Abstract
Background:
Alzheimer’s disease (AD) is the most common form of dementia. AD is also the leading cause of morbidity and mortality due to dementia worldwide. It has been shown that AD is associated with type 2 diabetes mellitus (T2DM) and brain insulin resistance. Rs1801278 is a polymorphism in insulin receptor substrate-1 (IRS-1) gene which changes the amino acid Arg972. This polymorphism has been found to be associated with susceptibility to AD in some populations.
Objective:
In the present study, our aim was to investigate the association of Arg972 IRS-1 (rs1801278) gene polymorphism and late-onset Alzheimer’s disease (LOAD) in an Iranian population.
Methods:
In this case-control study, 150 patients with LOAD and 150 unrelated healthy controls were recruited. Polymerase chain reaction (PCR) was performed to amplify a DNA segment of 263 base-pair (bp) length containing the single nucleotide polymorphism (SNP). The PCR product was then incubated with MvaI restriction enzyme to undergo enzymatic cleavage. Electrophoresis was thereafter carried out using agarose gel and DNA safe stain. The gel was ultimately visualized under a UV trans-illuminator. Allelic and genotypic frequencies were then compared.
Results:
A allele (mutant) of the gene was significantly associated with the risk of AD after adjustment for sex and age (p = 0.04, adjusted OR:1.77, 95% CI:1.00–3.11). Only AA genotype (mutant homozygote) was significantly associated with the risk of AD after adjustment for sex and age (p = 0.01, adjusted OR:2.39, 95% CI:1.22–4.66).
Conclusion:
SNP rs1801278 is significantly associated with the risk of developing AD in the studied Iranian population.
INTRODUCTION
Alzheimer’s disease (AD) is the most common form of dementia and also the leading cause of morbidity and mortality due to dementia worldwide [1]. AD is characterized by neuronal cell death [2] which arises from extracellular amyloid-β (Aβ) plaques and intracellular tau neurofibrillary tangles (NFTs) [3]. The World Alzheimer Report indicated about 47 million AD or AD-related dementia throughout the world in 2015 [4]. It is anticipated that, as the world’s population is aging, the number of patients with dementia, will grow to 130 million worldwide in 2050 [1, 4]. AD is classified into two forms of familial early-onset AD and sporadic late-onset AD (LOAD), constituting 3% and 97% of the AD cases, respectively [5]. The progression of LOAD occurs normally after 65 years of age [6].
It is believed that AD is associated with some gene mutations. Mutations of Aβ precursor protein and presenilin 1 and 2 in familial AD or LOAD and mutations of ɛ4 allele of APOE (APOE ɛ4) gene in sporadic cases of AD have been reported [7, 8]. AD is also associated with polymorphisms of TREM2 [9], MS4A6A [10], and CD33 genes [11].
Epidemiological and experimental studies suggest that insulin resistance is a key risk factor for type 2 diabetes mellitus (T2DM), AD, and other AD-related dementias [12]. An epidemiological study showed that T2DM is associated with an increased risk of AD [3]. The incidence of dementia increases by 50–100% relative to people without diabetes [13]. In Iran, prevalence of impaired fasting glucose and total DM were 14.60% (95% CI: 12.41–16.78) and 11.37% (95% CI: 9.86–12.89) among individuals with 25–70 years of age, respectively, in 2011 [14]. The number of DM patients is estimated to rise to 9.24 million cases by 2030 in Iran [15]. In a cohort study carried out in Guilan province of Iran (2014–2017), prevalence of DM was 24.1% [16].
Recently, it has been shown that the central nervous system (CNS) insulin resistance can be present apart from peripheral insulin resistance of T2DM in individuals with LOAD [17–20].
