Abstract
Background:
As the sixth-leading cause of death in the United States, Alzheimer’s disease (AD) entails deteriorating endothelial control of blood flow throughout the brain. In particular, reduced inward-rectifying K+ (KIR) channel function in animal models of aging and AD compromises endothelial function and optimal perfusion of brain parenchyma. Deficient endothelial KIR channels may result from aberrant interaction with plasma membrane cholesterol as a primary regulator of membrane fluidity and ion channels.
Objective:
We tested the hypothesis that mild methyl-β-cyclodextrin (MβCD) treatment to reduce membrane cholesterol may restore endothelial KIR channel function in brain endothelium of old AD mice.
Methods:
Membrane potential was continuously measured in isolated endothelial tubes from posterior cerebral arteries of young (1 to 3 months) and old (16 to 19 months) female
Results:
SKCa/IKCa channel function for producing hyperpolarization remained stable regardless of age group and MβCD treatment (
Conclusion:
In female animals, MβCD treatment of brain endothelium selectively restores KIR versus SKCa/IKCa channel function during AD. Thus, the endothelial cholesterol-KIR channel interface is a novel target for ameliorating perfusion of the AD brain.
INTRODUCTION
Alzheimer’s disease (AD) is the leading type of dementia (∼6.2 million people in the United States alone) and continues to rise in parallel with an increasingly aging demographic of individuals of age 65 years and older [1]. Moreover, females in the general population indicate a higher prevalence, severity, and rate of AD pathogenesis even after correcting for age [2, 3]. Thus, especially in the context of biological sex, the etiology of AD remains obscure with no effective treatment strategies in place accordingly. Emerging evidence suggests maintenance or restoration of cerebrovascular health for optimal brain perfusion as a solution. In particular, how endothelial function may be harnessed to prevent dementia per conditions of aging [4, 5] and development of AD pathology [6–8].
In addition to pre-capillary small- and intermediate-Ca2+-activated K+ (SKCa/IKCa) channels, endothelial inward-rectifying K+ (KIR) channels are integral to cerebral blood flow regulation [9, 10]. Our group has demonstrated that the hyperpolarizing function of endothelial KIR channels of posterior cerebral arteries (corresponding to vasodilation and maintenance of cerebral blood flow) is reduced during conditions of aging [5] and AD [7]. Further, another recent study observed lower KIR2 channel current density in brain capillary endothelium in 5xFAD mice (age, 12 to 13 months; severe AD pathology as neuronal loss [11]), with overall consequences of diminished functional hyperemia in response to both whisker stimulation and elevated K+ [8]. In turn, this study identified a potential therapeutic role for membrane lipid regulation via enhanced phosphatidylinositol 4,5-bisphosphate (PIP2) supply to restore deficient cerebrovascular KIR2 channels [8].
As an additional key component of lipid regulation while integral to development of AD pathology [12–14], membrane cholesterol may also inactivate vascular KIR2 channels during conditions of health [15] and hypercholesterolemia [16, 17], with the latter most pronounced for diminished endothelial KIR2 channel function in particular. This regulatory role for cholesterol has not been tested using direct electrophysiological measurements of freshly isolated and intact endothelium in a study model of AD. Thus, we have tested the hypothesis that
METHODS
Animal care and use
All animal care use and experimental protocols for this study were approved by the Institutional Animal Care and Use Committee of Loma Linda University and performed in accord with the National Research Council’s “
Solutions and reagents
Preparation and composition of all solutions have been previously described for measurements of Vm in intact endothelium freshly isolated from mouse posterior cerebral arteries [5, 22]. Briefly, physiological salt solution (PSS; pH 7.4) was prepared for continuous superfusion of cerebral endothelial tubes [(in mmol/L): 140 NaCl, 5 KCl, 2 CaCl2, 1 MgCl2, 10 HEPES, 10 Glucose]. For dissection of brain and arteries, PSS lacking CaCl2 contained 0.1%bovine serum albumin. For isolation of intact endothelium, an enzyme cocktail [0.31 mg/mL papain (P4762, Sigma), 0.5 mg/mL dithioerythritol (D8255, Sigma), 0.75 mg/mL collagenase (C8051, Sigma), and 0.13 mg/mL elastase (E7885, Sigma)] was prepared in dissociation PSS containing reduced Ca2+ (0.1 mmol/L CaCl2) and 0.1%bovine serum albumin. The 15 mmol/L KCl solution was prepared with an equimolar decrease of NaCl accordingly in order to maintain osmolarity. All reagents were purchased from Sigma-Aldrich (St. Louis, MO, USA) or ThermoFisher Scientific (Pittsburgh, PA, USA) unless otherwise indicated.
