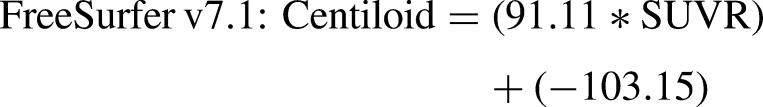Abstract
Background
Plasma biomarkers for Alzheimer's disease (AD) studies, but much remains unknown about the associations of plasma and PET biomarkers of amyloid-β (Aβ).
Objective
To determine the associations of plasma Aβ with PET Aβ accumulation and progression from PET A- to A + .
Methods
We evaluated PET A- participants with baseline plasma Aβ measurements and longitudinal indices of PET Aβ. Linear mixed effects models characterized the association of plasma biomarker outcomes with changes in PET Aβ values. Survival analysis evaluated the ability of baseline plasma biomarker A+/- status to differentiate trajectories of progression from PET A- to A + .
Results
Linear mixed effects models showed significant interactions between time and plasma Aβ42/40 with respect to longitudinal measures of PET Aβ burden. Survival analysis found that A status determined from plasma Aβ42/40 predicted distinct patterns of progression from PET A- to A + .
Conclusions
These models suggest that, in PET A- participants, baseline plasma Aβ42/40 can be used to predict both how much Aβ will accumulate over time and likelihood of becoming PET A + .
Introduction
Novel disease-modifying therapies for the treatment of Alzheimer's disease (AD) are focused on reduction of amyloid-β (Aβ),1–4 based on the premise of the Aβ cascade hypothesis. 5 While positron emission tomography (PET) and brain autopsy analyses indicate that the FDA-approved anti-Aβ therapies successfully remove Aβ from the brain, they have had limited success in slowing cognitive decline. 6 However, recent work has suggested that this may be due to the relative advanced disease stage in the participants included in the trials, and that initiating the removal of Aβ in earlier disease phases may be the key to delaying or reducing the risk of clinical AD progression. 7 If this is demonstrated conclusively, identification of individuals in the earliest stages of Aβ accumulation will be paramount in treatment success. While the utilization of cerebrospinal fluid (CSF) and PET imaging are used to qualify patients for Aβ immunotherapies, they are invasive, costly, and have limited accessibility.8,9 Plasma biomarkers offers a low-cost, accessible, and less invasive alternative to PET and CSF, have already shown promise as screening and diagnostic tools,10–12 and are also useful in disease staging in accordance with recent NIA-AA recommendations. 13
Two plasma biomarkers have emerged as leading candidates for indicating early Aβ deposition: the Aβ42/40 ratio and p-tau217. 14 Aβ42/40, a direct measure of the levels of Aβ40 and Aβ42 peptides in blood, has been shown to reflect both PET A status15–18 and Aβ PET SUVR values. 16 Similarly, p-tau217, a measure of phosphorylation of plasma tau species at the threonine 217 residue, has also been shown to predict PET A + status10,19,20 and Aβ PET SUVR values.21,22 While these findings support further investigation of Aβ42/40 and p-tau217 as plasma biomarkers of cerebral Aβ pathology, it should be noted that studies of their putative utility as such are based largely on cross-sectional studies that evaluated participants across a range of PET Aβ levels and clinical diagnoses. Few studies have focused on the utility of these plasma biomarkers for predicting incident Aβ in A- participants assessed longitudinally with PET, and those studies involved binarization of the plasma biomarkers as purely positive or negative as opposed to evaluating them as continuous measures. 23
In the present study, we conducted a comprehensive cross-sectional evaluation in PET A- participants that investigated the ability of the two most prominent plasma biomarkers of Aβ, Aβ42/40 and p-tau217, to predict longitudinal incident Aβ and provide quantitative estimation of Aβ accumulation.
Methods
Human participants
This study was approved by the University of Pittsburgh Institutional Review Board. Informed written consent was obtained from all participants. Data for the study cohort consisted of subjects recruited through the Alzheimer Disease Research Center and other population-based studies at the University of Pittsburgh. A total of 147 non-demented participants were included based on the availability of serial Aβ PET assessments and the availability of plasma Aβ40 and Aβ42 measures.
Included participants were diagnosed as non-demented by a multi-disciplinary panel, blinded to all imaging and blood results, and presented with a Clinical Dementia Rating (CDR) score of ≤0.5. Additionally, all participants demonstrated PET A- status at baseline based on a Centiloid value of 20. to best represent the presence of neuritic plaques. 24 A subset of 103 participants from the full cohort additionally included plasma p-tau217 data. All participants had one follow-up scan, resulting in two longitudinal Aβ PET assessments.
Image acquisition and analysis
All participants underwent 11[C]Pittsburgh compound-B (PiB) PET with and magnetic resonance (MR) imaging. PiB was produced under an approved United States Food and Drug Administration (FDA) Investigational New Drug (IND) application (IND#72200). PiB PET images were collected 50 to 70 min post-injection as previously described.
