education. Scatterplots above are corrected for age and education. A) Aβ42 versus Left Temporal (
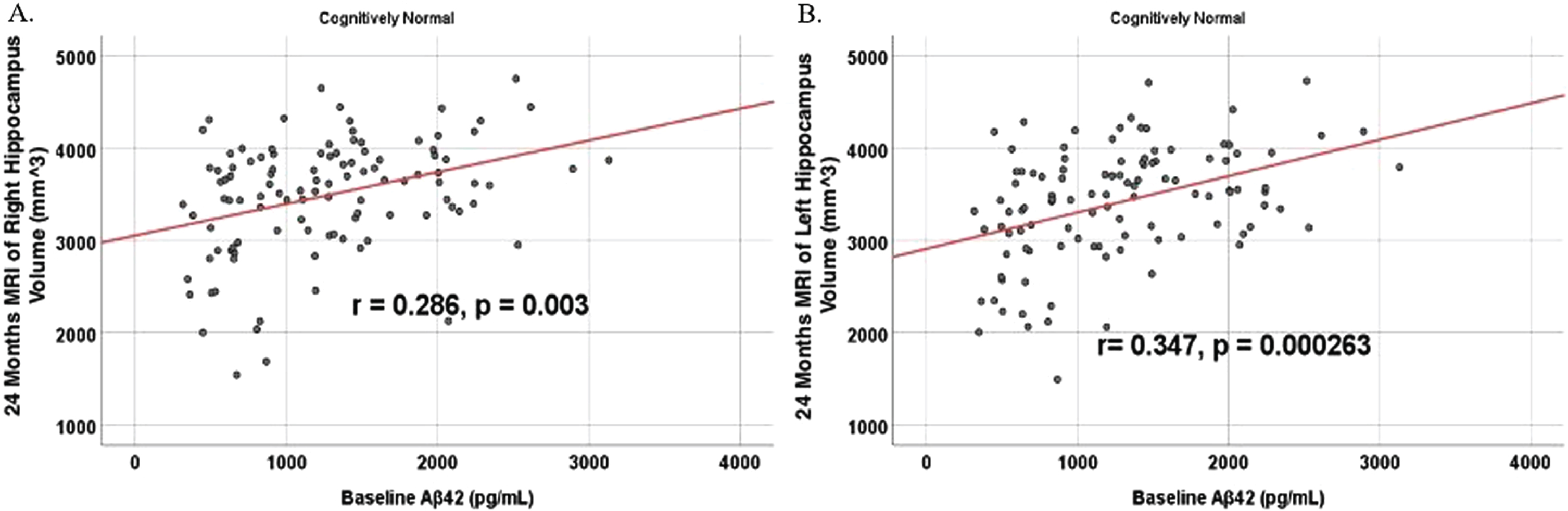
Partial correlations between baseline Aβ42 and 24 Month MRI brain volume regions in cognitively unimpaired control participants, corrected for age and education. Scatterplots above are corrected for age and education. A) Aβ42 versus Right Hippocampus (
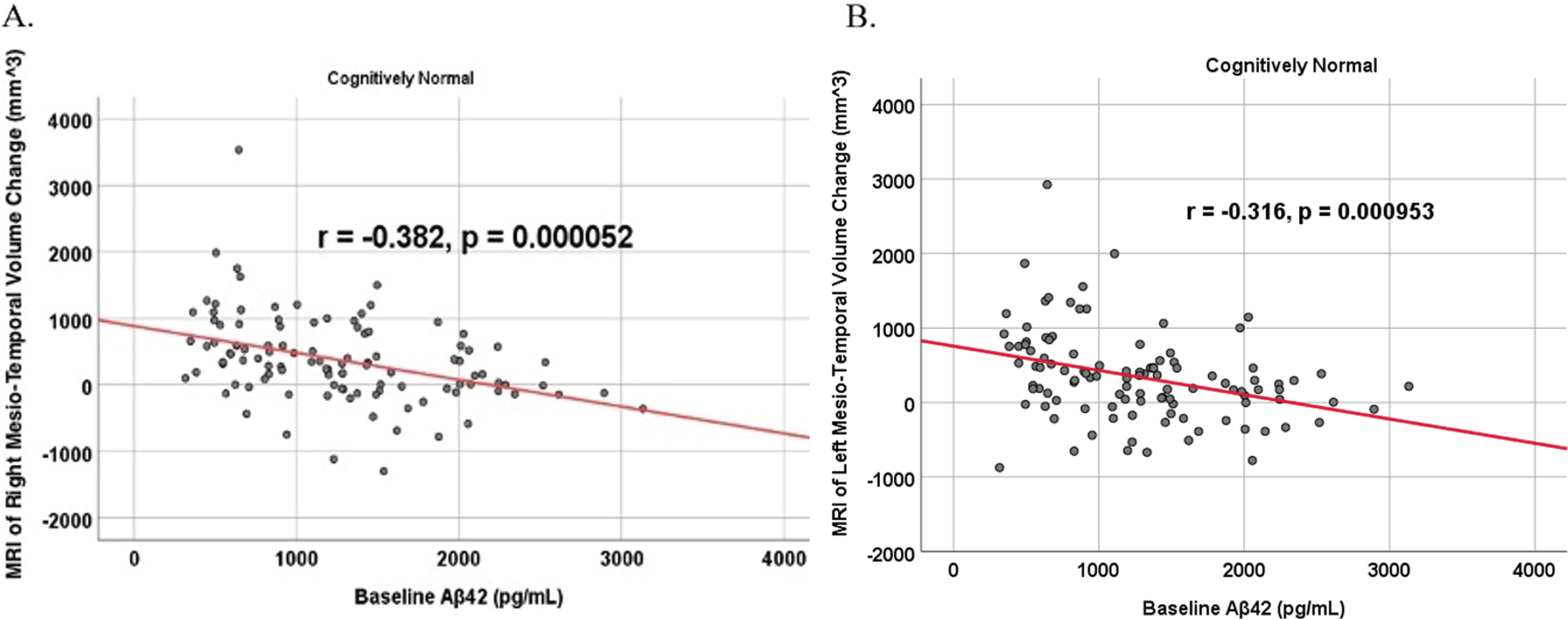
Partial correlations between baseline Aβ42 and MRI brain regional volume change in cognitively unimpaired control participants, corrected for age and education. Scatterplots above are corrected for age and education. A) Aβ42 versus Right Mesio-Temporal (
DISCUSSION
The main goal of this study was to evaluate a novel blood constituent, SDMA, against Aβ42 as a biomarker for AD dementia while reevaluating past inconsistent results of Aβ42 as a potential predictor of AD dementia progression, in addition to understanding the relationship between SDMA and Aβ42 with brain hypometabolism and atrophy as well as cognition and function. SDMA is an enantiomer of ADMA which is an inhibitor of nitric oxide synthase and a mediator of vascular endothelial function [36]. As a vascular risk factor, ADMA has been thought to contribute to the development of AD dementia; one study found ADMA plasma concentration level significantly increased and nitric oxide decreased in AD dementia patients [37]. Serum ADMA was increased in chronic kidney disease in parallel with increase of fecal tryptamine [38], which was shown to induce neurodegeneration in cells and animals [39, 40]. Additionally, in a prospective cohort study, serum ADMA concentration predicted worsening cognition in patients and was suggested to be a potential early detection marker for dementia and AD [41].