Dissection of cerebral arteries and isolation of endothelial tubes
Sequential dissection of mouse brain for preparation of posterior cerebral arteries and isolation of arterial endothelium have been previously described and illustrated in our previous studies [5, 22]. Briefly, following isoflurane inhalation for anesthesia and decapitation, the brain was acutely removed and placed in chilled (4°C) dissection PSS. Posterior cerebral arteries were carefully dissected from the brain and cut into segments. Arterial segments were partially digested in dissociation PSS containing the enzyme cocktail at 34°C for 10 to 12 min. Following digestion, arterial segments were triturated using borosilicate glass capillary tubes [1B100-4, World Precision Instruments (WPI), Sarasota, FL, USA] with an internal tip diameter of 80–120μm to remove adventitia, smooth muscle cells, and internal elastic lamina to isolate intact endothelial tubes. Endothelial tubes were continuously superfused with PSS at 37°C.
Recording of intracellular V
m
Recording of Vm in mouse cerebral endothelium has been previously described and illustrated [5, 22]. Briefly, Vm of isolated endothelial tubes was recorded with Axoclamp electrometers (2B and/or 900A; Molecular Devices, Sunnyvale, CA) using microelectrodes pulled (P-97, Sutter Instruments) from glass capillary tubes (GC100F-10, Warner Instruments, Holliston, MA, USA). Under continuous superfusion with PSS, an endothelial cell was penetrated with a microelectrode backfilled with 2 mol/L KCl (tip resistance: ∼150 M
Cholesterol assay of cerebral blood vessels
Cholesterol content was determined in total blood vessel content in the brain using Cholesterol Quantification Assay Kit (Sigma) in accord with manufacturer’s instructions. Brain microvessel fragments were isolated from the mouse brains using a previously illustrated protocol [23]. Cholesterol content (μmol/L) was normalized to total phospholipid (μmol/L) determined from the same blood vessel isolates using Phospholipid Assay Kit (Sigma) according to manufacturer’s instructions.
Cholesterol imaging of endothelial cell tubes
Endothelial cell tubes were secured on a glass coverslip with glass micropipettes using two micromanipulators and superfused with PSS while gradually raising the bath temperature to 37°C. Endothelial cell membrane cholesterol was detected by incubating cells with Filipin-III (50μg/ml, Sigma) at 37°C for ∼1.5 h under light-protected conditions. The cells were then washed with fresh PSS and images were acquired using a fluorescent microscope (Eclipse Ti-S; Nikon), a 60X objective [Plan Apo λ (numerical aperture: 0.95); Nikon)], a solid-state light source containing a violet channel (MIRA Light Engine; Lumencor, Inc., Beaverton, OR, USA), a 16-megapixel monochrome camera (DS-Qi2, Nikon), and Nikon imaging software (NIS Elements-F 4.60.00). For unpaired image acquisitions prior to staining with Filipin, endothelial cell tubes were treated with methyl-β-cyclodextrin (MβCD) for 20 min followed by 1 h washout period at 37°C. For ideal visibility, all images illustrating Filipin staining were pseudo-colored green.
