Abstract
The assimilation of carbon dioxide by heterotrophic bacteria was first established by Wood and Werkman 1 with the propionic acid bacteria. Recently the stable isotope of carbon (C13) 2 and radioactive carbon (C11) 3 have been used as tracers of the carbon dioxide fixed in the fermentation of glycerol. The fixed carbon has been shown to be predominantly in the products propionic and succinic acids. Wood, et al., 2 demonstrated, in addition, that the fixed carbon in succinic acid is exclusively in the carboxyl group. These authors. suggested that all the carbon dioxide is fixed by the following reaction
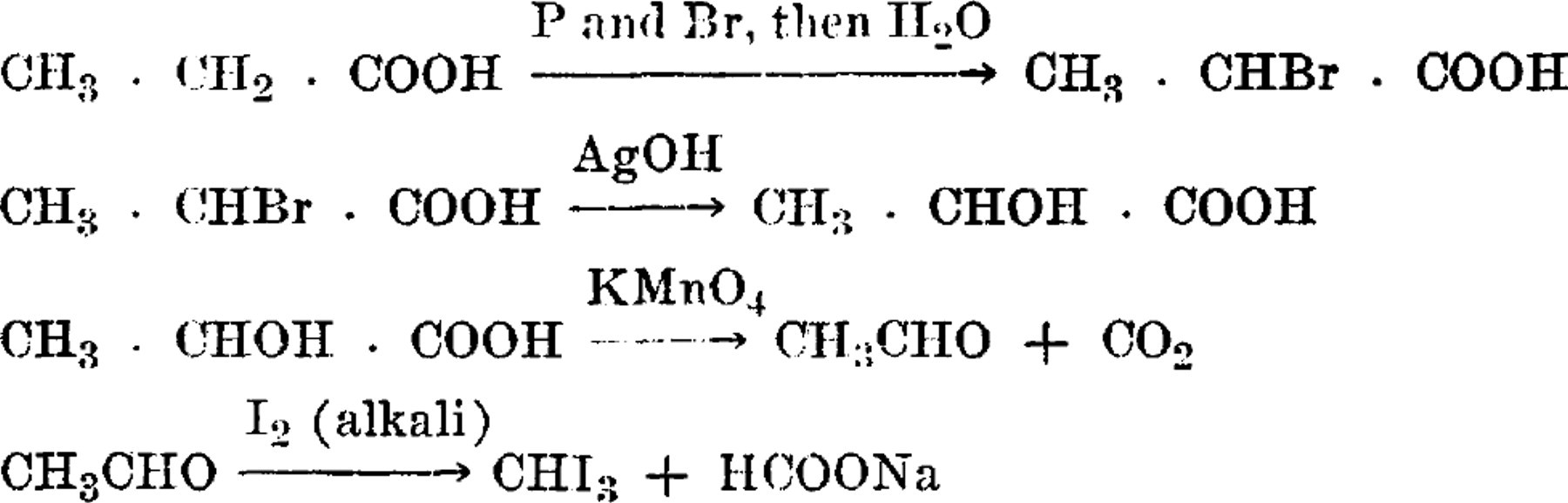
and that propionic acid containing fixed carbon is derived from the 4-carbon compounds. Accordingly, the fixed carbon of propionic acid would be in the carboxyl group. Therefore, the report by Carson, et al., 4 of fixed carbon occurring in the α- and β-carbons as well as in the carboxyl carbon of propionic acid is of particular interest. These results imply that, in addition to the mechanism shown by Wood, et al., 2 the propionic acid bacteria have another method of fixing carbon dioxide. The latter method might involve the synthesis of the entire chain from 1-carbon compounds, since the fixed carbon is suggested by Carson, et al., 4 to be distributed among the 3 carbons of propionic acid. This observation places in question the present schemes of fermentation, for the implication is that the substrate may first be broken clown to 1-carbon compounds and then synthesized to the final products of fermentation. While it is clear that autotrophic forms of life must perform such a synthesis, it does not seem necessary for heterotrophic forms of life to break down the substrate to a 1-carbon compound and then resynthesize it to final products. We have, therefore, reinvestigated the problem. Contrary to the report of Carson, et al., 4 we find the fixed carbon is exclusively in the carboxyl group of propionic acid.
Get full access to this article
View all access options for this article.