25
Sagittal T1-weighted magnetization-prepared rapid acquisition with gradient echo (MPRAGE) MR images were acquired using a 3.0 T Siemens Prisma scanner (Siemens Healthcare). PiB PET images were motion-corrected as needed and summed over 50–70 min before being registered to their corresponding MR images. MR images were processed with FreeSurfer v7.1.1 to construct a brain parcellation atlas that was then applied to the PiB PET images26,27 sample nine regions (anterior cingulate, anterior ventral striatum, superior frontal, orbitofrontal, insula, lateral temporal, parietal, posterior cingulate, precuneus) to obtain a volume-weighted global PiB SUVR value, relative to cerebellar gray matter.
25
SUVRs were transformed into Centiloids based on the following transformation equation, derived based on methods described elsewhere.
28
Plasma biomarker acquisition and analysis
Participants’ blood samples were collected via venipuncture by experienced nurses using K2EDTA blood tubes (BD #366643) and centrifuged within 30 min of blood collection for 10 min at 2000 xg at 4°C. Plasma samples were aliquoted into LoBind microtubes and frozen at −80°C until use. Samples were limited to a maximum of three cycles to minimize the impact of freeze-thaw cycles, in accordance with published literature. 29 Plasma concentrations of Aβ40, Aβ42, and p-tau217 30 were measured using Single molecule array (Simoa) methods on a Quanterix HD-X as previously described. 31 Plasma samples were thawed, vortexed to mix, and centrifuged for 10 min at 4000 x g at 4°C to remove particulates. Plasma Aβ40 and Aβ42 were measured using the Neurology 4-Plex E (N4PE; #103670) kit and plasma p-tau217 was measured using the Janssen p-tau217 + assay, developed at Janssen Research & Development using Janssen's proprietary antibodies which measures p-tau217 as well as p-tau212 and p-tau214. Quality control (QC) samples were analyzed at the start and the end of each run to assess the reproducibility of each assay. The N4PE assay had within- and between-plate CVs of <10% and <16%, respectively. The Janssen p-tau217 + assay had within- and between-plate CVs of 4.0 and 7.3%, respectively. These CVs were within accepted ranges. 32
Statistical analysis
All statistical analyses were performed using R (version 4.2.1, R Foundation for Statistical Computing, Vienna, Austria), 33 using participant APOE*4 status, sex, age, and years of education as covariates. Plasma Aβ was expressed as the Aβ42/40 ratio while p-tau217 was natural log transformed to account for skewness in the data in accordance with previous studies.31,34,35 Associations between plasma biomarker measures and longitudinal change in PET Aβ were evaluated for the full cohorts, as well as for plasma A+/- subsets, using linear mixed effect models that included the baseline Centiloid value and an interaction term between PET follow-up interval and baseline plasma biomarker measures. The full cohorts were evaluated with the addition of age, sex, APOE*4 status, and years of education as covariates. Alternate versions of the full cohort model were run including race as a covariate given the racial diversity of the cohort; including cognitive status (CU versus MCI) to ensure that MCI participants were not driving the results; and with a spline fit for Aβ42/40 to allow for non-linear effects of baseline Aβ42/40. Models were duplicated with the variables standardized as validation. Associations between plasma measures and longitudinal progression from PET A- to A + were evaluated using Kaplan-Meier survival curves and log rank tests, using time between baseline scan and first PET A + scan as the outcome. Cox proportional hazard models were also used to assess participants’ continuous baseline plasma against risk of progression from PET A- to A + . Data included in the survival analyses was right-censored to a time of 8 years. These models additionally included baseline Centiloids as a covariate. Subsets were split into plasma A + and plasma A- for each of the plasma biomarkers, Aβ42/40 and p-tau217, independently, based on the following values: Aβ42/40, 0.095; p-tau217, 0.084. These cutoffs were derived in accordance with the BETTH methods described elsewhere 36 to maximize effect size. For both Aβ42/40 and p-tau217, the optimal method was GMM.
Results
Participant characteristics, including age, sex, cognitive status, APOE*4 status, and race, for the full cohort and subset cohort are shown in Table 1. All participants were diagnosed by a multi-disciplinary panel, blind to all imaging and blood results, as being non-demented (CDR 0.07 ± 0.18).
Cohort characteristics.