Pharmacology
An effective half-maximal concentration of NS309 (EC50, ∼1μmol/L; 3895, Tocris, Bristol, England) was used to directly evaluate function of SKCa/IKCa channels to produce hyperpolarization [5, 22]. As consistent with our previous studies [22], DMSO solvent (≤0.1%) for working stocks of NS309 was found to have no effect on endothelial Vm on its own for≤5 min applications used for respective drug treatments in cerebrovascular endothelial tubes. Elevated level of extracellular KCl ([K+]o: 15 mmol/L) was used to stimulate endothelial KIR2 channels for hyperpolarization [22, 24]. MβCD (1μmol/L; 332615, Sigma) was prepared in PSS to reduce cholesterol in the endothelial cell membrane [25, 26]. Respective K+ channel activating agents were applied before and after washout of a 20 min application of MβCD treatment. Resting Vm was typically allowed≥2 min to stabilize before application of a pharmacological agent, whereby each application was allowed sufficient time (≥3 min) to record peak Vm responses. In between individual applications, the endothelial tube was washed with PSS to baseline conditions.
Data and statistical analysis
For quantification of cholesterol levels normalized to total phospholipid content,
For simultaneous comparisons among both age groups±MβCD treatment, statistical analysis included a two-way Analysis of Variance (ANOVA) with Tukey’s
RESULTS
The goal of this study was to ascertain the mechanism of how cerebrovascular endothelial KIR2 channel function is reduced during AD pathology as observed in our recent study [7]. In accord with our current hypothesis, we examined the potential impact of membrane cholesterol on endothelial K+ channel function for hyperpolarization of Vm as components of EDH during development of AD pathology. As the most impacted biological sex in the clinical AD population, we chose to center the current studies on use of females [2, 3]. It should be noted that female
