Abstract
This communication was confined to a report on studies of (a) flavones, a group of yellow pigments, characterized by the production, in their solutions, of intense yellowish-brown color on the addition of ammonia, and of (b) anthocyanins, a group of red, violet, or blue pigments, which, in solution, change to bluishgreen on the addition of alkali, and pink on the addition of acid.
These pigments were obtained from tulips: flavone, from “La Reine”; anthocyanin, from “Crimson King.” Both varieties of flowers were collected at the N. Y. Botanical Garden through the courtesy of Dr. A. B. Stout.
The chemical relationship of flavones and anthocyanins.—Wheldale and others believe that flavone is convertible into anthocyanin by oxidation. The Armstrongs regard this conversion as due to processes of oxidation and reduction. Combes and Willstätter consider that reduction alone effects the change. The results of our own experiments accord with the view of the latter investigators. We find that active (“nascent”) hydrogen reduces flavone to anthocyanin. The latter can be further reduced to a leuco-base, which in turn, by exposure to air or, more rapidly, by addition of an oxidizing agent, is reconverted into anthocyanin, thus possibly explaining the facts observed by the Armstrongs:
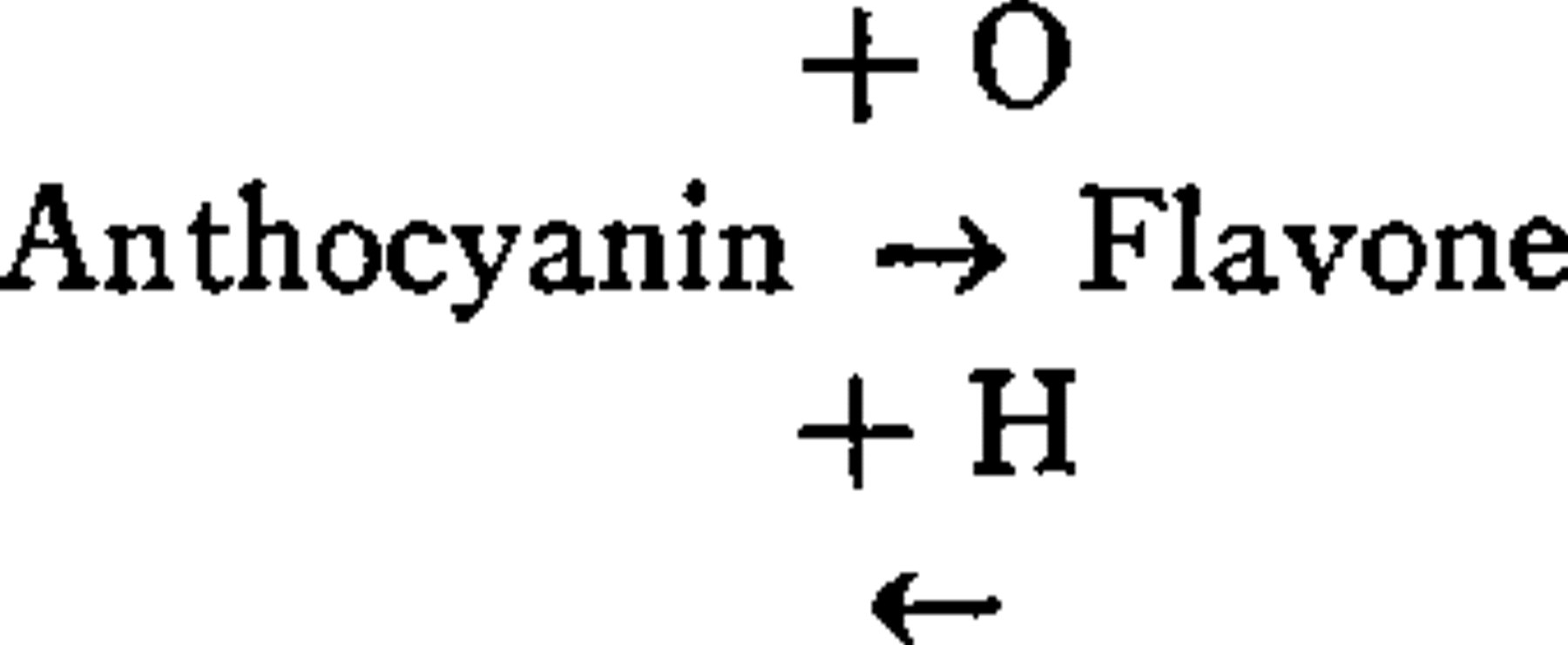
We have also succeeded in oxidizing anthocyanin to flavone. This was done most effectively by first isolating anthocyanin in a fairly pure condition, by Willstatter's method. The only oxidizing agent we have been able to use with success thus far is hydrogen peroxide. Oxidase from potato, as well as oxidase from Crimson King tulip, could not be used as a substitute for hydrogen peroxide.
Flavone thus obtained from anthocyanin can be reduced back again to anthocyanin:
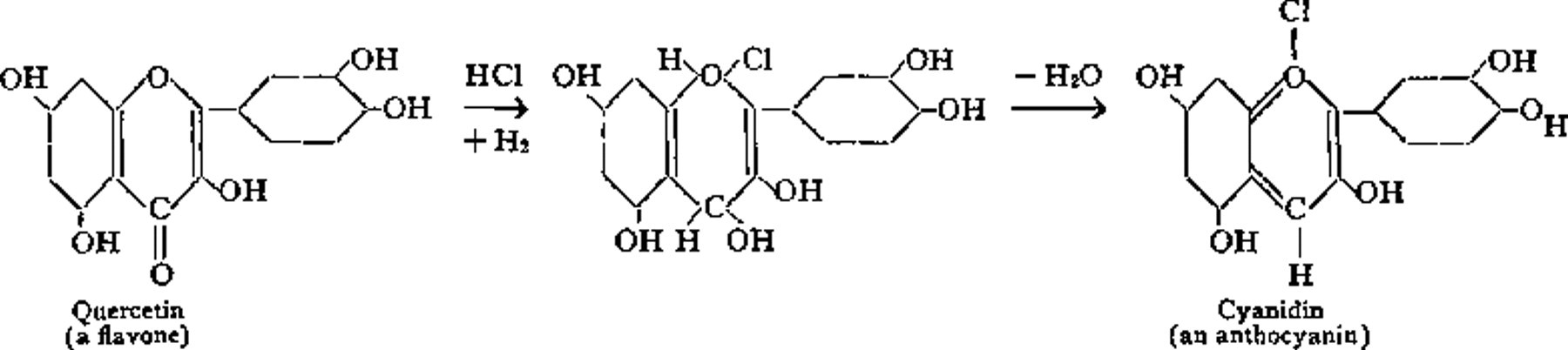
The chemical relationship between flavones and anthocyanins may be expressed thus (Willstatter):
Get full access to this article
View all access options for this article.