Based on our data analysis, we do not have sufficient evidence to conclude that either SDMA or Aβ42 as measured with 2D-UPLC tandem mass spectrometry is a reliable predictor of future cognitive status. For SDMA, our study is the first to our knowledge to look at SDMA as a single biomarker measurement for predicting diagnosis of prodromal AD dementia. Previous studies with SDMA included it with other renal biomarkers and have shown promising results discriminating between MCI and AD dementia participants [21, 42], but our study differs in its conclusion. Of note, one of the studies had a much smaller sample size and compared normal controls to MCI and AD dementia patients at the time of their illness; but did not examine baseline SDMA or Aβ42 to predict a future diagnosis [42]. Additionally, the other study found SDMA to be positively associated with AD dementia cognitive status and global AD brain pathology burden; however, SDMA was not analyzed via serum like this investigation but by concentrations in the brain from autopsy donors, and the study also had a smaller sample size [21]. The patients in that study were also significantly older at an average of 90 years compared to both groups in this study. To investigate the prognostic value of SDMA and Aβ42 further, we looked at the relationship between these biomarker levels and cognitive decline and neuroimaging outcome variable within prodromal AD dementia and cognitively unimpaired control subject cohorts. A previous study found higher SDMA, in contrast to our results, was associated with objective and subjective cognitive impairment [25]. However, the SDMA concentrations they found to be associated were much higher than the average SDMA concentration of either this investigation’s control group and prodromal AD dementia group. Nevertheless, we found no correlation between SDMA and MRI, FDG-PET, and neurocognitive scores in either the prodromal AD dementia group or the cognitively unimpaired controls.