Total cholesterol content in cerebral blood vessels during conditions of pre-AD and AD
Cholesterol dyshomeostasis is known to underlie conditions of atherosclerosis per aging [29] while the presence of the apolipoprotein E (
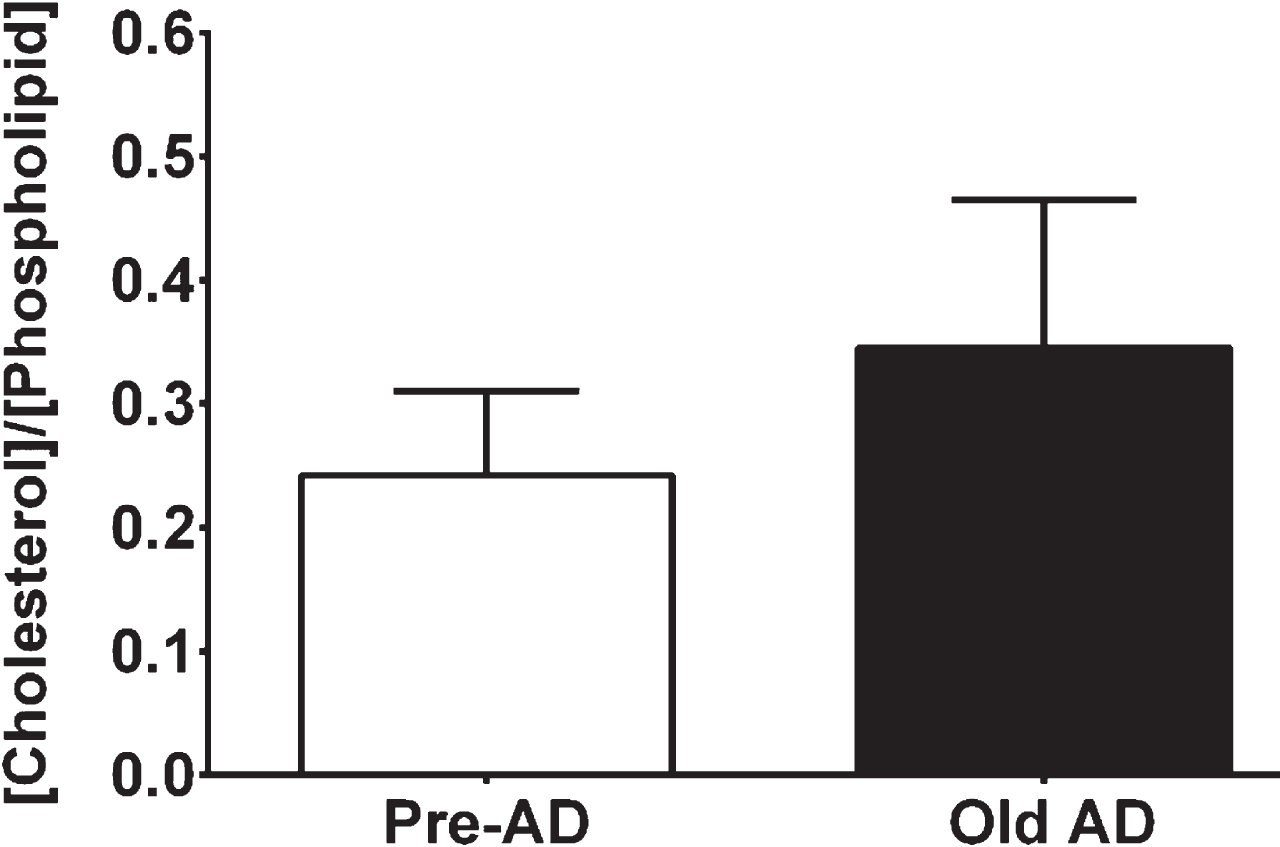
Cerebrovascular cholesterol content among pre-AD and AD groups. Data represent molar ratio of cholesterol content per total phospholipid content for respective groups.
Membrane cholesterol in endothelial cell tubes during conditions of pre-AD and AD
Presence of cholesterol inclusion bodies or “crystals” appear in endothelial cells during conditions of disease such as atherogenesis [26] and can be removed using MβCD [32]. With use of Filipin to stain membrane cholesterol [26, 32], we observed fluorescent crystals in endothelial tubes during AD pathology (Fig. 2F; 15.3±0.7 crystals/mm2) that were significantly reduced in number (Fig. 2H; 3.6±0.9 crystals/mm2) following mild MβCD treatment (1μmol/L; 20 min incubation, 1 h washout) or in young, pre-AD animals regardless of MβCD treatment (Fig. 2B, D; 0.2±0.1 crystals/mm2). These data demonstrate that membrane cholesterol crystals are a feature of cerebrovascular endothelium during AD pathology which may impact function of K+ channels crucial to endothelial function underlying perfusion of the brain.
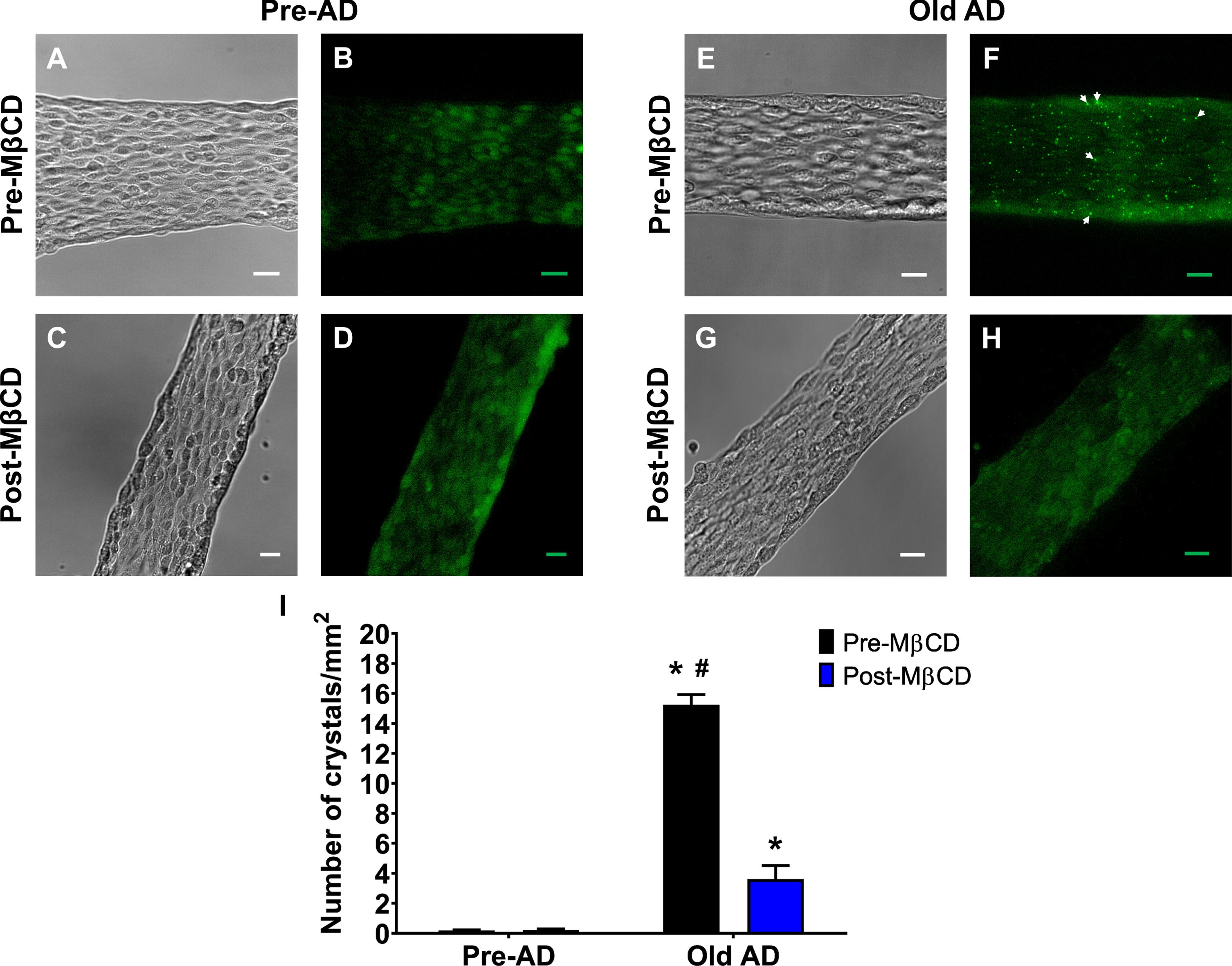