Neuronal growth, development, and neurogenesis have important roles in insulin function in the CNS [18, 19]. The role of insulin in neuronal cells is mediated by two main signaling pathways, that is to say, insulin –insulin receptor substrate (IRS) –protein kinase B (AKT) pathway and mitogen-activated protein kinase pathway [20]. Insulin –IRS1 –PI3K (phosphoinositide 3-kinase) –AKT –TBC1D4 signaling induces cognition and emotional function in hippocampus. In addition, insulin –IRS1 –PI3K –AKT –GSK3β (glycogen synthase kinase 3) has a role in neuroplasticity and neurogenesis [20].
In animal models, brain insulin level alterations can influence neuronal glucose uptake and metabolism due to glucose transporter type 4 (GLUT4) translocation arising from insulin –IRS1 –AKT signaling pathway in the basal forebrain, hippocampus, amygdala, and to a lesser degree, in the cerebral cortex and cerebellum [21–24]. Under the condition of high-energy demand during spontaneous alternation tests of spatial working memory, this mechanism can promote cognitive activity in the hippocampus in rats [21–23].
Brain insulin resistance can result from insulin-like growth factor-1 (IGF-1) resistance and IRS-1, 2 malfunctions induced by Aβ accumulation [25]. There is a two-sided relationship between brain insulin resistance and Aβ plaque deposition in the brain [26].
Arg972 IRS-1 gene polymorphism (rs1801278) results in a Gly/Arg substitution at codon 972 (Arg972) that impairs IRS-1 ability to activate the downstream PI3K signaling pathway. Therefore, it can lead to insulin signaling malfunction [6]. Lack of insulin signaling is proposed to upregulate amyloidogenic and Tau NFT-genic pathways. There is a higher prevalence of this polymorphism in T2DM [27] and AD patients [28].
Albegali et al. [29] and Alharbi et al. [30] have found significant association of rs1801278 variant with T2DM (p < 0.001 and p = 0.04, respectively). Li et al. [31] in a meta-analysis, found significant association between rs1801278 and T2DM risk in recessive model (AA versus GA + GG, p = 0.043) and codominant model (AA versus GG, p = 0.007).
In contrast, some studies have shown no association of IRS-1 gene polymorphisms with AD in Caucasian populations [27, 32–35], but a Chinese study evaluating the association of rs1801278 polymorphism with AD in a Han Chinese population revealed a significant association of the polymorphism and AD [28].
An explanation for this discrepancy could be the possible effects of ethnic populations on the mentioned association [28].
As little is known about the association of Arg972 IRS-1 (rs1801278) with AD in the Iranian ethnic population, we set out the present study to test the hypothesis of the presence of the association between the polymorphism and AD in an Iranian population in the north of Iran.
MATERIALS AND METHODS
Subjects
Given the results of previous published research [28] and considering type 1 error (α) = 0.05, type 2 error (β) = 0.1 (power of study = 90%), and a case-control ratio of one-to-one, a minimum sample size of 148 participants in each group was calculated and considered as a prerequirement for this study. Therefore, 150 patients with LOAD and 150 unrelated healthy subjects were enrolled in Guilan province in the north of Iran. Inclusion criteria included patients with probable LOAD diagnosed according to the National Institute on Aging and Alzheimer’s Association (NIA-AA) diagnostic criteria of AD [36]. Exclusion criteria for the patients were the history of head trauma, stroke, motor neuron disease, congenital dementia, neurological infections, neuropsychologic systemic lupus erythematosus, sarcoidosis, multiple sclerosis, other neurodegenerative diseases, and familial history of AD in first-class relatives. Informed consent was obtained from all the participants. This study was approved by the ethics committee of Guilan University of Medical Sciences.
Genotyping
Five milliliters of peripheral blood sample were collected from the participants and stored in Tris-EDTA 5% buffer at –30°C. DNA was extracted by a salting-out method from white blood cells. Forward and reverse primers were synthesized and used for PCR amplification. Restriction enzyme MvaI was used for Restriction Fragment Length Polymorphism (RFLP) as previously described [37].
