Abstract
Quantitative studies of the mechanism of calcification require a knowledge of the physico-chemical constants of the blood equilibria involved in the process. The tertiary dissociation constant of phosphoric acid, K3, thus far available seems incompatible with the apparent dissociation constant pK3 from the dissociation curve for the phosphates which is of an order of 1 x 1012.
The value of K3 was determined experimentally from a study of aqueous systems of the tertiary sodium and potassium phosphates at equilibrium, from the standpoint of hydrolysis. The value thus obtained was correlated with calculations made from the data obtained by the electrometric titration of phosphoric acid.
The tertiary phosphates hydrolyze to a great extent, the secondary and primary salts inappreciably. In the mechanism of the first stage hydrolysis of the tertiary salts, the essential equilibrium, may be expressed ionically:
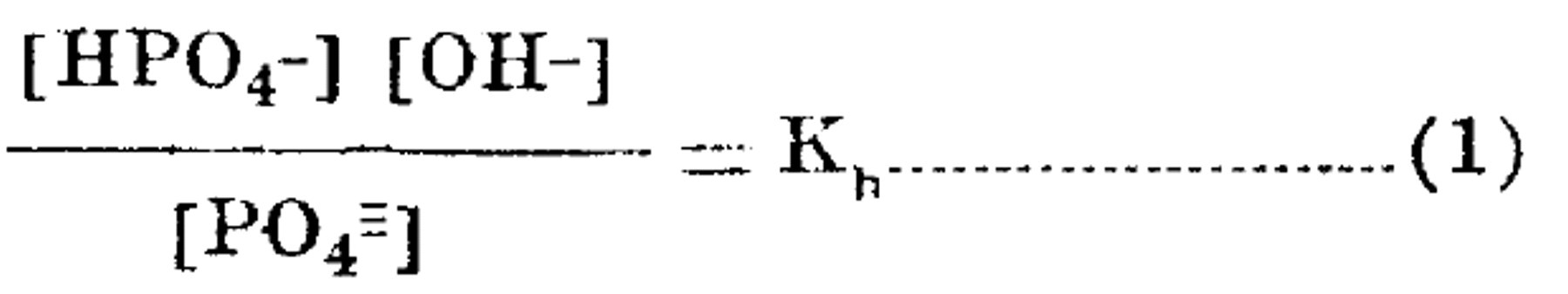
Where the brackets include the ion activities, and Kh is the activity hydrolysis constant.
Get full access to this article
View all access options for this article.