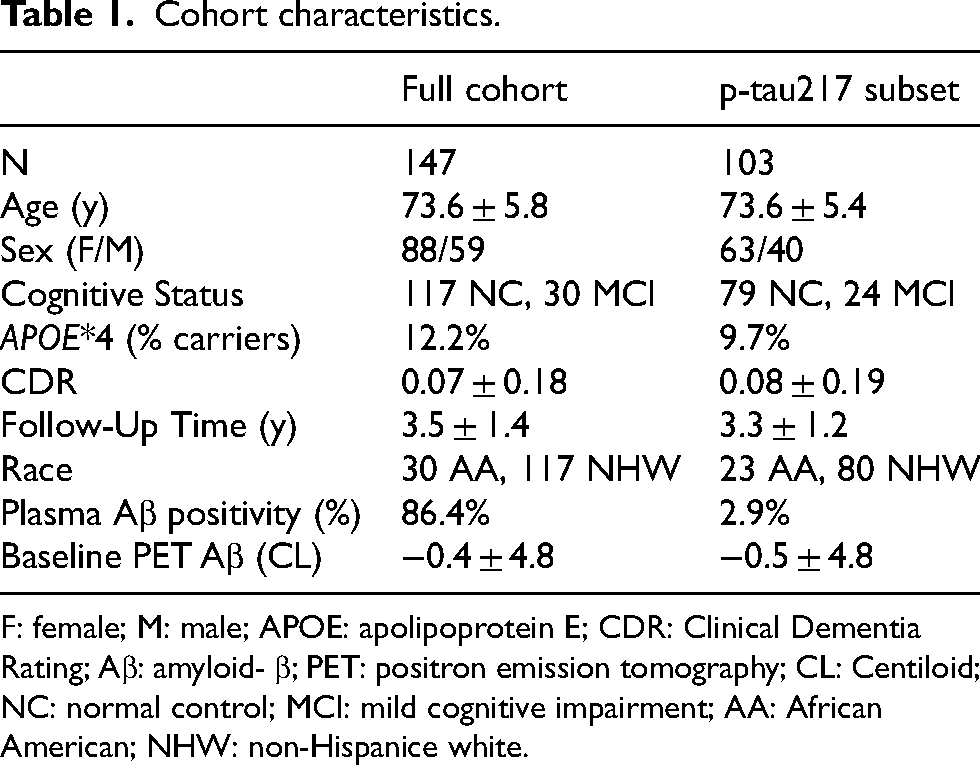
F: female; M: male; APOE: apolipoprotein E; CDR: Clinical Dementia Rating; Aβ: amyloid- β; PET: positron emission tomography; CL: Centiloid; NC: normal control; MCI: mild cognitive impairment; AA: African American; NHW: non-Hispanice white.
Prediction of change in Centiloids
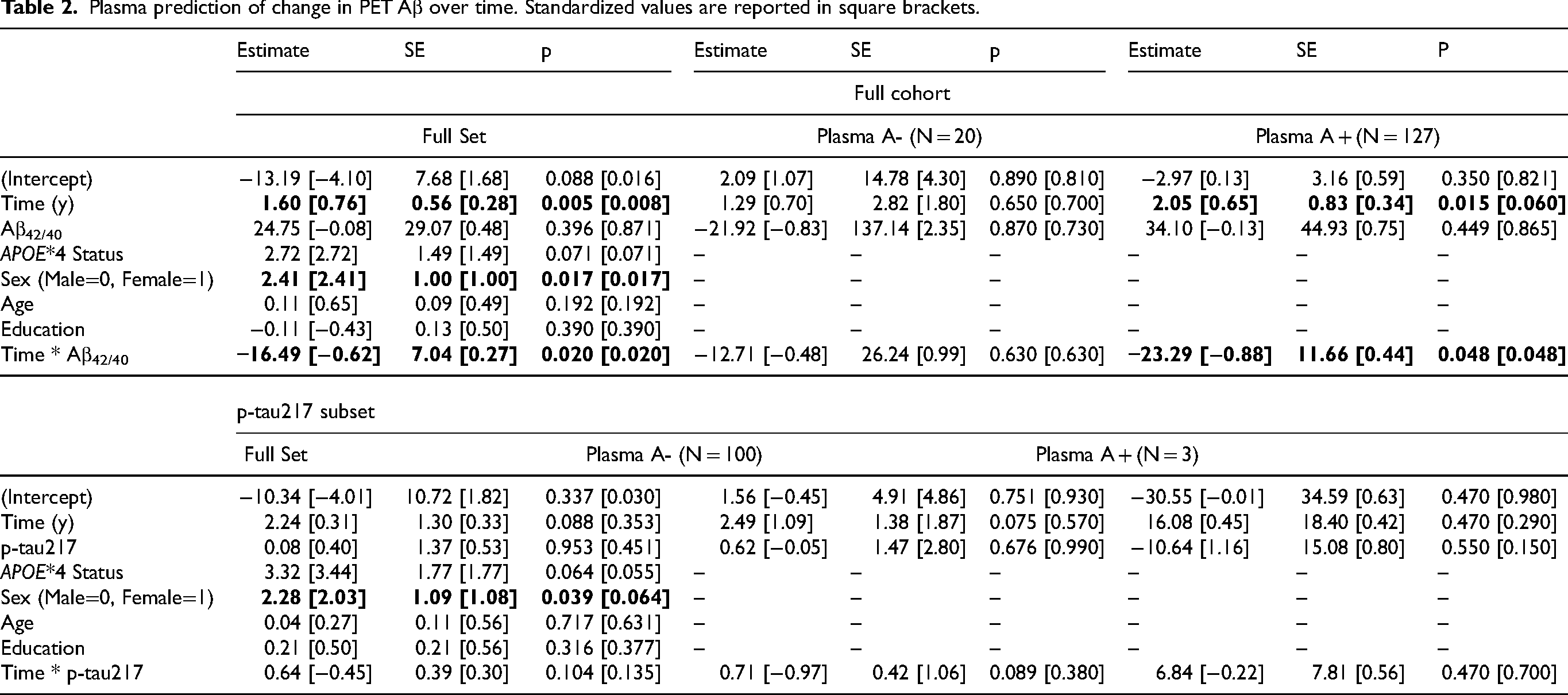
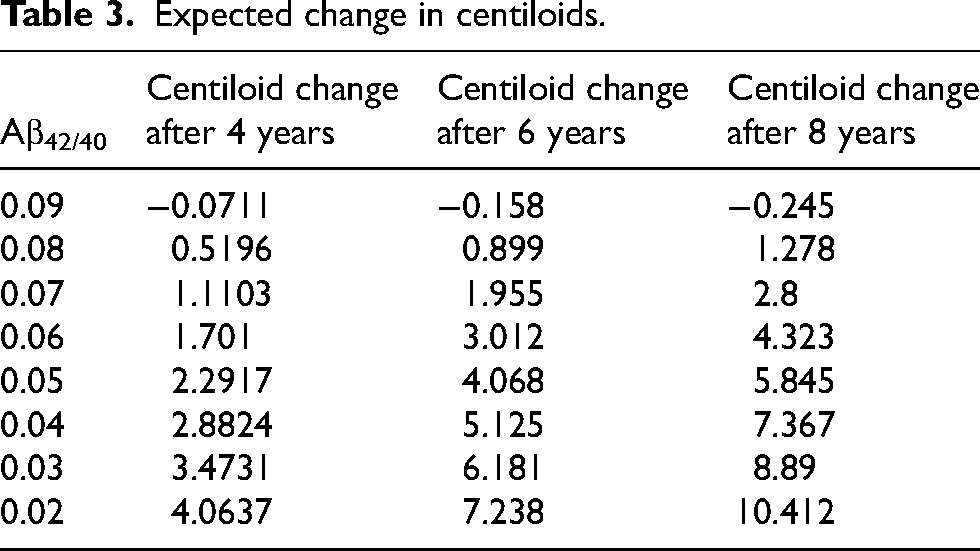
In the linear mixed effects models with respect to change in PET Aβ (Table 2), time and the interaction between time and baseline plasma Aβ42/40 were significant (p = 0.020) such that decreasing levels of Aβ42/40 over time was significantly associated with increased PET Aβ. The same was found for the plasma A + models (p = 0.048), but not for the plasma A- models (p = 0.630). Time was independently significant in the full (p = 0.005) and A + (p = 0.015) models, but not the A- model (p = 0.650), with increased time associated with increased PET Aβ. Sex was additionally independently significant (p = 0.017), with female participants displaying greater change in Aβ than male participants. In the p-tau217 subset, only sex was significant in the full cohort (p = 0.039), again with female participants displaying greater change in Aβ. Neither race nor cognitive state was significant in the models. The consideration of non-linear effects of baseline Aβ42/40 did not change any of the significances found in the linear model (Supplemental Table 1). Figure 1 shows the baseline trajectories, with standard errors, for the Aβ42/40 model for plasma A + and plasma A- participants. Examples of expected change in Centiloids for varying baseline levels of Aβ42/40 after 4, 6, and 8 years, based on the full models, are shown in Table 3.
