Abstract
The effects of various salts on the coagulation time of milk by rennin have been studied. It was found that CaC12, MgSO3, and NaC1 in certain dilute solutions favor coagulation (shortening the coagulation time), in other more concentrated solutions counteract it.
The coagulation of milk by rennin is sometimes spoken of as a precipitation of the hypothetical caseinogen by calcium salts. That it is not a true precipitation reaction is shown by the above mentioned action of salts. These to the same extent as they favor the stability of the milk colloids also favor coagulation, and vice versa; in other words, to the extent salts increase or decrease the surface charges of the colloids they act as coagulants or anti-coagulants. The effect of salts on the surface charge is expressed by the well-known equation:
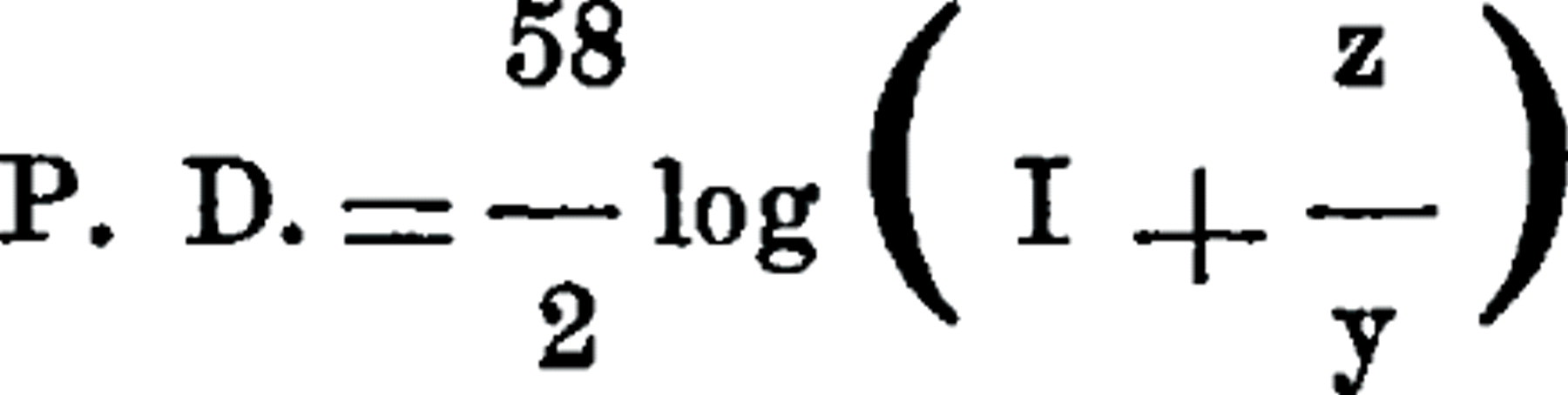
where z stands for ions in combination with the colloids, y for the same ions in the medium.
A condition under which an increase in the surface charges would favor the cementing together of the colloids may occur if colloids with the opposite surface charge make their appearance, in this case colloids which in Loeb's terminology can be written RNH2 COOH. Some calcium compounds are of that nature because they have their isoelectric point far toward the alkaline side. Calcium caseinate has, according to Loeb, its isoelectric point at pH 10.53.
Get full access to this article
View all access options for this article.