While all the results were negative for SDMA, some results did point to an association between lower CSF levels of Aβ42 and cognitive and functional decline. For Aβ42, our results are in support of a review conducted of 26 previous studies that also found insufficient evidence in using plasma and CSF Aβ42 in the diagnosis or even risk assessment of AD dementia as a single measurement [43]. This review and other recent studies are in support of measuring Aβ42 and Aβ40 together for better prediction of amyloid pathology in prodromal AD dementia [7, 44]. In contrast, our results did find levels of CSF Aβ42, and not SDMA, positively correlate with temporal and hippocampal brain volume, brain metabolic activity, and neurocognitive scores in cognitively unimpaired controls but not in prodromal AD dementia participants, which is in line with previous research [45, 46]. In the prodromal AD dementia participants, the absence of a correlation between Aβ42 and the anatomical and functional changes that often typify AD may suggest that these pathologies are independent of one another, or alternatively, that CSF Aβ42 levels might behave differently across the lifespan [47]. For example, a study found higher and lower CSF Aβ42 in patients <75 years was indicative of future MCI/AD dementia, but over the age of 75 years, Aβ42 could not predict MCI/AD dementia [48]. In our investigation, correlations were smaller between Aβ42 and MMSE at 24 months and absent in prodromal AD dementia participants, suggesting a possible plateau effect, perhaps stemming from a more advanced age and/or disease-stage. The association of Aβ42 with markers of neurodegeneration (i.e., MRI and FGD-PET) might also be the result of a plateau effect that is dependent not only upon age but also the disease stage. Although not exactly the same, a similar effect has been reported in the literature in which Aβ42 plateaus as a patient progresses into MCI and then decrease in concentration as they advance into AD dementia [49]. In normal controls, however, there are varying reports on the behavior of Aβ42 with age, with some reports that Aβ42 increases with age consistently and another that it decreases in early life, increases in midlife, and then becomes unassociated with age at later years [50]. For both the normal controls and prodromal AD dementia patient groups, Aβ42 was inversely associated with age.
While Aβ42 may be a poor biomarker as a standalone measure, performance is improved when combined with Aβ40 as a ratio, as Aβ42/Aβ40 has proven to be a more precise and reproducible marker for amyloid-positivity [51]. For example, replacing Aβ42 for Aβ42/40 improved the concordance with amyloid PET by 15% in patients with MCI [52]. Apart from amyloid-positivity, studies have also found better diagnostic performance of AD dementia utilizing Aβ42/40 compared to Aβ42 alone [53, 54]. Altogether, based on prior reports pointing to the usefulness of Aβ42 as a standalone measure and its even more promising role as part of the Aβ42/40 index, our results suggest consideration of this ratio in future studies, as well as alternative explanations of our otherwise negative findings [3, 4]. For this reason, we explore the role of a multiplicity of potentially limiting and confounding factors.
Sample size
Another potential limitation to our study is the sample size. Although several studies of anti-amyloid therapies comprise smaller sample sizes [55], it is worth mentioning that positive findings have been published in studies with sample sizes larger than ours in regards to Aβ42 [53, 56–59]. However, this study is currently believed to be the largest in regards to assessing SDMA as a predictor of AD dementia, though there are very few select investigations of its utility in dementia. Future investigations of larger sample sizes are therefore encouraged in order to support the generalization of these results for both SDMA and Aβ42.
Comorbidities
Hepatic, renal, and neurodegenerative diseases can influence biomarker measurements in the older adult population. Biomarkers, such as phosphorylated tau (p-tau), neurofilament light chain, and total tau, can elevate by 2 to 4 folds in individuals with cirrhosis and show correlations with creatinine levels [60]. The liver makes albumin and filters proteins from blood, so cirrhosis may alter biomarker binding, especially sticky Aβ, and reduce protein elimination, whereas glomerular diseases of the kidneys may result in proteinuria that alters the level of blood protein [61]. Severe liver and kidney diseases, and peripheral neuropathy, such as diabetic neuropathy, can increase the concentration of neurodegenerative biomarkers [62, 63]. Our study has been able to rule out renal and hepatic diseases, with mean lab values within normal limits and no significant differences between the normal control and prodromal AD dementia cohorts. In the normal control cohort, the mean liver enzyme values are ALT 23.33, AST 26.18, and alkaline phosphatase 73.38. Renally, the mean creatinine is 0.88 and urea nitrogen is 19.82. In the prodromal AD dementia cohort, the mean liver enzyme values are ALT 22.63, AST 24.97, and alkaline phosphatase 68.97. Renally, the mean creatinine is 0.85 and urea nitrogen is 18.56. The prevalence of diabetes in the United States in 2019 is 11.3% of the population (37.3 million individuals), and among seniors, 29.2% of those aged 65 and older have diabetes (15.9 million individuals) [64]. One study has demonstrated that in patients with AD dementia, the prevalence of diabetes is higher (35% compared to 18% in control participants) and glucose tolerance is more impaired (46% compared to 24% in control participants) [65]. Although studies often exclude comorbid conditions, investigation of these factors can be valuable in understanding the clinical interpretation of biomarkers.