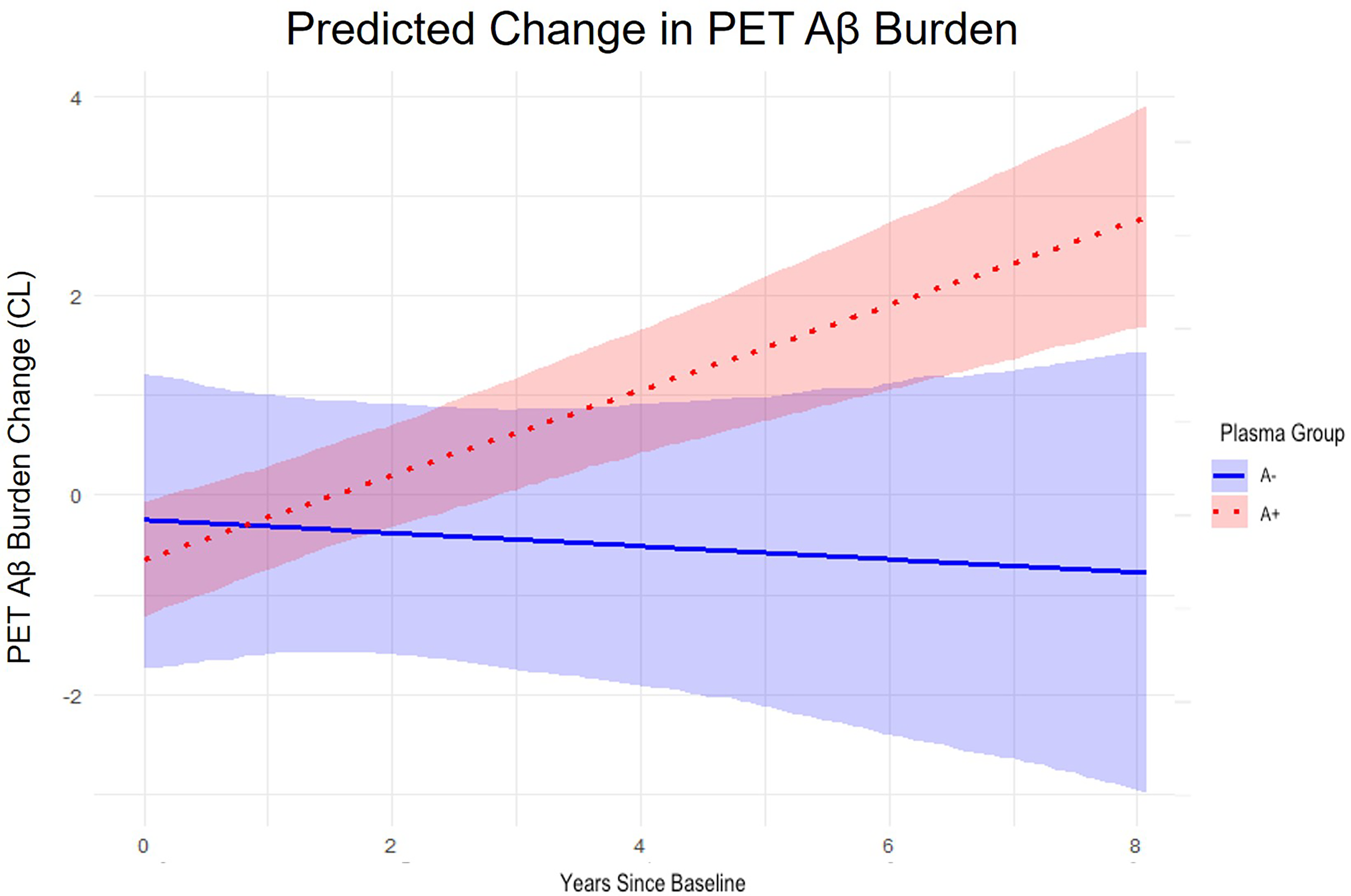
Plasma A+/- Centiloid trajectories over time.
Plasma prediction of change in PET Aβ over time. Standardized values are reported in square brackets.
Expected change in centiloids.
Progression from PET A- to A+
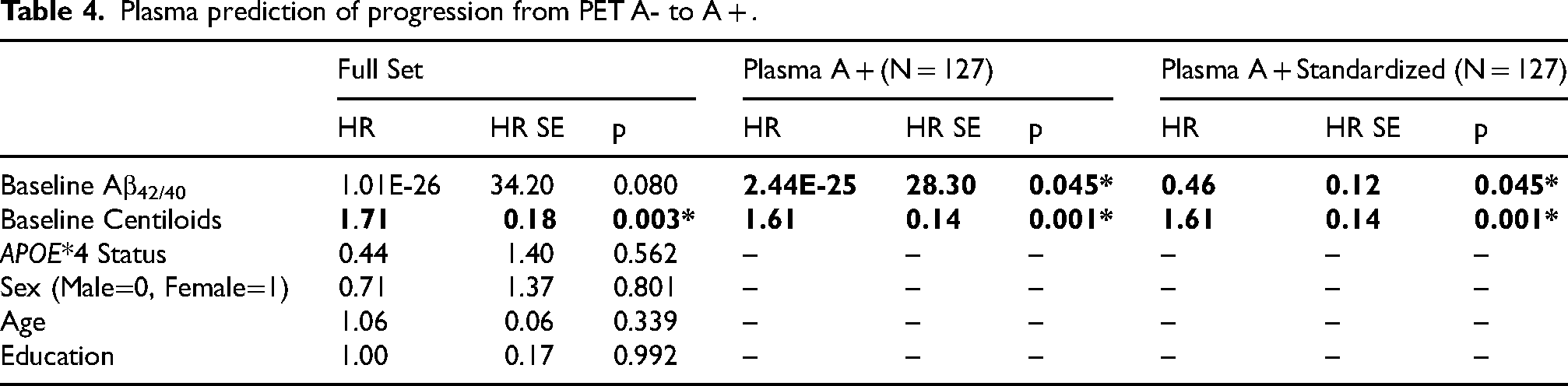
The Kaplan-Meier curves (Figure 2) visually differentiated the plasma A- and A + participants for Aβ42/40, with plasma A + participants starting to progress to PET A + after 2 years. Based on the log rank tests, the plasma A + group had significantly more progression from PET A- to A + than the plasma A- group (p < 0.05). In the Cox proportional hazard models testing the full cohort's baseline Aβ42/40 with respect to progression from PET A- to A + (Table 4), higher baseline Centiloids were significantly associated with