PCR amplification was performed for all samples using respective forward and reverse primers. In the next step, PCR products were incubated and digested by MvaI. PCR-RFLP characteristics are shown in Table 1.
Enzymatic digestion reaction characteristics
Electrophoresis was carried out using 3% agarose gel and visualized by DNA-safe stain using a UV trans-illuminator (Figs. 1 2). A few samples were sequenced to illustrate wild-type and variant allele sequences.
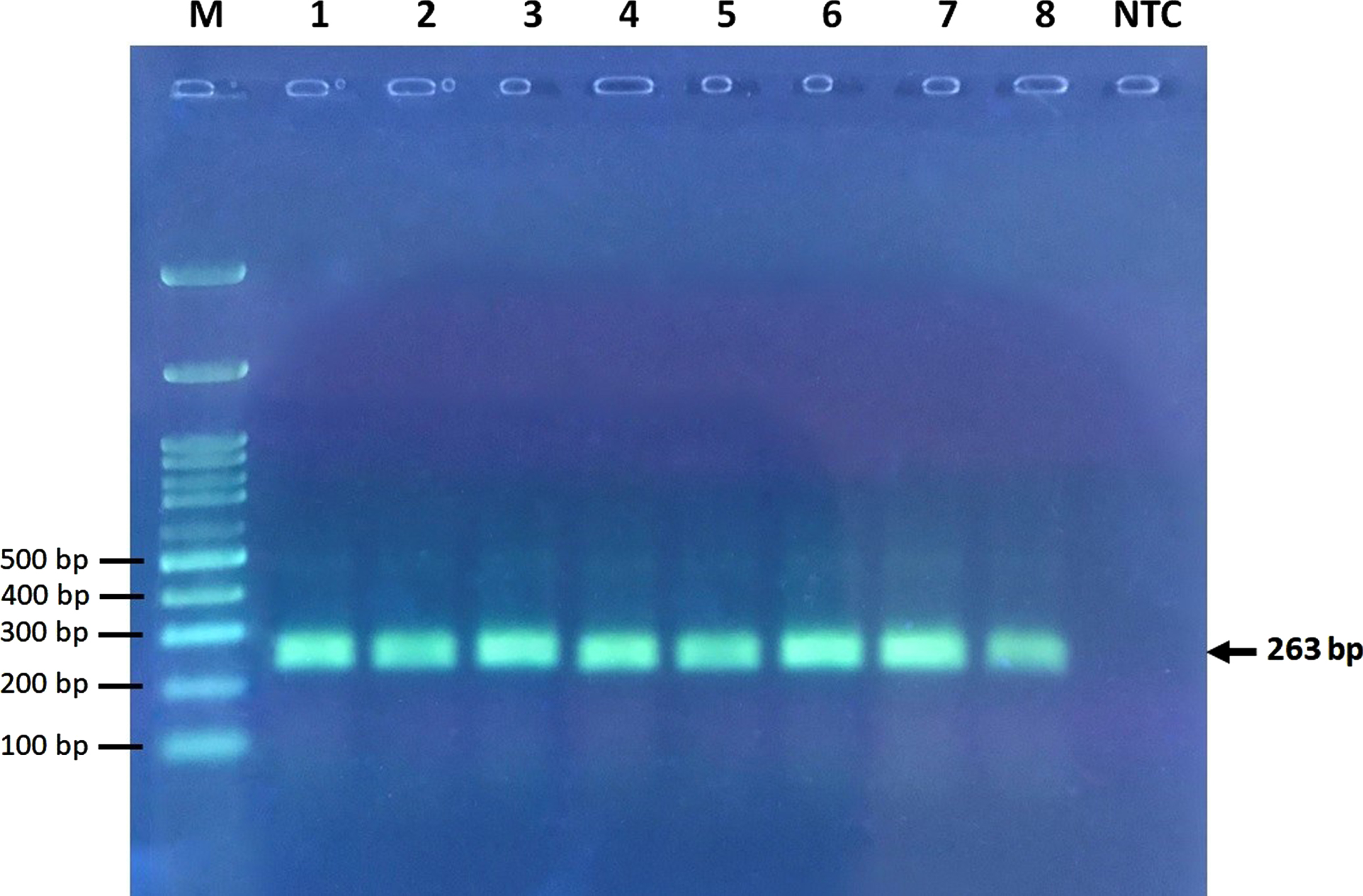
Electrophoresis of undigested PCR products. M, marker ladder; NTC, no template control ladder.
