Abstract
In recent years there has been increasing interest in examining the role of empathic abilities in Alzheimer’s disease (AD). Empathy, the ability to understand and share another person’s feelings, implies the existence of emotional and cognitive processes and is a pivotal aspect for success in social interactions. In turn, self-empathy is oriented to one’s thoughts and feelings. Decline of empathy and self-empathy can occur during the AD continuum and can be linked to different neuroanatomical pathways in which the cingulate cortex may play a crucial role. Here, we will summarize the involvement of empathic abilities through the AD continuum and further discuss the potential neurocognitive mechanisms that contribute to decline of empathy and self-empathy in AD.
INTRODUCTION
Alzheimer’s disease (AD) is the most prevalent form of neurodegenerative dementia. It is typically characterized by early and progressive episodic memory loss. The progressive neurodegeneration initially involves the hippocampi, the entorhinal, and cingulate cortex, and, subsequently, it spreads to the entire temporal, parietal, and frontal lobes [1]. Research in AD has recently focused on examining the concept of empathy. It is a complex and multidimensional construct in relation to the ability to understand and share the emotional and mental states of others [2, 3]. Empathy plays a pivotal role in social interactions as an enhancer of prosocial behaviors and adaptive social responses to context. Furthermore, empathy correlates positively with the degree of life satisfaction, emotional intelligence and self-esteem [4].
On the other hand, self-empathy can also be distinguished from empathy. Self-empathy is oriented to oneself and, thus its study may help us understand better one’s thoughts and feelings. In both cases, impairment of empathy and self-empathy can occur during the course of the AD and can be observed by anatomical inspection in different brain areas. AD patients have been found to show impaired social behavior, theory of mind, empathy, emotion recognition, and inaccurate self-awareness, and, although uncommon as first symptoms, severe behavioral alterations such as disinhibition, social awkwardness, and apathy [5]. Here we will discuss the involvement of empathy and self-empathy through the AD continuum, also providing an in-depth revision of the shared neural pathways by both empathy and self-empathy.
EMPATHY
Empathy enables individuals to participate in social situations, allowing them to perceive and recognize thoughts, emotions, and behaviors of others. Specifically, the ability to recognize and understand emotions and intentions in others is crucial for maintaining successful social interactions. There is considerable consensus that empathy comprises two main components [3, 6]. First, affective empathy refers to the ability to recognize, understand, and share the affective other’s states, emotions, or feelings. Second, cognitive empathy refers to the ability to understand and predict other’s beliefs, thoughts, or intentions and may be considered as equivalent to the so-called construct of theory of mind. This conceptualization is supported by the involvement of different neural pathways that underlie the two dimensions of empathy [6, 7]. Regions underlying the functions of affective empathy are the inferior frontal cortex, inferior parietal lobe, anterior cingulate cortex (ACC), and anterior insula [7–11]. Conversely, cognitive empathy relies on the medial prefrontal cortex, the superior temporal sulcus, the temporo-parietal junction, and the temporal poles [7]. Further evidence in favor of this dissociation have been found in separate EEG event-related potentials associated with affective and cognitive empathy [12]. In any case, it should be noted that the development of empathy must be contextualized in the socio-cultural environment in which an individual grows up. That would explain the premorbid interindividual differences of empathy. Factors such as education, gender, and social engagement have been examined to better understand the role of possible modulators between aging, AD, and empathy. Thus, some studies have reported that characteristics such as higher education, female gender, and successful interpersonal relationships correlate with greater empathic abilities [13–15].
THE ROLE OF EMPATHY IN HEALTHY AGING AND AD CONTINUUM
There is growing evidence indicating that older age is associated with reduced capacity for empathy [16, 17]. Research findings suggest that older adults may have deficits in both affective and cognitive empathic dimensions. Aging is associated with deficits on both first- and, more specifically, second-order theory of mind tasks, which underlies a specific age-related deficit in the ability to infer one’s own and other people’s thoughts, feelings, and mental states [4]. Likewise, older adults also show decline in affective recognition, particularly for negative facial expressions, with positive emotions relatively preserved [18]. These changes, rather than be due to selective declines in brain regions involved in perceiving specific emotions, seem to be related to the way in which older adults process faces [19].