progression from PET A- to A + (p < 0.05), with a hazard ratio (HR) of 1.71, while baseline Aβ42/40 had a trend-level significance with progression from PET A- to A + (p = 0.080), with an HR of 1.01E-26. In the plasma A + cohort, both baseline Aβ42/40 and Centiloids were significantly associated with progression from PET A- to A + (p < 0.05), with HRs of 2.44E-25 and 1.61 respectively. To ease interpretability, the Aβ42/40 measure was standardized, relative to a mean value of 0.0692 and standard deviation of 0.0137, in a secondary plasma A + analysis. In this model, the HR for Aβ42/40 was 0.46. The plasma A- cohort is excluded as the models were unable to generate results from the limited number of participants.
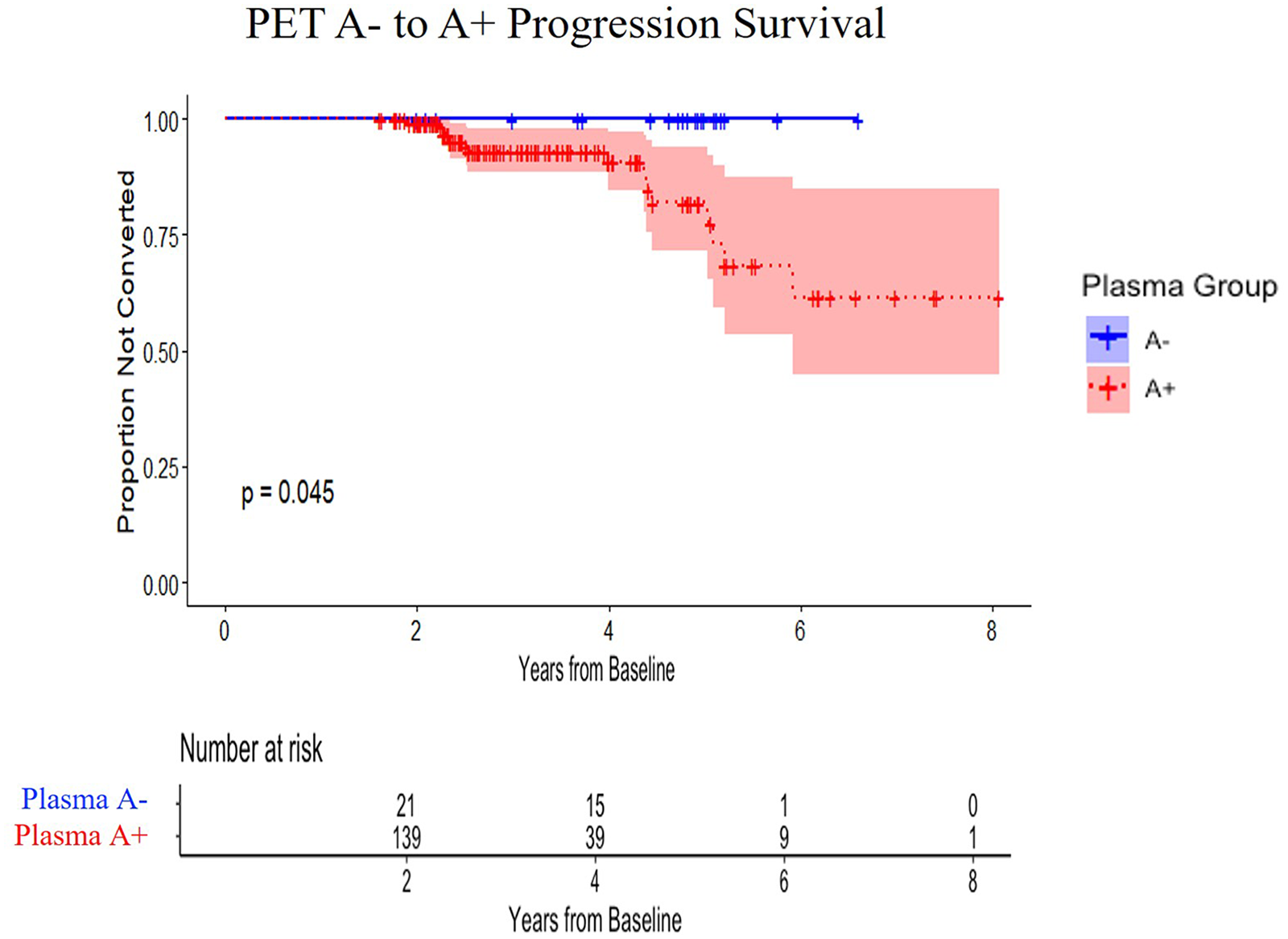
Survival curves for risk of progression from PET A- to A +.