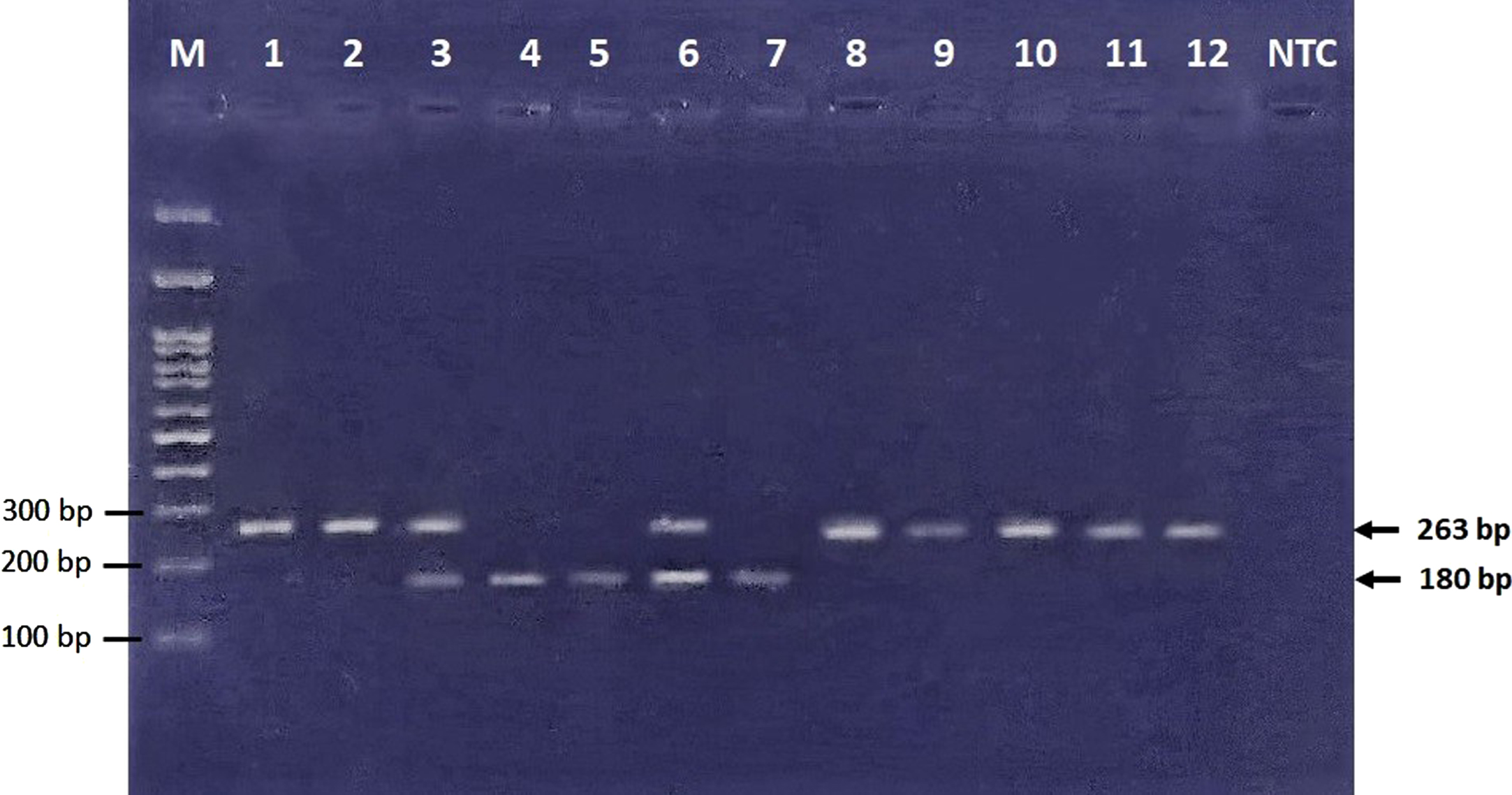
Electrophoresis of restriction products. 83 bp band cannot be visualized in this figure due to usage of 3% agarose gel, which can show DNA fragment size of larger than 100 bp. GG (wild type homozygote genotype) ⟶ Bands No. 1, 2, 8–12 (263 bp). GA (heterozygote genotype) ⟶ Bands No. 3, 6 (263, 180, and 83 bp). AA (mutant homozygote genotype) ⟶ Bands No. 4, 5, 7 (180 and 83 bp).
