Abstract
It has been noted by Howell, Collip, Dale and Evans, the author and others that the intravenous injection of sodium bicarbonate may act as either a respiratory or circulatory stimulant, eliciting hypernea or a marked rise in the blood pressure. Such injection obviously increases the hydrogen ion concentration of the blood and inasmuch as it produces a slight dilution, it decreases the amount of carbon dioxide in the blood eliciting the stimulation. The increased respiration is, therefore contrary to the usually accepted laws of respiration. The only apparent change in the blood which might elicit stimulation is the greatly increased number of HCO3 ions. Collip, therefore, suggests that the HCO3 ion exerts a specific stimulating action on the respiratory center.
We believe, however, that this anomalous result may be otherwise explained. When the carbon dioxide is dissolved in water it exists primarily in three forms—dissolved CO2 molecules, dissolved undissociated H2CO3—molecules and dissociated H2CO3—thus 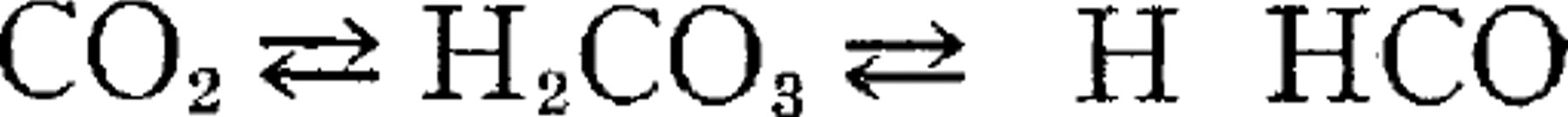 . Addition of sodium bicarbonate to the solution pushes the reaction to the left increasing both the dissolved CO2 and the undissociated H2CO3. If we accept the view that the dissolved CO2, and the undissociated H2CO3, diffuse freely into cells while the ions as such do not penetrate to any appreciable extent it is apparent that the injection of sodium bicarbonate increases the freely diffusable forms of carbon dioxide at the expense of the poorly diffusible ions and in that way increases the acid effects of the blood, at least on the interior of the cells (and possibly in the lymph bathing the cells as will be discussed in a later paper) even though the actual sum total of the original dissolved carbon dioxide in its various forms is not increased and the hydrogen ion concentrate of the blood is actually decreased.
. Addition of sodium bicarbonate to the solution pushes the reaction to the left increasing both the dissolved CO2 and the undissociated H2CO3. If we accept the view that the dissolved CO2, and the undissociated H2CO3, diffuse freely into cells while the ions as such do not penetrate to any appreciable extent it is apparent that the injection of sodium bicarbonate increases the freely diffusable forms of carbon dioxide at the expense of the poorly diffusible ions and in that way increases the acid effects of the blood, at least on the interior of the cells (and possibly in the lymph bathing the cells as will be discussed in a later paper) even though the actual sum total of the original dissolved carbon dioxide in its various forms is not increased and the hydrogen ion concentrate of the blood is actually decreased.
Get full access to this article
View all access options for this article.