Plasma prediction of progression from PET A- to A + .
Discussion
As the need for early detection of AD pathology in the brain becomes increasingly necessary in the search for a greater anti-Aβ immunotherapy efficacy, inexpensive, non-invasive, and widely accessible biomarkers that can reliably predict incident cerebral Aβ pathology is crucial for identifying at-risk individuals likely to benefit the most from anti-Aβ interventions. Plasma biomarkers that are well positioned to meet this need.
To determine the potential utility of plasma Aβ42/40 and p-tau217 for the early prediction PET Aβ accumulation and progression to PET A+, we evaluated baseline measures of plasma Aβ42/40 and p-tau217 against longitudinal change in PET Aβ burden and risk of progression from PET A- to A +. We found that the interaction of Aβ42/40 with time significantly predicts the magnitude of PET Aβ longitudinal change, particularly in plasma A + participants, and that baseline Aβ42/40 status has significant associations with future progression from PET A- to A + in plasma A + participants. P-tau217 did not show any significant associations with changes in Aβ load or progression to A + .
Models of the interaction between time and Aβ42/40 level with respect to PET Aβ found that both time and the interaction term were significant. The beta values in the models suggest that while PET Aβ is expected to increase with time, lower baseline levels of Aβ42/40 suggest accelerated rates of Aβ deposition. Table 3 demonstrates this. A plasma Aβ42/40 level of 0.06 predicts an increase of 4.3 Centiloids in 8 years. If baseline plasma Aβ42/40 level is 0.04, that same Centiloid increase is expected in just over 6 years, and a plasma Aβ42/40 level of 0.02 projects that change in approximately 4 years. Furthermore, this was significant only for plasma Aβ42/40 A + participants, not A-. This indicates that, once an individual becomes plasma A+, we can use their continuous plasma Aβ42/40 level to project when they will accumulate enough Aβ deposits in the brain to reach a level of abnormality on PET Aβ.
In addition to its prediction of future brain Aβ accumulation, Aβ42/40 status has significant associations with progression from PET A- to A + (Table 4). Plasma Aβ42/40 status differentiates distinct patterns of progression risk, with none of the plasma A- participants in the cohort progressing to PET A + during the observation period, while conversion to PET A + among plasma A + participants was observed after as little as 2 years. Furthermore, in the Cox proportional hazard models, plasma A + participants’ plasma Aβ42/40 continuous values had a significant association with risk of progression to PET A + . Specifically, after standardizing the Aβ42/40 measure, we found that the Aβ42/40 hazard ratio was 0.46, indicating that every 0.0137 decrease in Aβ42/40 below 0.0692 was associated with a 217% risk of progression to PET A + . This follows the results found in the assessment of continuous plasma Aβ42/40 and time with change in PET Aβ.
P-tau217 did not have any significant associations in the models evaluating plasma and time with respect to Centiloid changes. This is likely due to plasma p-tau217's close correlation with abnormal levels of insoluble Aβ in the brain.14,37,38 This is also the probable reason that the p-tau217 data could not be assessed in the survival models because less than 3% of the PET A- participants were p-tau217 A + . These results suggest that, while p-tau217 has been shown to be very robust, even better than plasma Aβ42/40, for detecting PET A + individuals,14,37,38 it may not be suitable for predicting future brain Aβ accumulation in PET A- individuals.
