Abstract
As a solution is cooled in a freezing-point determination, crystallizing does not start at the freezing temperature. The temperature falls below the freezing-point, to an extent varying in different cases, before the freezing process commences. As soon as ice does start to form, the temperature climbs rapidly toward the freezing-point. The heat for this rise of temperature is derived from the freezing process, the latent heat of fusion of ice being 80 calories. The formation of ice is equivalent to the removal of water, and hence the solution will be concentrated. The freezing-point observed will, therefore, represent a solution more concentrated than the original solution; or, to express the fact differently, the freezing-point depression observed will be greater than the true depression for the solution.
The solution, if the amount of solid present is so small that it can be neglected, will be concentrated s u/1 times for u degrees of undercooling (Jones
1
), where s equals the specific heat of the solution and 1 the latent heat of fusion. Thus the true freezing-point, Δ, will equal the observed freezing-point, Δ
1
, minus 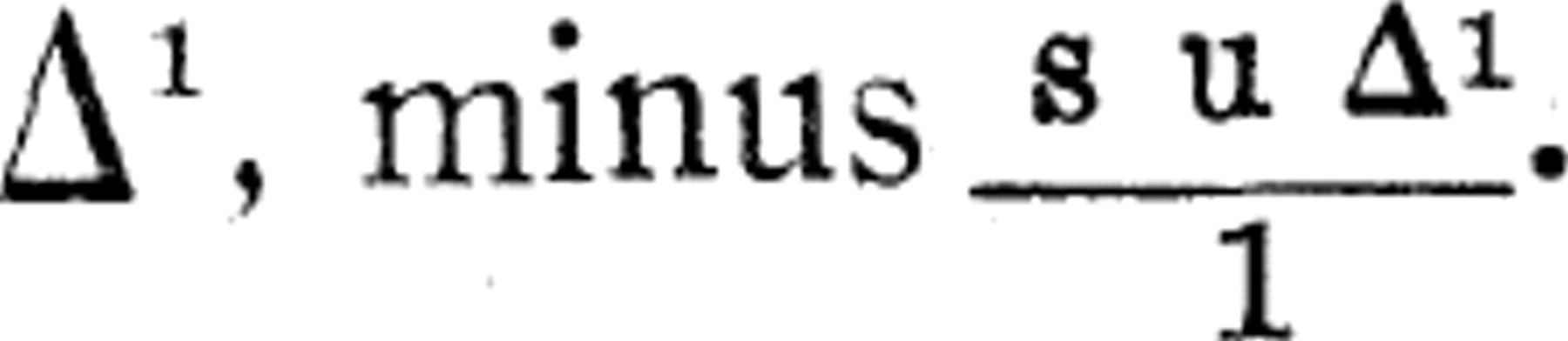 .
.
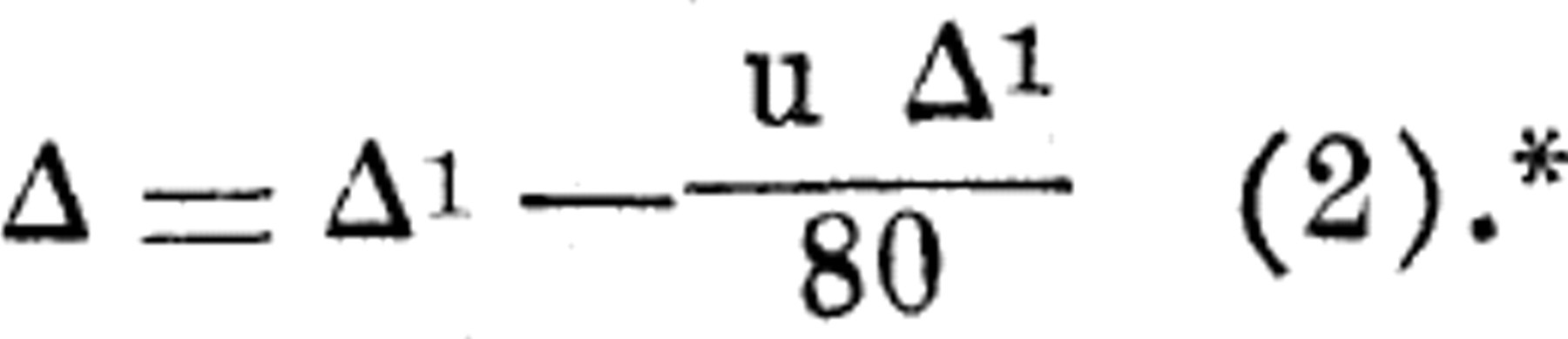
Assuming the specific heat to be unity and 1 to be 80,
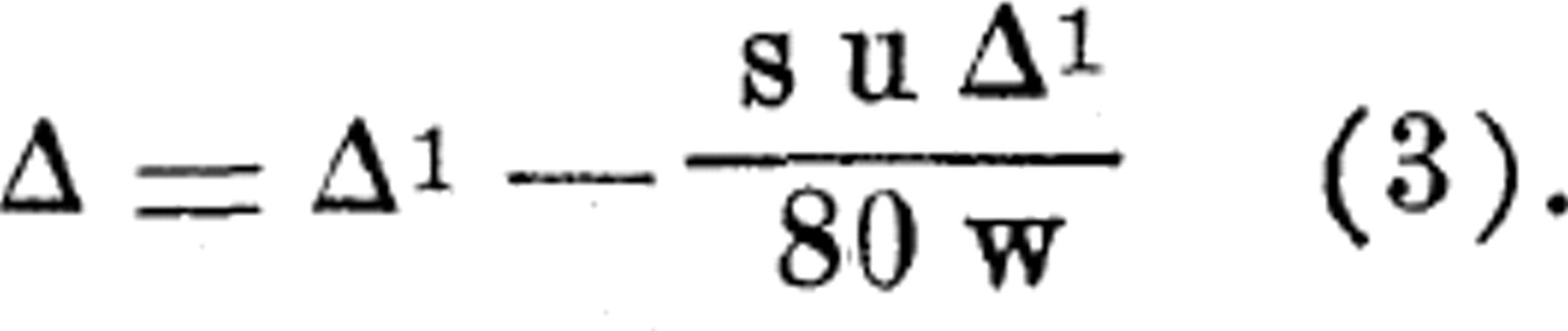
However, if there is a large percentage of solid present in the material, then, for the same amount of undercooling, there will be a greater concentration in such material than in a solution containing only a negligible amount of solid. The same quantity of ice must be formed in each case (assuming the specific heats to be the same), and hence the concentration will be greater in the material of high solid content. If the solids are considered, the correction equation becomes
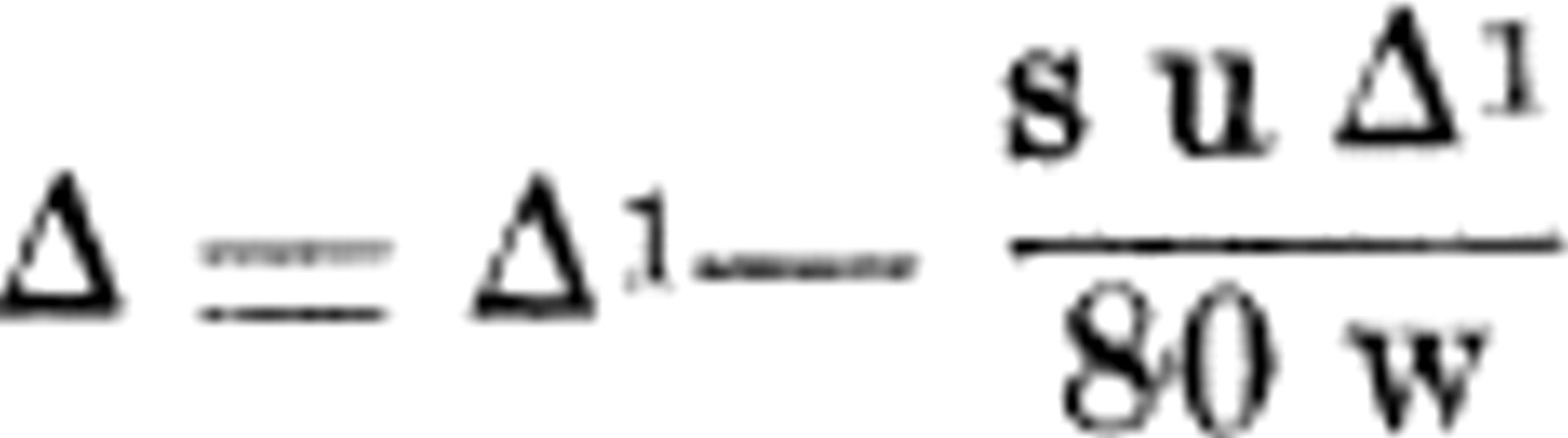
The term, s, is the specific heat of the material, and w represents the fraction by weight of water acting as a solvent (more exactly, this water plus the soluble constituents dissolved in it).
Get full access to this article
View all access options for this article.
