Abstract
The dilution method for determining lung volume, invented by Davy and modified by Bohr and his coworkers, and recently by Lundsgaard and Van Slyke, rests on the principle of mixing the air in the lungs with a known volume of foreign gas (H2 or O2), and calculating the air content of the lungs from the extent to which either the foreign gas or the nitrogen of the lung air (Lundsgaard and Van Slyke) is diluted. This method yields satisfactory results when the subject can breathe deeply, so that 4 or 5 respirations cause complete mixture of the lung air with the diluting gas. When the subject, however, because of weakness or respiratory disturbance, cannot greatly increase the depth of his respirations, so much time is required for complete mixture that volume changes due chiefly to absorption of oxygen make accurate results unobtainable.
We have been able to avoid this difficulty by modifying the principle, and basing the calculation not upon the dilution of one gas, but upon the volume ratio of two gases. The subject breathes to and from a bag containing oxygen, sufficient to satisfy his requirements for the length of the experiment, and mixed with the oxygen a known volume of hydrogen approximating the volume of the nitrogen in the lungs. The CO2 is removed by a scrubber of sodium hydroxide shells. After mixture is complete, the N2 and H2 in the gas are determined. Since 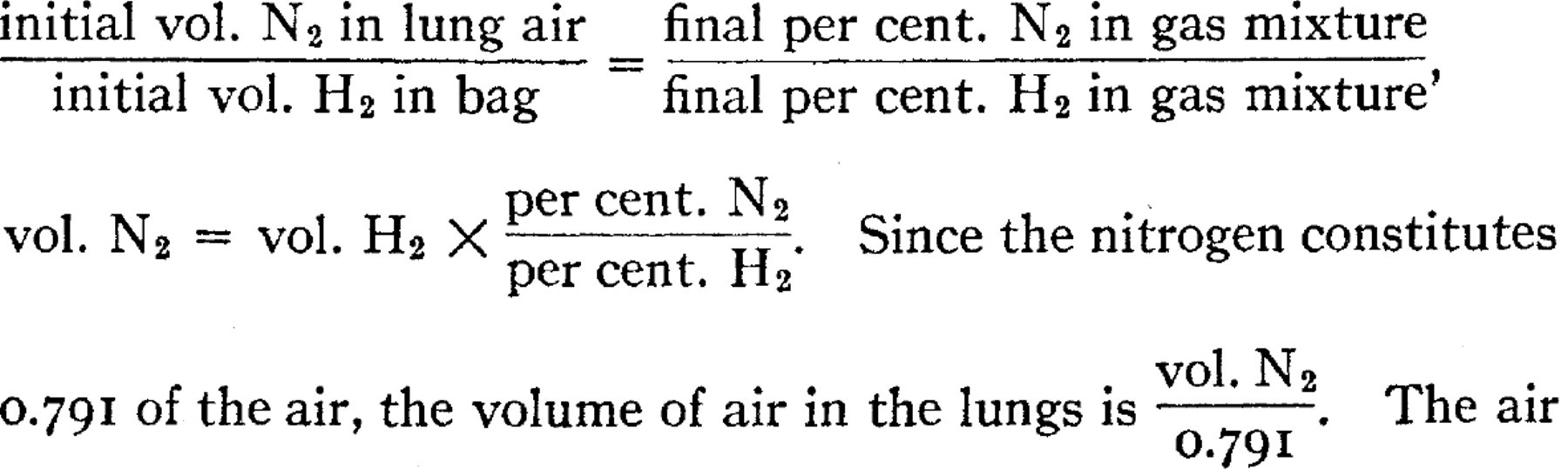 volume thus determined is corrected by subtracting air present in the dead space of the apparatus at the start, also any air present as impurity in the oxygen or hydrogen used. Changes in volume of oxygen or CO2 in the system do not affect the results, and absorption of hydrogen is negligible.
volume thus determined is corrected by subtracting air present in the dead space of the apparatus at the start, also any air present as impurity in the oxygen or hydrogen used. Changes in volume of oxygen or CO2 in the system do not affect the results, and absorption of hydrogen is negligible.
Get full access to this article
View all access options for this article.