During aging, there is a decrease in the reported social network sizes [20, 21]. However, this seems not to be a random decrease since a possible explanation is that older people prioritize positive social interactions over other ones [22]. It has been suggested that such prioritization occurs in normal aging, but this phenomenon would probably not operate in the same way in the very early preclinical stages of AD. Thus, an incipient sign for the development of a later neurodegenerative disorder such as AD could be the presence of subtle changes in social functioning that lead to violation of social conventions, social withdrawn, and isolation [23]. However, it should be noted that those changes could be secondary to the general cognitive decline that accompanies AD [24, 25]. In particular, cognitive empathy has been found to be mediated by executive functions [26, 27].
Changes in the ability of recognizing and understanding emotions and mental states in others seem to occur early in the AD continuum independently on the presence of cognitive deficits [28, 29]. Since social interaction has beneficial effects on cognitive status and specific domains such as memory [30]; a decrease in the quantity and quality of social contacts may be indicative of a neurodegenerative disorder onset. Indeed, social isolation has been associated with faster progression of cognitive impairment and higher mortality [31].
Different types of positive social interactions, especially related to empathy and other personal connections may be involved in the early stages of AD [8]. Indeed, loss of empathy has been reported in AD, particularly the cognitive component of empathy versus a relatively preservation of the affective component [32]. To confirm this association with the disease, some studies have been carried out with cases of familial Alzheimer’s disease (FAD) [33, 34]. The findings of those studies have highlighted the presence of specific early social behavioral changes in carriers of autosomal dominant mutations in AD [33]. These results have been to some extent confirmed in mouse models that express the human genes containing FAD mutations; thus, it has been proven that these mouse models with cognitive dysfunctions also develop early social disturbances [9, 36].
Regarding neuroanatomical bases underlying the link between empathy and AD, the ACC seems to play a crucial role as it is involved in the ability to recognize emotions in others [37]. In fact, there is growing evidence pointing out that empathic relationships are not only related to the “mirror system” but also to the ACC [8]. However, active social decision-making is critical to establishing and maintaining successful social relationships, involving not only the ACC but also the orbital frontal cortex (OFC) as depicted in Fig. 1 [38]. For social decision-making, the ACC would evaluate different predictions about the emotional states of others, while the OFC would judge the value of possible choices of behavior and determine social preferences [10, 39]. Hence, damage in any of these areas could affect social cognition. Although more work needs to be done to find a possible mechanism to link changes in ACC to the alterations in social behavior associated with the early stages of AD, FAD is already providing important clues. In FAD, amyloid is deposited very early in the precuneus [40], which is a brain structure that interacts with ACC through various cortical connections [41]. It could be argued that a decrease in these interactions could affect the functioning of the ACC and, secondarily, empathy.