Presence of cholesterol crystals in AD animals and reduction following MβCD treatment. Representative (A, C, E, G) brightfield and (B, D, F, H) Filipin-III images (green pseudo-color) of cerebrovascular endothelial cell tubes isolated from female animals. A, B) Pre-AD images without MβCD treatment. C, D) As shown in (A) & (B) respectively after MβCD treatment (1 mmol/L, 20 min incubation, & 1 h wash). E-H) As shown in (A), (B), (C), and (D) respectively for AD animals. White arrows indicate cholesterol “crystals” in endothelial tubes during AD conditions in the absence of MβCD treatment. The scale bar throughout panels represents 20μm. I) Summary data for number of crystals per unit area (mm2).
Effect of MβCD on cerebrovascular endothelial SK
Ca
/IK
Ca
channel function
Endothelial SKCa/IKCa channels are integral to a major blood flow regulation pathway as EDH in cerebral arteries [22, 33] and arterioles [34]. We have previously found that SKCa/IKCa channel function is preserved in females while modestly enhanced (∼20%) in males during AD pathology [7]. Accordingly, we found that peak hyperpolarization to NS309 (1μmol/L) as SKCa/IKCa channel activation was not altered by advanced AD pathology or MβCD treatment (1μmol/L) (Fig. 3;
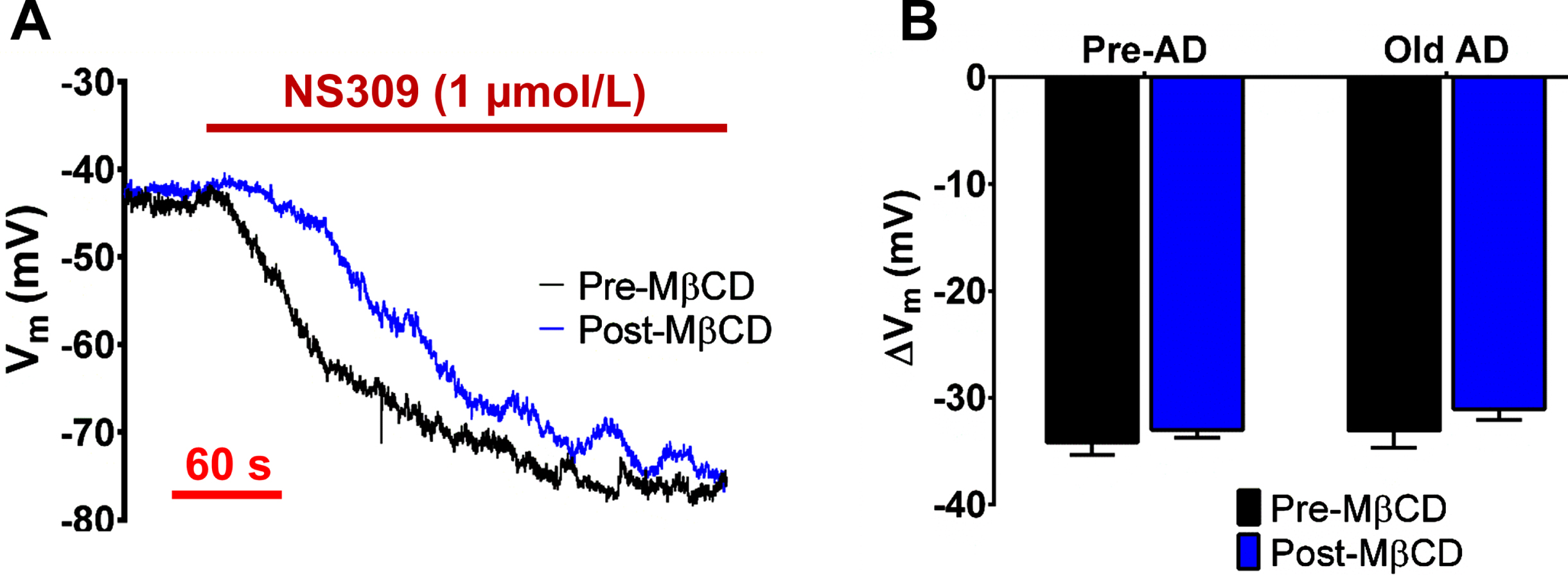
SKCa/IKCa channel function is not impacted by conditions of AD or membrane cholesterol regulation. A) Representative recordings of SKCa/IKCa channel hyperpolarization in Old AD females in response to NS309 (1μmol/L) before and after 30 min washout of MβCD (1 mmol/L, 20 min). B) Summary data for peak
Impact of MβCD treatment on cerebrovascular endothelial K
IR
2 channel function
