Abstract
A summary is given of work being reported in detail in U. S. Public Health Reports. The equilibrium potentials at 30° of the indophenol system of oxidation-reduction indicators are precisely formulated by the equation:
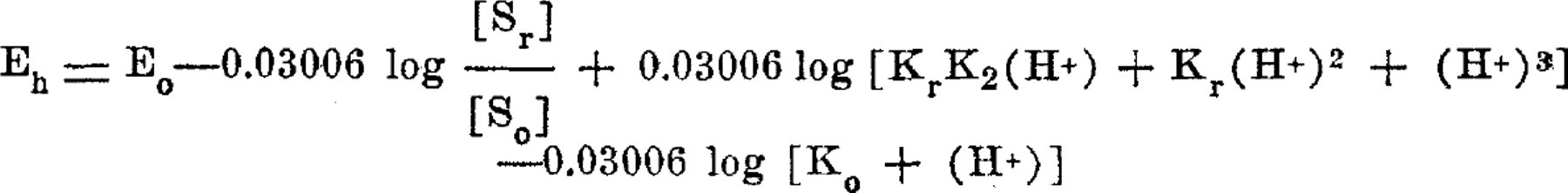
The potentials are determined not only by the proportions of oxidant and reductant but also by the hydrion concentration. The indophenol system lies at a more positive (more oxidizing) range than any other system of indicators studied by us up to this time. Substitutions in the indophenol molecule of halogen or alkyl groups produce rather small changes in the oxidation-reduction potential and relatively more important changes in the acid-base properties of the compounds. The latter permits selection of compounds compatible with physiological ranges of pH, and possibly useful for indicating certain intensities of oxidation-reduction in biological processes.
One such compound is phenol indo-2,6-dibromo phenol, whose acid dissociation constant (pKo = 5.7) is at a point where the dye retains its deep blue color in mildly acid solutions. Many biological fluids reduce this compound in the presence of air.
Get full access to this article
View all access options for this article.