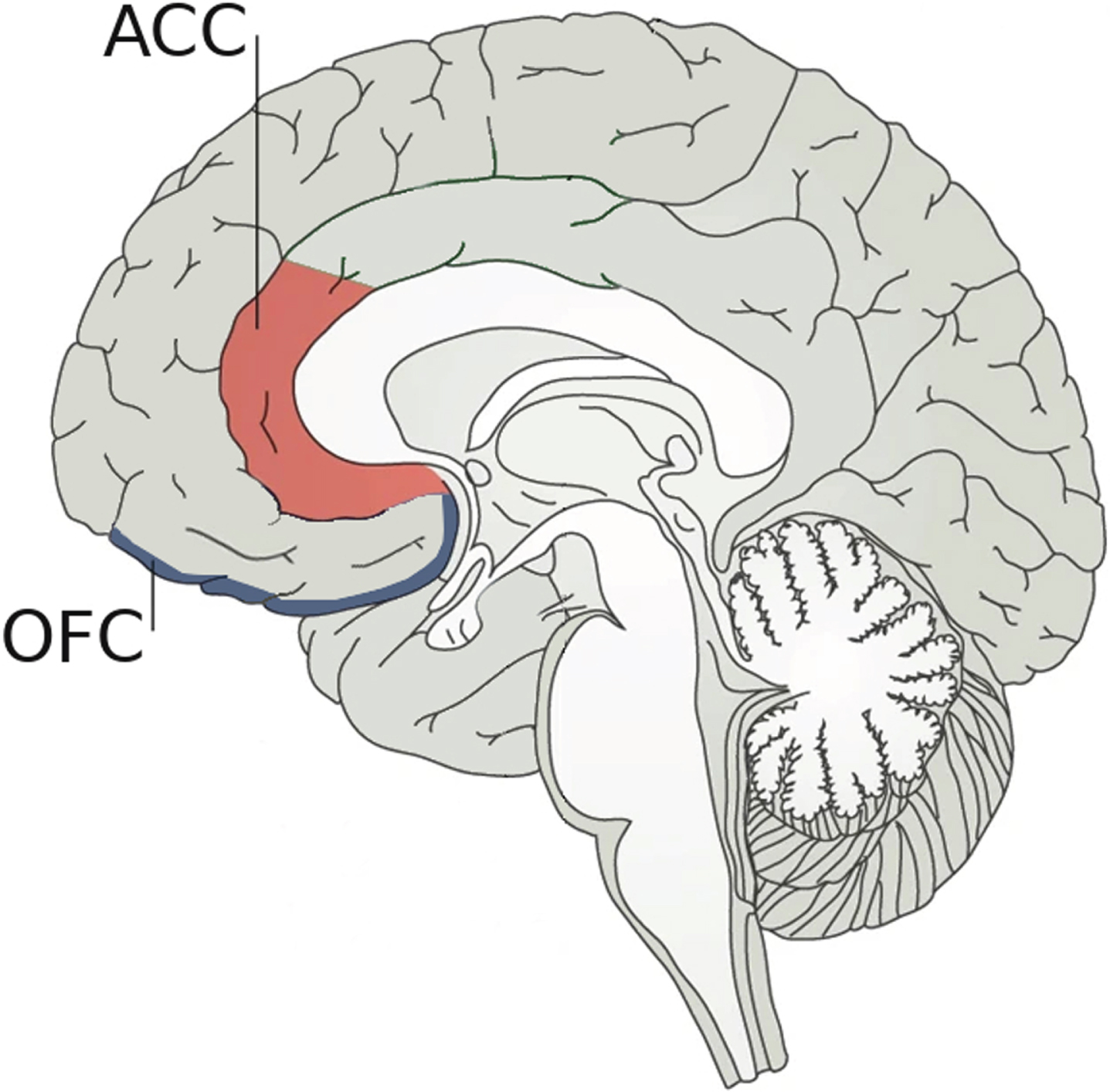
Neuroanatomical bases underlying social decision-making. Social decision-making, which is crucial to establishing and maintaining successful social relationships, is supported by two pivotal brain regions: 1) the anterior cingulate cortex (ACC) that plays an important role in the ability to recognize emotions by evaluating predictions about the emotional states of others; and 2) the orbital frontal cortex (OFC) that judges the possible choices of behavior. Damage in any of these two areas could affect social cognition.
THE ROLE OF SELF-EMPATHY IN AD CONTINUUM
Self-empathy refers to a personal facet of oneself by which one can recognize and become aware of what is going on in one’s own mind [42]. As with empathy, there are two dimensions for self-empathy related to the affective and cognitive components of one’s inner mind. Most importantly, self-empathy provides a unique framework in which to integrate and compare the events of one’s current and past experiences; in this same framework, it is also possible to compare one’s own experiences with those of other people.