Statistical analysis
Hardy-Weinberg equilibrium analysis was performed with chi-square goodness-of-fit test. Categorical variables were expressed as n (%) and analyzed with chi-square test or Fisher’s exact test where appropriate. Continuous variables were expressed as mean±standard deviation (SD) and analyzed with independent samples T-test. Comparison of genotype and allele frequencies between patients and controls were made using the logistic regression analysis to assess odds ratio (OR) and 95% confidence interval (CI). Statistical analyses were performed using SPSS (version 26). α= 0.05 was used as statistical level of significance.
RESULTS
Data from 300 subjects in two groups of cases and controls were collected and analyzed. The mean±SD of age in the case and control groups was 83.01±7.24 and 76.94±9.54, respectively (p < 0.001). Thirty-eight (25.3%) and sixty-two (41.3%) were male in the case and control groups, respectively (p = 0.002).
The genotype frequencies of the IRS-1 gene rs1801278 polymorphism were not in Hardy-Wein-berg equilibrium in both groups.
Figure 3 displays sequencing results of the wild-type and variant alleles of the Arg972 IRS-1 gene polymorphism (rs1801278).
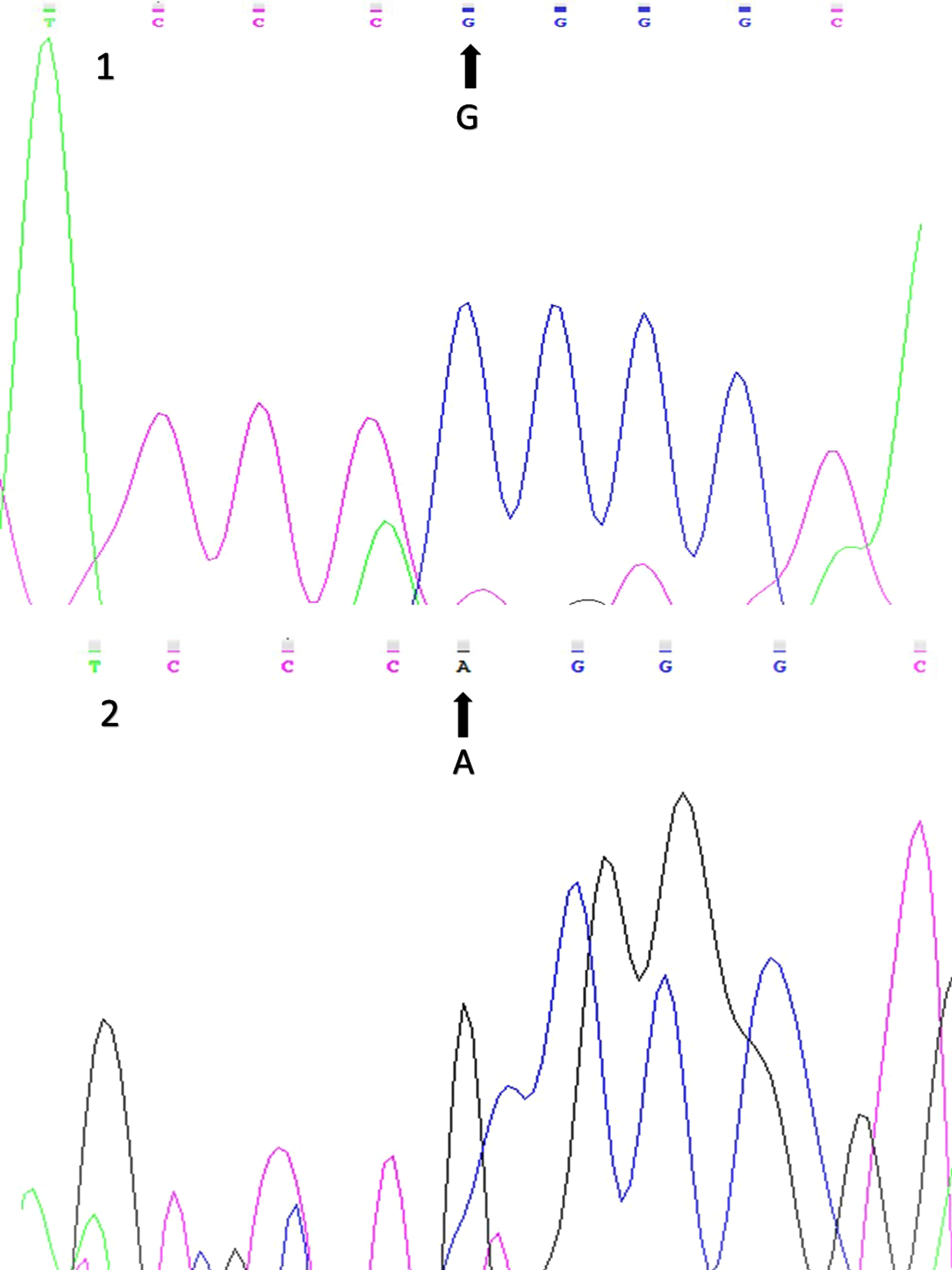
Sequencing results for the Arg972 IRS-1 (rs1801278) gene polymorphism (1: wild-type allele; 2: variant allele).
