Abstract
According to the electrode equation,
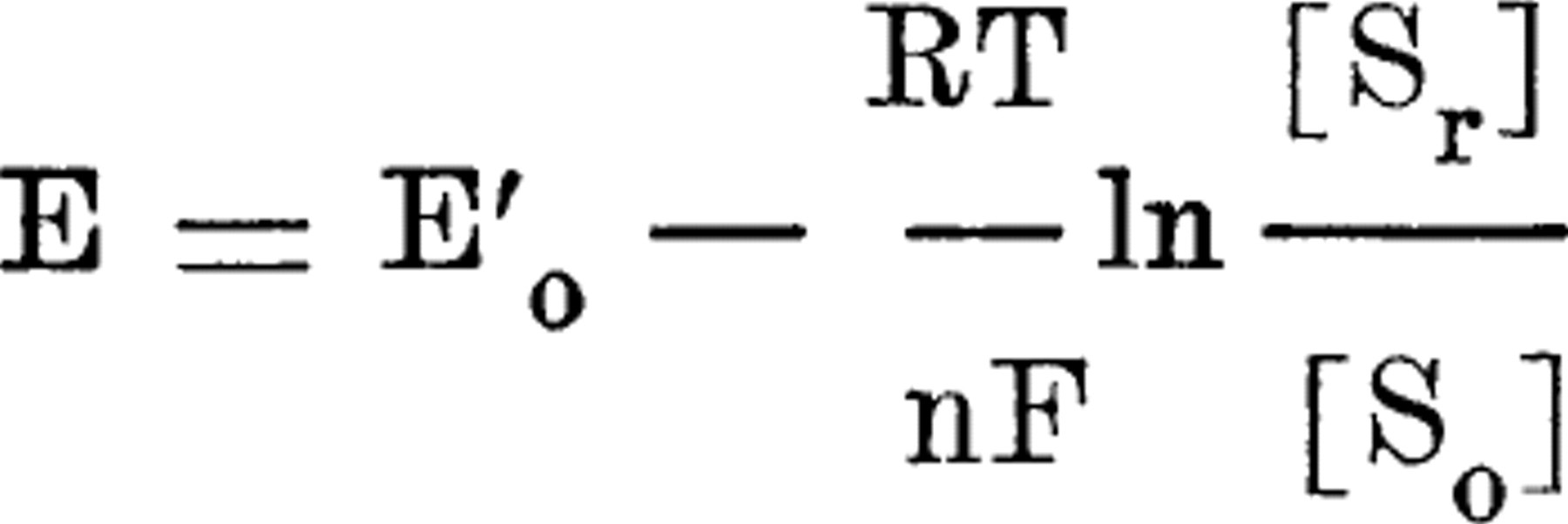
the oxidation-reduction potential, E'o, of a given oxidation-reduction system at a given pH can be calculated from the potential observed, E, when a blank noble metal electrode is placed in a mixture in which the ratio of the concentration of the total reductant, [Sr], to that of the total oxidant, [So], is known. For the benzoquinone-hydroquinone system, the equation becomes
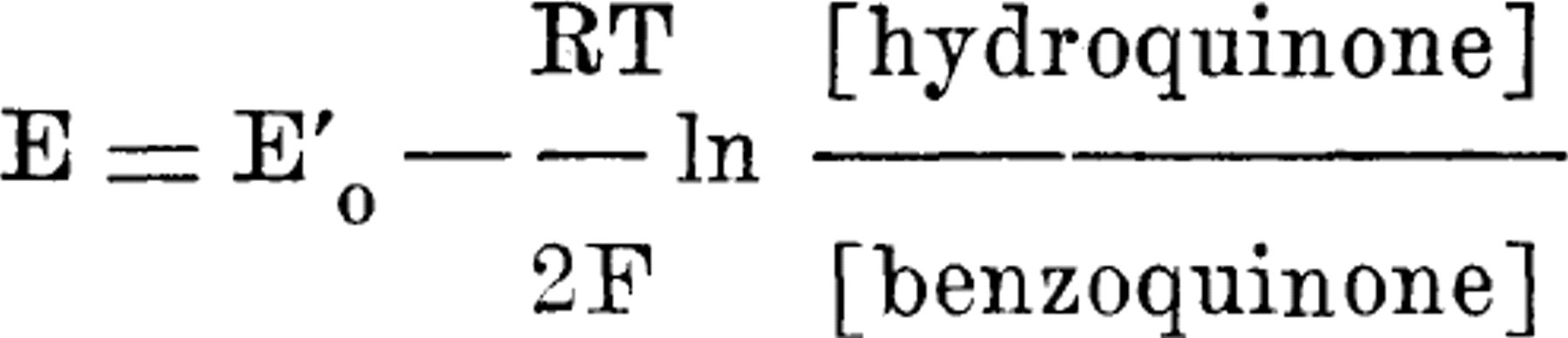
Theoretically the potential is independent of the total concentration of the two components of the system and independent of the size of the electrode.
The mechanism through which the electrode responds to the composition of the mixture is not clear, but in a homogeneous solution the number of the reductant particles acting upon any unit area of the electrode must bear to the number of the oxidant particles acting, the same ratio as that in the solution. Slight difference in the uniformity of the electrode surface should make no difference in the electrode behavior. Assuming the surfaces of the electrode on both sides to be identical, we should obtain the same potential, if we could separate the 2 species of particles and place only pure species on each side of the electrode. Similarly if we could split one electrode into 2 identical halves and immerse them separately in 2 solutions, one containing the pure reductant and the other, the pure oxidant, the potential observed should not differ from that observed when the original electrode is placed in the solution made by mixing the 2 pure solutions.
In the present experiment, this hypothesis was tested using the benzoquinone-hydroquinone system. Instead of splitting one electrode into 2 identical halves, 2 electrodes as nearly identical as possible were prepared. One of them was placed in a solution of benzoquinone of known concentration while the other was placed in a solution of hydroquinone of the same or different concentration. Both solutions were prepared with the same buffer mixture.
Get full access to this article
View all access options for this article.