KIR2 channel function is crucial for cerebral vasodilation and blood flow [10, 15] and is impacted by conditions of hypercholesterolemia [16, 17]. Also, we have recently found reduced KIR2 channel function with advancing age [5] while exacerbated by conditions of AD [7] in both biological sexes. Thus, we tested whether treating with a relatively low dose of MβCD, an agent that lowers membrane cholesterol [25, 26], could restore KIR2 channel function in endothelial tubes from AD mice. Relative to SKCa/IKCa channel activation, there is a modest Vm hyperpolarization response to activation of cerebrovascular endothelial KIR2 channels with 15 mmol/L KCl (
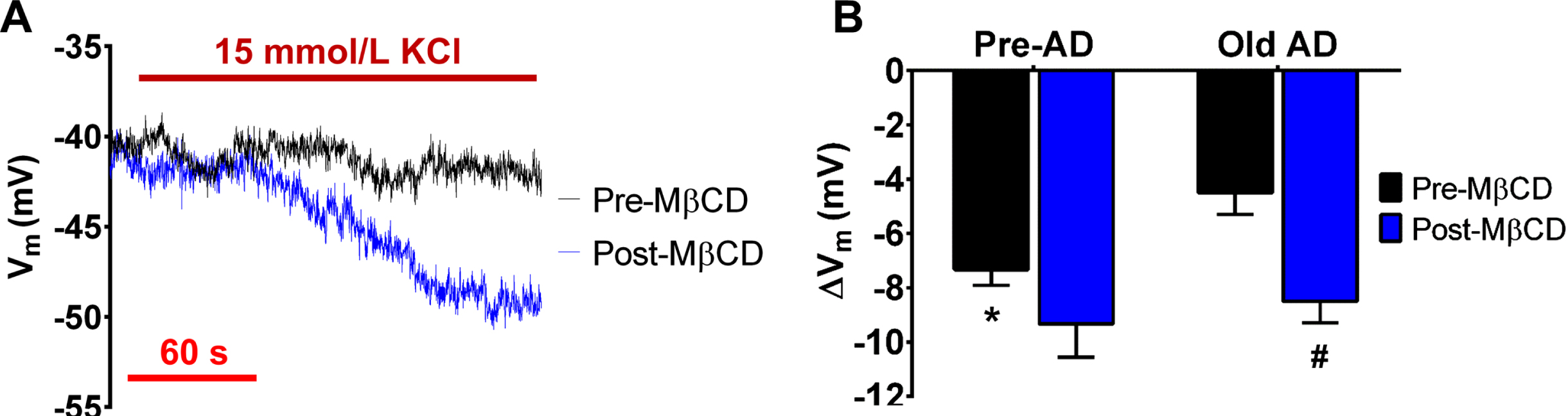
Reduction of membrane cholesterol restores endothelial KIR2 channel function during AD pathology. A) Representative recordings of KIR2 channel-mediated hyperpolarization in Old AD females in response to 15 mM KCl before and after 30 min washout of MβCD (1 mmol/L, 20 min). B) Summary data for peak
DISCUSSION
Understanding blood flow regulation throughout the cerebrovascular network is crucial because chronic, inadequate delivery of oxygen and nutrients throughout the brain contributes to neurodegenerative pathology such as AD [6, 35]. Further, with recognition of the endothelium as an organ that “feeds” all other organs throughout the body [18], naturally, endothelial function has become pivotal for fundamental studies of brain health [9, 37], the aging brain [4, 38], and select conditions of AD [7, 8]. In particular for AD, there are remaining knowledge gaps for potentially altered functions of cerebrovascular endothelial ion channels per blood flow regulation, primarily as the transient receptor potential [39–41] and K+ channels (e.g., KCa and KIR subtypes; [7, 42]). In a recent study [7], we have demonstrated that endothelial KIR channel function of posterior cerebral arteries is reduced during AD by ∼50%, whereas SKCa/IKCa channels are relatively stable. This general