Race and ethnicity
Our study uses ADNI data, which includes around 10% of African American and Hispanic participants, specifically our data includes 91.67% White participants in the cognitively unimpaired cohort, and 91.78% White participants in the prodromal AD dementia cohort which is a limitation of the data set. Although one ADNI study that controlled covariates did not find differences between racial groups, ADNI itself is not population-based and unable to represent the general population [66]. Future studies should examine a larger and more diverse cohort of participants in a longitudinal study. It is important to ensure that studies are representative of the population in terms of racial and ethnic diversity, as well as social factors that are associated with dementia and pathology outcomes.
Age
Another explanation for why neither biomarker showed a significant difference between cognitively unimpaired control and prodromal AD dementia is the confounding variable age. Age has been documented to be the main predictor of CSF and plasma Aβ42 before, with mixed results in other studies for SDMA [67–69]. Although we adjusted for this variable in our multivariable analysis, we believe further prospective investigations are needed into these biomarkers that control for age using restriction or matching as part of the initial study design. An important limitation in our analysis is that we only collected baseline Aβ42 and SDMA levels. We found that these early baseline values were not significantly capable of predicting AD dementia from cognitively unimpaired controls, though we did not perform longitudinal analyses of constituent changes in relation to changes in brain volume, metabolic activity, and cognitive decline. Additionally, our study’s time frame of 24 months may be too narrow of a window for indicators to develop. Individuals with MCI have a 10 to 15% incidence of developing dementia each year [64]. One-third of individuals with MCI develop dementia due to AD within five years [65]. A timeframe of 24 months may not have been robust enough to see significant distinctions in imaging and biomarkers between cognitively unimpaired controls and prodromal AD dementia participants.
Further studies should be conducted that collect Aβ42 and SDMA levels over time to determine if there is a critical window during which these biomarkers have predictive capability of AD pathogenesis and progression. This approach has been fruitful in the past, with at least one prospective cohort study showing that repeated testing of serum Aβ42 may be useful in the detection of participants at risk for cognitive decline [70].
Socioeconomic
Social determinants of health (SDOH) are factors that can alter an individual’s health and wellbeing, including chronic disease management and comorbidities related to dementia. Important SDOH factors should be examined are financial and housing stability, social support, discrimination, food deserts, education, access to care, and quality of care. Our study was limited by our methods and did not examine most of these relevant SDOH factors. We did not find any studies that examined the relationship between SDOH factors and fluid AD dementia biomarkers. One study noted that neurodegenerative diseases may correlate with homelessness in individuals with challenging socioeconomic and unsupportive environments [71]. ADNI is a multi-site study and enrolls mostly college-educated participants with over 16 years of education. Previous studies have shown an association between higher education and reduced cognitive decline [72]. High education levels can inflate MMSE scores, possibly due to structural and cognitive process differences in individuals with more education. Additionally, ADNI limits study locations by including less diverse zip codes, especially areas with greater disparities and disease burdens. Further, individuals with little or no practical access to care and lack of trust in healthcare providers might go to less preventative and follow-up visits. Many chronic health conditions, including dementia and other comorbidities, may not be diagnosed and treated early on, thereby worsening disease outcomes. Future studies should examine the impact of SDOH factors on AD dementia biomarkers through prospective studies and interviews.
Conclusion
Our results are in support of other investigations concluding that there is insufficient evidence in utilizing baseline CSF Aβ42 in the diagnosis and risk assessment of AD dementia as a single measurement. Nevertheless, our results by no means preclude its use as part of a Aβ42/40 ratio, a use strongly supported by the current literature [3, 51]. We were also unable to find conclusive evidence to support the usage of SDMA as a novel biomarker capable of predicting AD pathogenesis. However, our results suggest that Aβ42 may be associated with neurodegeneration in the earliest phases of the disease when a ceiling or plateau effect of the biomarker appears less likely. Future larger studies should be conducted that collect Aβ42 and/or Aβ42/40 as well as SDMA concentration levels over time to determine if there is a critical window during which these biomarkers may have predictive capability of AD pathogenesis and progression.
Footnotes
ACKNOWLEDGMENTS
The authors would like thank all subject participants who voluntarily contribute themselves to the advancement of medical science.
Data collection and sharing for this project was funded by the Alzheimer’s Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12-2-0012). ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: AbbVie, Alzheimer’s Association; Alzheimer’s Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb Company; CereSpir, Inc.; Cogstate; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd and its affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd.; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics. The Canadian Institutes of Health Research is providing funds to support ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health (![]() ). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
Data collection and sharing for this project was funded by the Alzheimer’s Disease Metabolomics Consortium (National Institute on Aging R01AG046171, RF1AG051550 and 3U01AG024904-09S4).]
FUNDING
The authors have no funding to report.
CONFLICT OF INTEREST
The authors have no conflicts of interest to report.
DATA AVAILABILITY
The data supporting the findings of this study are available on request from the corresponding author.
