Abstract
The fact that the addition of sodium citrate to calcium containing solutions reduces the concentration of ionic calcium is well known. The nature of the combination between calcium and citrate has not been definitely established. It has been sometimes thought that a neutral compound Ca3(citrate)2, exists, but this is not consistent with the evidence that the combination between calcium and citrate is negatively charged. 1
For this reason the frog heart preparation which had been shown to be sensitive to calcium ion changes under controlled conditions was used to assay the calcium ion concentration in solutions of varying total calcium and citrate content.
The results of such experiments are shown in the accompanying figure. The experimental points show the concentrations of calcium and citrate present in solutions which are iso-active with corresponding solutions containing no citrate, but containing a certain concentration of calcium ions. Each series of symbols represents physiologically isoactive points. The relationship for a given concentration of calcium ions appears to be linear for the limits within which this technique is accurate, i. e., calcium ion concentrations from 0.3 to 1.2 millimols per liter. This has been found to be true when the total calcium concentration is as high as 30 millimols per liter.
These results are consistent with the interpretation that (1) calcium combined with citrate does not affect the amplitude of contraction of the frog heart and (2) that one mol of calcium combines with one mol of citrate to form a calcium citrate compound carrying one negative charge. The straight lines in the figure fit the equation,
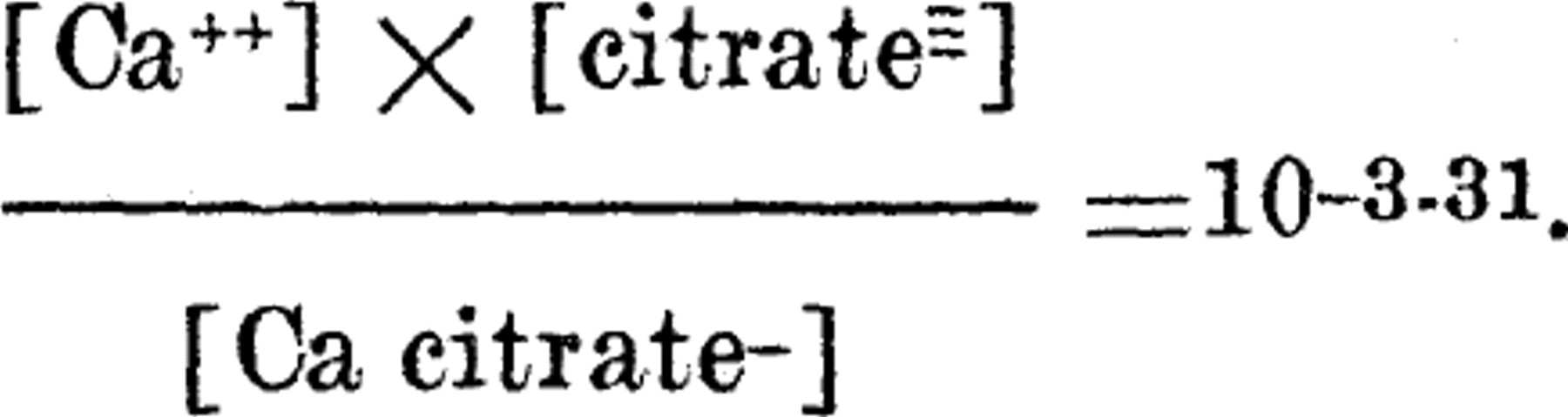
The same problem has also been studied chemically by equilibrating salt solutions containing citrate with calcium carbonate and calculating the calcium ions from the solubility product constant of the solid phase.
Get full access to this article
View all access options for this article.
