Abstract
The direct titration of the arsenic in arsphenamine with a standard iodine solution was first used by Gaebel, 1 who also found that the reaction was a reversible one, namely
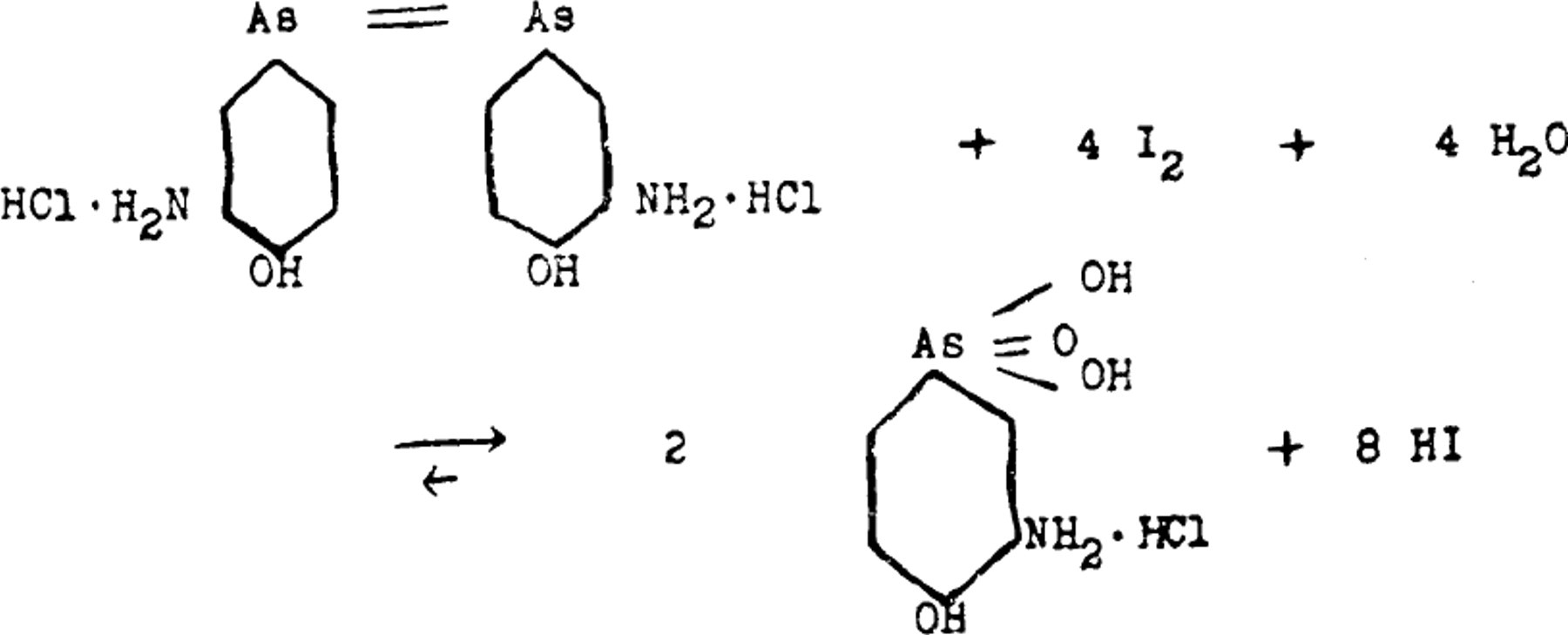
The titration of inorganic arsenious compounds with iodine is also a reversible reaction, but may be carried to completion, as shown by Washburn and Strachan, 2 by maintaining the pH of the reaction mixture between 9 and 4. Gaebel found, however, that this was not the case with the reaction between arsphenamine and iodine.
Myers and Du Mez 3 from a comparative study of several methods of quantitative determination of the arsenic in arsphenamine, rejected Gaebel's titration method because of the low results obtained. Nevertheless, the direct titration of both arsphenamine and neoarsphenamine with iodine has been employed by a number of workers, most of whom have not taken into account the fact that the reaction does not go to completion. The method has been used quite extensively, for instance, for the simultaneous oxidation of arsenic and sulphur in the differential analysis of the composition of neoarsphenamine and sulpharsphenamine. The accuracy of the conclusions drawn from such simultaneous analyses, particularly in regard to the sulphur determinations, depends, of course, upon the absolute accuracy of the values assigned to the arsenic oxidation.
Macallum, 4 quoting 3 analyses of a single sample of arsphenamine, with a 3.5% titration error between samples, and assuming 31.3% arsenic content for the arsphenamine (anhydrous arsphenamine contains 34.2% arsenic; with 1 mol. of CH3OH 31.85%; with 2 mols. of H2O 31.57%) derived the relationship of 7.755 mols. (theoretical 8 mols.) of iodine for the titration of arsphenamine with iodine. Elvove, 5 in making differential analyses of the composition of samples of neoarsphenamine and sulpharsphenamine, employed the factor thus derived by Macallum.
It was felt, therefore, that the quantitative determination of sulphur fractions in neoarsphenamine and sulpharsphenamine by the simultaneous oxidation of arsenic and sulphur were of doubtful value, unless it could be shown that the reaction between the iodine and arsenic attained a constant point of equilibrium under the conditions of the titration, and the equilibrium point of the reaction was accurately determined.
Get full access to this article
View all access options for this article.