In the first phases of the AD continuum a progressive transition takes place between the non-demented asymptomatic stage, in which one continues to live a normal life with appropriate cognitive and social functioning, and the beginning of an incipient subjective cognitive decline plus (SCD-P) phase [43–45]. SCD-P refers to a self-experienced persistent decline in cognitive functioning in comparison with a prior normal status and independent of the objective performance on neuropsychological tests. Self-empathy allows a person to become aware of the first subtle cognitive complaints experienced in the SCD-P phase by comparing one’s current cognitive performance with her own premorbid status as well as with the cognitive performance of other people with similar age or condition.
The most frequent symptom in SCD-P, but not solely, is memory loss. Damage to the connections between entorhinal cortex (EC) and hippocampal areas seems to be at the base of memory complaints [46, 47]. Structural changes in medial prefrontal cortex and temporo-parietal junction [48] as well as in the medial temporal lobe have also been reported in individuals with SCD-P [49]. In the hippocampus, CA1 is the minimum region essential for the functioning of episodic memory [50–53]. Mainly, connections between EC and CA1 are involved in the acquisition of new memories [54]. Injury to these connections can lead to characteristic memory loss in AD. However, not only individual resilience and compensating factors such as cognitive reserve may help to slow down memory loss [55, 56], but also alternative pathways for connecting the cerebral cortex and CA1 have been recently identified. It has been described an alternative neural circuitry that could delay the transition from a non-demented stage to a mild cognitive impairment (MCI) [57]. This alternative pathway, that can connect cerebral cortex to CA1 without going through the EC [58], includes posterior cingulate cortex (PCC), mammillary bodies, and hippocampal CA2. In this circuitry the involvement of PCC appears to be different to the early deficits found in ACC [59]; the functional decrease of PCC occurs later in the AD continuum, specifically in the MCI stage when a decreased connectivity between PCC and hippocampus may be evidenced [59].
From the molecular perspective, it has been identified a high proportion of amyloid-β peptide (Aβ) and a low concentration of tau in the cingulate cortex [60, 61]. However, the opposite trend can be observed in the EC, where more abundant tau deposits and a lower proportion of Aβ have been reported [60]. In this sense, it has been postulated that the alternative neural pathway may lose its functionality and, therefore, its ability to compensate, once the PCC is impaired. The damage in this region could occur when the tau deposits of the EC are transported and reach the PCC. Thus, the transport time of tauopathy from EC to PCC could be related to the time of disease progression from the preclinical and prodromal phases to the dementia stage [62].
As the disease progresses and cognitive impairment worsens, self-empathy also deteriorates. Self-empathy is a necessary precondition for self-awareness of one’s inner state; both are qualitatively different concepts and can be distinguished [7, 65]. From a phenomenological point of view, impairment in self-empathy causes an impairment in self-awareness. This is what can be observed from late MCI to subsequent stages of AD dementia, where patients may begin to suffer from self-empathy deficits that immediately affects self-awareness and prevents them from recognizing the changes caused by the cognitive impairment associated with the disease. Self-awareness would include three different components, namely: 1) the recognition of a specific deficit; 2) the emotional response elicited by that deficit; and 3) the understanding of the functional impact that the deficit has on daily life [63]. Lack of self-awareness of memory deficits in MCI is important because it is a marker for conversion to AD [64].
In 1914, Babinski coined the term anosognosia which, in medical settings, means that a patient is unable to perceive his or her own acquired deficits as well as their functional implications in everyday life [66]. More generally, anosognosia is the most extreme loss of self-awareness that affects to the introspective capacity [67] and produces a lack of free will or freedom of choice, leading to a total dependence from others [68–71]. In the field of dementia, anosognosia is a common symptom of later stages of AD; overall, anosognosia is present in AD patients with an estimated frequency ranging 20–80% [72–75]. It is commonly related to a metacognitive executive dysfunction [76]; indeed, the executive neural networks show some overlap with those that support empathy and self-empathy [77, 78], so neurodegeneration at these networks can lead to the appearance of anosognosia. In particular, the most consistent correlations between anosognosia and cognitive processes include cognitive flexibility, mental control, problem solving and frontal behaviors such as prehension, utilization, imitation, inertia, and indifference [64].
