Abstract
The ethylene bis-iminodiacetic acid (Synonym: ethylenediamine tetra-acetic acid) a polyaminocarboxylic acid derivative∗ is one of the most powerful deionizing agents for alkaline earth and metals. It is available as the free acid, and as di-, tri- and tetrasodium salt, under the trade name of Sequestrene. The structural formula of the free acid is as follows:
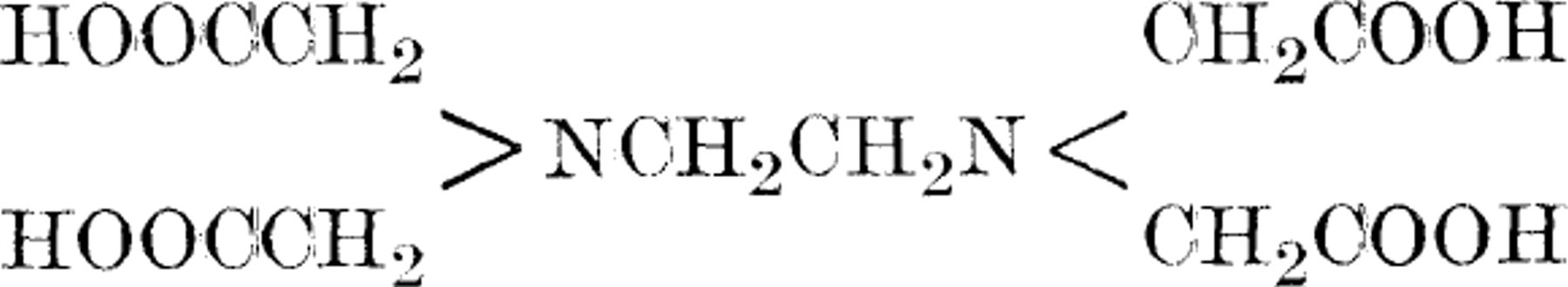
The free acid is only slightly soluble in water, while the sodium salts are readily soluble. The free acid and its salt do not hydrolyze in aqueous solution, and are stable and effective over a wide range of temperatures and pH.
Dyckerhoff( 1 ), et al., were the first to investigate the anti-coagulant effect of the di-sodium salts of Sequestrene. They found that one half to one mg completely inhibited the coagulation of human and animal blood, and that it was 10 times more effective than sodium citrate. The anti-coagulant effect is due to the binding of the blood calcium ion. It has no effect on blood clotting in vivo. The di-sodium Sequestrene has been used in our laboratory for routine blood work for over a year. Because of its powerful anticoagulant properties, only minimum quantities are necessary. It is less damaging to the cellular elements of the blood, and preserves them longer than sodium citrate and the oxalates. For routine blood examination, a 10% aqueous solution (maximum solubility) is preferable; 0.005-0.01 cc will keep 5 cc of blood from coagulating.
If blood is kept at 5°C; the red cell and leucocyte counts do not show any significant changes for from 3-5 days. If blood is collected in paraffmized containers, the platelet count remains unchanged for 24 hours; after this time a slight decrease is noted (6%).
Get full access to this article
View all access options for this article.