finding has also been reinforced for KIR channel function in capillary endothelium during AD [8], altogether establishing a premise for mechanistic investigation and corresponding therapeutic strategies involving cerebrovascular K+ channels. With a basis of KIR channels modulated by membrane cholesterol [17, 43] and cholesterol dysregulation playing a key role in AD etiology [14, 31], the goal of this study was to investigate whether mild MβCD treatment to reduce membrane cholesterol would significantly improve cerebrovascular KIR channel function in old
Endothelial membrane cholesterol and use of cyclodextrin for treatment of AD
Cholesterol is essentially present in the plasma membrane of all mammalian cells as one of the major lipid components for cellular growth and function, whereby its excess beyond physiological levels is generally associated with the development of disease including AD [14, 44–46]. Although not statistically significant, we have found that the mean for cerebrovascular cholesterol (normalized to total phospholipid content) was ∼40%higher in AD versus pre-AD animals (Fig. 1). Thus, our data for cellular cholesterol content suggest that cerebral blood vessels may or may not be enriched with excessive levels of cholesterol among individual animals during conditions of AD. However, with use of fluorescent Filipin staining, we observed apparent cholesterol crystals in endothelial membranes of AD animals that were substantially reduced in number by ∼75%following MβCD treatment (Fig. 2). In females, elevated cholesterol levels in human frontal cortex [47] and rodent endothelial plasma membranes [48] have been shown during conditions of aging as a key risk factor for cognitive decline towards AD [1].
