Abstract
Conclusions
The above data appear to indicate that gelatin gels have a structure and that this structure is more or less fixed at the time that gelation takes place. It would appear that the gelatin aggregates or micelles are more and more interlaced at increasing concentrations of gelatin. This structure is apparently not appreciably altered by drying at a temperature below the “melting” point of the gel. The fact that drying does not markedly influence the gel structure appears to argue against the formation of micelles by the adventitious coming in contact of dispersed particles of gelatin, for certainly many gelatin particles must touch each other in the dried sheet, but apparently they do not cohere to each other with any appreciable force, certainly not with a force at all comparable with the force of coherence between particles or micelles originating at the time of gelation. This 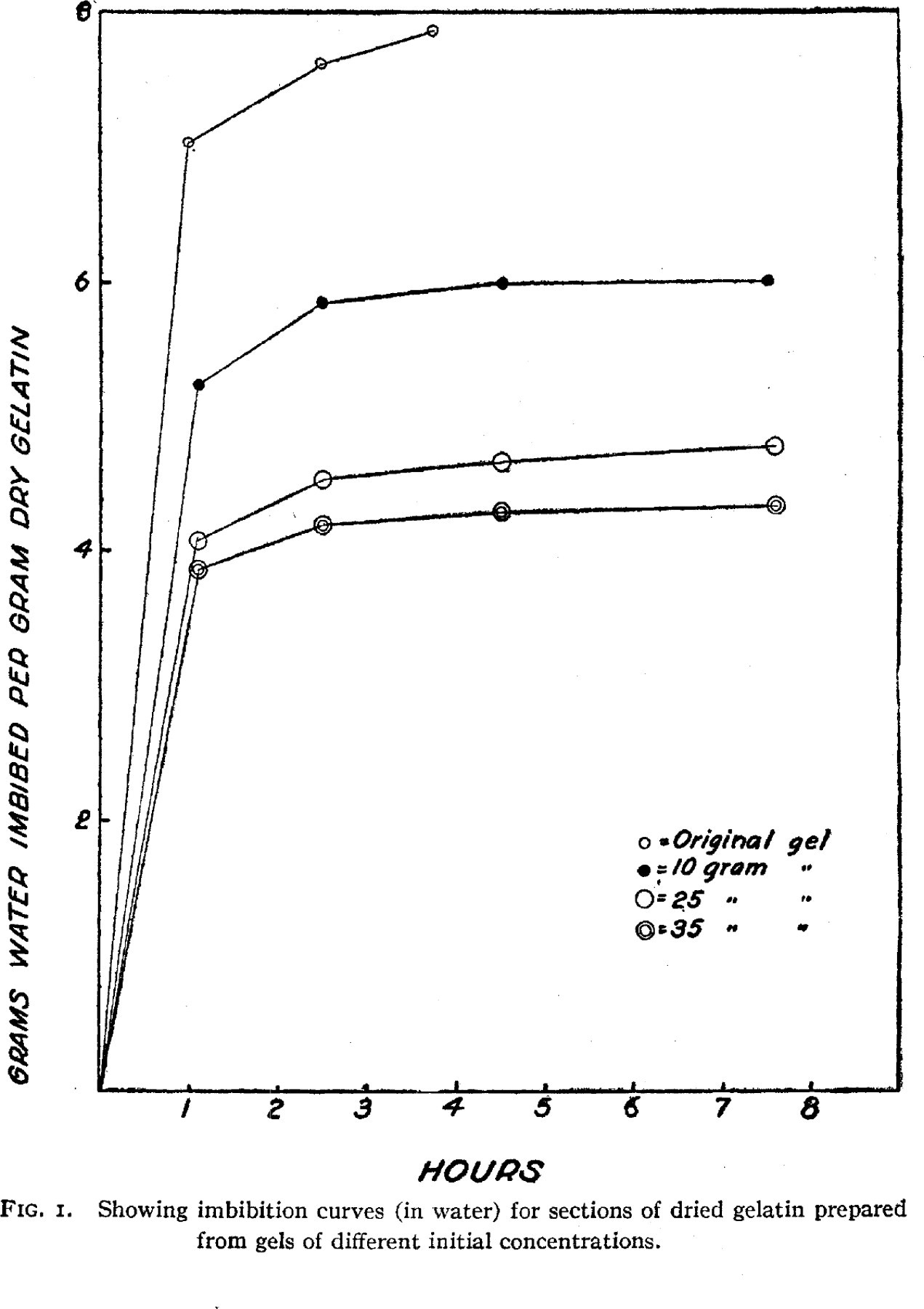 might be explained by a crystal structure where the crystals melt or soften at the gelation temperature. The micelles would then be formed by the solidification of crystals and later when micelle touched micelle the hardened surface of the crystal would prevent cohesion. We have recently shown' that a crystal gel may be very dilute and yet possess considerable rigidity so that a crystal structure is not incompatible with the properties of gelatin gels.
might be explained by a crystal structure where the crystals melt or soften at the gelation temperature. The micelles would then be formed by the solidification of crystals and later when micelle touched micelle the hardened surface of the crystal would prevent cohesion. We have recently shown' that a crystal gel may be very dilute and yet possess considerable rigidity so that a crystal structure is not incompatible with the properties of gelatin gels.
These experiments likewise show the marked influence that hysteresis may have on experiments where gelatin is involved. This is particularly noticeable in Fig. 4 where uniform-sized particles prepared from gels of different concentration were used. Inasmuch as a dried sheet of gelatin apparently reflects the structure of the original gel from which it was prepared, the production of differerit samples of dry gelatin possessing uniform physicochemical properties would appear to be extremely difficult.
Get full access to this article
View all access options for this article.