This study is not without its limitations. While the PET measures in the study were longitudinal, the plasma measures were cross-sectional. Future work should include longitudinal plasma measures to additionally factor in change in plasma biomarker levels over time. Many of the analyses relied on the application of binary thresholds, which are dependent on both the dataset they are derived from and their method of determination. Future work should evaluate the effect of applying differing or multiple, more granular thresholds. The models considered in this work were linear. Future work should consider whether non-linear models provide a better fit for the data. APOE*4 carrier status was included as a covariate in the models, but only 12.2% of participants in this cohort were carriers. This is substantially lower than the estimated 25%. 39 This may reduce the generalizability of the findings. Years of education was included as a covariate, but previous work has suggested that other social determinants of health, particularly racialization, needs to be given full consideration in plasma and imaging biomarker assessment. 40 While this analysis did not find any significant association with racialization, likely due to the small size of the cohort, future work should to consider including covariates such as socioeconomic status, type of employment, food security, neighborhood disadvantage, access to healthcare, social engagement, and stress to further improve generalizability. 41
Conclusions
Baseline plasma Aβ42/40 levels significantly interacted with time in association with change in PET Aβ and plasma Aβ42/40 status significantly predicted patterns of progression from PET A- to A + . P-tau217 did not show any significant associations. These findings suggest that plasma Aβ42/40 may be useful in selecting individuals for anti-Aβ treatments who are accumulating Aβ at subthreshold PET levels and are at risk of progressing from PET A- to A+, allowing a closer follow-up potentially leading to earlier therapeutic interventions for Aβ removal to yield the most meaningful clinical response. Future work in larger and diverse cohorts should validate these results and evaluate how the inclusion of longitudinal plasma measurements may improve the predictions as well as consider how best to standardize plasma Aβ measurements to allow for comparability across assays.
Supplemental Material
sj-docx-1-alz-10.1177_13872877261422406 - Supplemental material for Plasma amyloid-β predicts amyloid-β accumulation in PET A- non-demented participants
Supplemental material, sj-docx-1-alz-10.1177_13872877261422406 for Plasma amyloid-β predicts amyloid-β accumulation in PET A- non-demented participants by Alexandra Gogola, Ann D. Cohen, Brian J. Lopresti, Milos D. Ikonomovic, Beth Snitz, Dana Tudorascu, Alexandria Reese, Xuemei Zeng, Davneet Minhas, Julia Kofler, Cristy Matan, Howard Aizenstein, Oscar Lopez, Thomas K. Karikari and Victor L. Villemagne in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
The authors have no acknowledgments to report.
Ethical considerations
This study was approved by the University of Pittsburgh Institutional Review Board.
Consent to participate
Informed written consent was obtained from all participants.
Consent for publication
Not applicable
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institute of Aging grants P50 AG005133, RF1 AG025516, R01 AG052446, R01 AG052521, P01 AG025204, R01 AG063752. TKK and the Karikari Laboratory were supported by NIH/NIA (R01 AG083874, U24AG082930, P30 AG066468, RF1 AG077474, R01 AG083156, R37 AG023651, R01 AG025516, R01 AG073267, R01 AG075336, R01 AG072641, P01 AG025204), NIH/NINDS (U01 NS131740, U01 NS141777), NIH/NIMH (R01 MH108509), Aging Mind Foundation (DAF2255207), DoD (HT94252320064), the Anbridge Charitable Fund, and a professorial endowment from the Department of Psychiatry, University of Pittsburgh. The content of this article is solely the responsibility of the authors and does not necessarily represent the official views of the funders.
National Institute on Aging, (grant number P01 AG025204, P30 AG066468, P50 AG005133, R01 AG025516, R01 AG052446, R01 AG052521, R01 AG063752, R01 AG072641, R01 AG073267, R01 AG075336, R01 AG083156, R01 AG083874, R37 AG023651, RF1 AG025516, RF1 AG077474, U24AG082930).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TKK has consulted for Quanterix Corporation, SpearBio Inc., Neurogen Biomarking LLC., and Alzheon, has served on advisory boards for Siemens Healthineers and Neurogen Biomarking LLC., outside the submitted work. He has received in-kind research support from Janssen Research Laboratories, SpearBio Inc., and Alamar Biosciences, as well as meeting travel support from the Alzheimer's Association and Neurogen Biomarking LLC., outside the submitted work. TKK has received royalties from Bioventix for the transfer of specific antibodies and assays to third party organizations. He has received honoraria for speaker/grant review engagements from the NIH, UPENN, UW-Madison, the Cherry Blossom symposium, the HABS-HD/ADNI4 Health Enhancement Scientific Program, Advent Health Translational Research Institute, Brain Health conference, Barcelona-Pittsburgh conference, the International Neuropsychological Society, the Icahn School of Medicine at Mount Sinai and the Quebec Center for Drug Discovery, Canada, all outside of the submitted work. TKK is an inventor on several patents and provisional patents regarding biofluid biomarker methods, targets and reagents/compositions, that may generate income for the institution and/or self should they be licensed and/or transferred to another organization. These include WO2020193500A1: Use of a ps396 assay to diagnose tauopathies; US 63/679,361: Methods to Evaluate Early-Stage Pre-Tangle TAU Aggregates and Treatment of Alzheimer's Disease Patients; US 63/672,952: Method for the Quantification of Plasma Amyloid-Beta Biomarkers in Alzheimer's Disease; US 63/693,956: Anti-tau Protein Antigen Binding Reagents; and 2450702-2: Detection of oligomeric tau and soluble tau aggregates.
Data availability statement
Data utilized in this work is available upon request and with the necessary data sharing agreements.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.