The prevalence of A (mutant) allele in case and control groups were 31.67% and 19.67%, respectively. By using logistic regression analysis and considering age, sex, and allelic frequencies as cov-ariates, A allele was significantly associated with the risk of AD after adjustment for sex and age (p = 0.04, adjusted OR: 1.77, 95% CI: 1.00–3.11) (Table 2).
Comparison of genotypic and allelic frequencies of Arg972 IRS-1 gene polymorphism between AD cases (N = 150) and healthy controls (N = 150)
aAdjusted OR is based on the logistic regression with covariates to be age, sex, and genotypic frequency. bAdjusted OR is based on the logistic regression with covariates to be age, sex, and allelic frequency.
The prevalence of AA (mutant homozygote IRS-1 polymorphism) genotype in case and control groups were 26% and 11.33%, respectively (Table 2). The prevalence of GA (heterozygote IRS-1 polymorphism) genotype in the case and the control groups were 11.33% and 16.67%, respectively. By using logistic regression analysis and considering age, sex, and genotypic frequencies as covariates, only AA genotype was significantly associated with the risk of AD after adjustment for sex and age (p = 0.01, adjusted OR: 2.39, 95% CI: 1.22–4.66). GA genotype was not associated with the risk of AD after adjustment for sex and age (p = 0.37, adjusted OR: 0.71, 95% CI: 0.34–1.48).