From a neuroanatomical point of view, anosognosia has been associated with extensive brain damage, mainly affecting the right hemisphere, specifically the inferior parietal lobe and frontal inferior and dorsolateral regions [79]. Specifically, in MCI anosognosia is associated with a reduced gray matter volume of the ACC and of the inferior frontal cortex [64] as well as a reduced PCC functioning and a decreased hippocampal metabolism [80]. In the first phases of AD, the presence of anosognosia is associated with different structures of the medial temporal lobe [81] and a specific disconnection within the medial temporal subsystem of the default mode network [82, 83].
DISCUSSION
Empathy and self-empathy abilities have been little researched in the field of AD. In general, as the disease progresses, it is possible to find a correlation between changes in both abilities and brain degeneration [84, 85]. Specifically, it is well-known that through the AD continuum there is a degeneration of the EC [46, 86]. This degeneration has two direct and fatal consequences: on the one hand, the connectivity of the hippocampal CA1 is undermined [87] and, on the other hand, the functionality of the PCC is diminished [59, 88]. In this way, connections between the cerebral cortex and the CA1 are impaired, either through the EC or through the PCC, leading to loss of memory [57]. Thus, by looking at the rate of decline of the cingulate cortex and the empathic abilities in the AD continuum, we can glimpse a link between them (Table 1). Here, we hypothesize that in the early stages of the disease, changes in the ACC would occur and produce a decrease in cognitive empathy and emotion recognition that lead to a decreased in maintaining social relationships; whereas in the later AD stages, changes in PCC would appear and lead to a self-empathy impairment and anosognosia [89].
Proposed neuroanatomical changes at cingulate cortex in AD continuum and their association with changes in empathic abilities
Proposed correlation between dysfunction at different regions of cingulate cortex and changes in empathic abilities through the AD continuum. ACC, anterior cingulate cortex; PCC, posterior cingulate cortex.
All the above has practical implications insofar as empathic abilities are an inherent part of the human condition. On the one hand, a subtle impairment of the cognitive empathy, such as that observed in the early preclinical stages of AD, entails socio-behavioral changes that, in turn, reflect changes in personality traits, and lead to more or less apparent limitations in adapting effectively to the social environment [90]. Such limitations would produce a consequent deterioration of social activity either by loss of initiative of the individual himself or by lack of interest of others in relating to a person who does not adjust to social norms. As a vicious circle, social isolation would increase the progression of cognitive impairment [91]. On the other hand, the progressive accumulation of Aβ that typically occurs from late MCI, produces a decline of self-empathy and a consequently loss of awareness of one’s own deficits and limitations in daily life [92]. The appearance of anosognosia worsens the prognosis of patients to the extent that they do not perceive the need to undergo pharmacological and non-pharmacological therapeutic interventions; in addition, anosognosia also has a negative impact on the main caregivers of these patients, increasing their levels of anxiety and stress and diminishing their quality of life [69].
In sum, a better understanding of the changes in empathy and self-empathy associated with AD, as well as its neuroanatomical basis, may help identify early individuals at risk of progression, improve and personalize interventions on patients for the purpose of modifying the course of the disease, and ultimately improve the quality of life of patients and their caregivers.
Footnotes
ACKNOWLEDGMENTS
We wish to thank warmly to the Carlos III Institute of Health and Queen Sofia Foundation for its sponsorship and continuous support. This study was funded by the Spanish Ministry of Science, Innovation and Universities –State Research Agency–under the Grant RTI2018-098762-A-C32 by the European Regional Development Fund. Complimentary, it was supported by the multidisciplinary Research Program of the Fundación General de la Universidad de Salamanca (FGUSAL) in the framework of the Centro Internacional sobre el Envejecimiento (CENIE) co-funded by the Interreg V-A Programme, Spain - Portugal, (POCTEP), 2014-2020 under the Grant 0348_CIE_6_E by the European Regional Development Fund.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
