Abstract
When NaOH is added to solutions of hemoglobin, a reaction is initiated whose progress may be followed very accurately by means of spectrophotometry. The changes which occur may, for convenience, be divided into 2 successive main reactions, and may be represented as follows:
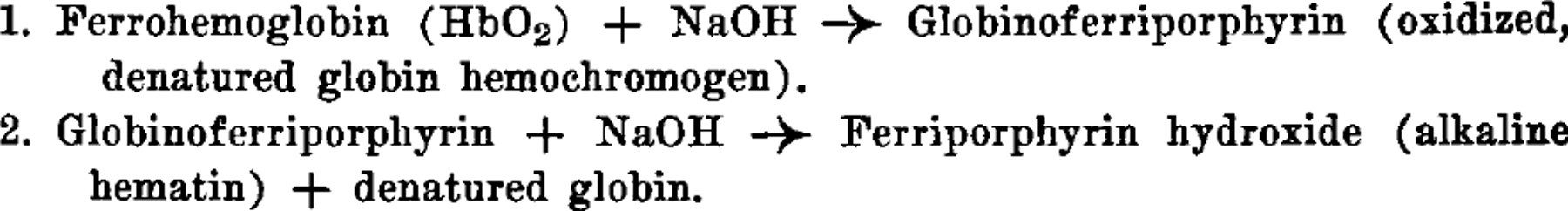
Under the conditions which have been used the reactions are unidirectional, but the addition of a reducing agent, Na2S2O4, shifts reaction 2 in favor of the hemochromogen, as follows:
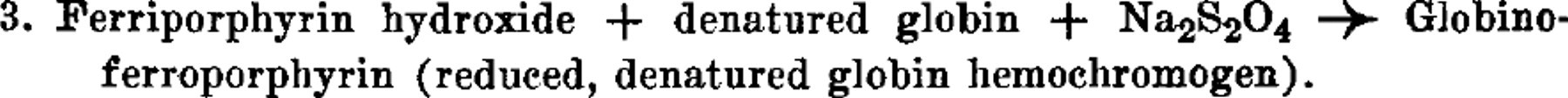
When Na2S2O4 is added to globinoferriporphyrin (reaction 1), globinoferroporphyrin also is produced. The terminology which is employed is unambiguous, and has been proposed by the writer. 1 The protein component of the hemoglobin is probably irreversibly denatured in this process. It has already been pointed out by Drabkin and Austin 2 that the first reaction takes time and is the basis for the von Krüger reaction. 3
It has been found by the writer that essentially similar reactions to the above occur when hemoglobin is denatured by reagents other than alkali—notably urea and acetamide in high molar concentrations (4 to 6 M), and HCl. In the case of the latter reagent, denaturation results in the production of the familiar acid hematin and even with fairly strong acid (0.1M) it is well recognized that the reaction takes time. During the course of this reaction the writer has found that, if enough alkali is added to just dissolve the material precipitated at the iso-electric point as the acidified solution is neutralized, the presence of the typical globinoferroporphyrin (the reduced hemochromogen) is disclosed upon the addition of Na2S2O4.
In studying denaturation reaction rates conditions were chosen to provide for a very slow reaction with alkali. The concentration of hemoglobin (HbO2,) in the final solution was 0.1 mM per liter (where 1 mole is equivalent to 1 mole of iron porphyrin), and the concentration of total alkali in the final solution was 0.008M.
Get full access to this article
View all access options for this article.