DISCUSSION
In this case-control study, we enrolled 150 LOAD patients and 150 unrelated controls for evaluation of the association of Arg972 IRS-1 gene polymorphism with the risk of AD in an Iranian population. A allele was significantly associated with the risk of AD, compared to G allele after adjustment for sex and age. Furthermore, AA genotype was significantly associated with the risk of AD, in contrast to GG and GA genotypes after adjustment for sex and age.
To the best of our knowledge, only one study evaluated the association of Arg972 IRS-1 (rs1801278) gene polymorphism with the risk of AD [28]. Wang et al. [28] in a case-control study in 2014, evaluated 1113 AD patients and 1113 healthy controls which were matched for sex, age, body mass index, residence area, and education level. The authors showed that 5.7% of the patients and 2% of the controls had AA genotype (mutant homozygote), respectively and 42.3% of the patients and 20.1% of the controls had GA genotype (heterozygote Arg972 IRS-1), respectively. Moreover, the authors found that both genotypes were significantly associated with an increased risk of AD after adjustment for comorbidities including T2DM, coronary artery disease, and hypertension (p < 0.001) [28]. Also, 26.8% of the patients and 12% of the controls carried the mutant A allele, respectively and the A allele was significantly associated with an increased risk of AD after adjustment for the above-mentioned comorbidities (p < 0.001). On the other hand, in each age sub-group of the cases, the prevalence of AA genotype, unlike GG genotype, in patients with a Mini-Mental State Examination (MMSE) score≤14 was high, compared with that in patients with MMSE score≥15–26 (p < 0.001). Furthermore, it was demonstrated that patients with GG genotype had higher MMSE score in each age group, compared with GA and AA genotypes (p < 0.05). It was concluded that Arg972 IRS-1 (rs1801278) gene polymorphism is significantly associated with AD in Han Chinese population. Moreover, the authors stated that this polymorphism may also have a prognostic value for AD [28].
The findings of the study of Wang et al. [28], were in agreement with those of our study demonstrating a significant association of A allele and AA genotype with increased risk of AD. Unlike the findings of Wang et al. [28], we did not find a higher prevalence of GA genotype in the patients even after adjustment for sex and age.
Hamilton et al. [33] investigated the role of insulin signaling-related candidate gene polymorphisms in 954 LOAD patients higher than 60 years of age and 1,106 controls in a Caucasian population in England. The authors showed that there was no significant association of IRS-1 polymorphism (rs1801123) with AD.
Giedraitis et al. [32] used available data from two genome-wide association studies [34, 35]. They showed that IRS-1 polymorphism (rs10187726) had a significant association with LOAD in one of the mentioned studies [34].
In addition, the literature suggests no association of other IRS-1 gene variants (rs2234931, rs2943634) with AD in Caucasian populations [28, 39].
The above-mentioned studies suggest possible ethnic influences on the association between IRS-1 gene variants and the risk of AD.
There are caveats with respect to the present study. First, data concerning comorbidities and clinical characteristics such as T2DM, plasma glucose, heart disease, hypertension, weight, height, body mass index, and waist/hip ratio of the participants were not collected and therefore we were not able to adjust the results accordingly. For example, an association of rs1801278 with the risk of T2DM has been reported [29–31]. Therefore, future studies with adjustment of these parameters would be helpful. Second, the genotype frequencies of the gene variant were not in Hardy-Weinberg equilibrium. However, inasmuch as 10% of all genotype–phenotype association studies show deviation from Hardy-Weinberg equilibrium [40], the results of our trial cannot be considered abnormal. Nevertheless, we suggest future investigations with larger sample sizes.
To sum up, the findings from the present study indicate a significant association of mutant A allele and mutant homozygote AA genotype with the risk of AD after adjustment for sex and age. Further investigation is needed to assess the effects of rs1801278 polymorphism on the severity of cognitive impairment to assess prognostic values.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
Guilan University of Medical Sciences provided financial support for this study.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