Cyclodextrins such as MβCD are cyclic oligosaccharide compounds and have been widely used to bind to cholesterol in a concentration-, time-, temperature-, and cell type-dependent manner [25]. With a reference point of 5 mmol/L MβCD (30 to 60 min incubation) for patch-clamp measurements of rodent vascular cells (brain [15], mesentery [16]), we determined that 1 mmol/L MβCD (20 min) was sufficient for restoration of reduced KIR channel-dependent hyperpolarization for AD animals following 30 min of MβCD washout. In addition to the precision in time for various drug-response measurements, it should be noted that a major strength of our electrophysiological abilities encompasses a scale of hours (versus seconds or minutes) [21] and thus, we encompassed paired examinations of Pre-MβCD and Post-MβCD conditions throughout individual endothelium preparations.
SK
Ca
/IK
Ca
channel function is unaffected, whereas K
IR
channel function is restored by mild MβCD treatment of cerebrovascular endothelial membrane cholesterol during AD
Endothelial SKCa/IKCa channels are vital for cerebral blood flow regulation [33, 34], whereby their function is not impaired by conditions of AD alone [7]. Accordingly, the current study demonstrates that MβCD has no significant impact on SKCa/IKCa channel activity to produce membrane hyperpolarization (Fig. 3), suggesting that membrane cholesterol does not play a major role for regulation of cerebrovascular SKCa/IKCa during health and disease. Overall, this lack of effect of MβCD treatment on SKCa/IKCa channel function is consistent with previous studies (reviewed in [49]). As highly expressed in smooth muscle cells [50], large conductance KCa (BKCa) channels are suppressed [51] or activated in response to membrane-enriched cholesterol depending on expression of
KIR channels are integral to neurovascular coupling and the overall coordination of cerebral pre-capillary vasodilation and blood flow [10, 53] with a unique capacity for also boosting SKCa/IKCa channel hyperpolarization [54, 55]. Elevated membrane cholesterol suppresses endothelial KIR2 channel currents [16, 45] by interacting with a CD loop region of the C-terminus as a cytosolic component of the channel [56]. Remarkably, we found that KIR2 channel-dependent hyperpolarization increased progressively with time of washout following MβCD treatment to ∼9 mV and ∼14 mV for both female Pre-AD and AD animals at 30 min and 60 min respectively (Fig. 4 and Results text). The enhancement of hyperpolarization responses in pre-AD animals was unexpected but previous evidence suggests that, even in healthy humans, normal cholesterol levels can suppress endothelial function [57]. Also, we should note that mechanisms of local control of KIR channels can occur via glycocalyx regulation as well [43]. Relative contributions of membrane cholesterol-dependent versus -independent modulation of endothelial ion channels remain to be resolved for conditions of AD.
Experimental considerations and potential treatment strategies
Although KIR2 channels are also present on cerebrovascular smooth muscle cells [10, 15], we studied the key component of coordinated electrical signaling of cerebral blood flow control as the endothelium [9, 22]. In addition to retention of
SUMMARY AND CONCLUSIONS
Impairment of cerebral blood flow during AD is due, in large part, to endothelial dysfunction in the brain. Therefore, it is crucial to locate mechanisms and therapies that center on improvement of cerebral perfusion. With following up from our recent studies demonstrating a functional decrease of endothelial KIR2 channels, the current study was strategically undertaken to restore KIR2 channel-dependent hyperpolarization in response to elevated extracellular K+. We found that mild application of a cholesterol removal agent as MβCD progressively restored and even enhanced KIR2 channel-dependent hyperpolarization while SKCa/IKCa channels were unaffected. Further, this finding was observed in females as the representative biological sex of the clinical AD population [2, 3]. Altogether, these findings suggest that the endothelial cholesterol-KIR channel interface may be a novel therapeutic target for ameliorating cerebrovascular function to optimize perfusion of the AD brain.
Footnotes
ACKNOWLEDGMENTS
This research has been supported by National Institutes of Health grant R56AG062169 (to E.J.B.). The content of this original article is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
