Abstract
The remarkable observation made by Dakin and Dudley
2
that “racemized” casein is not attacked by pepsin, trypsin, or erepsin, is apparently excreted unchanged when injected subcutaneously or given by mouth to a dog, and is unaffected by bacteria, would indicate that the chemical structure of casein had been decidedly altered during the process of “racemization.” Dakin
3
suggests that the process is a tautomeric change of the keto-enol type in the manner 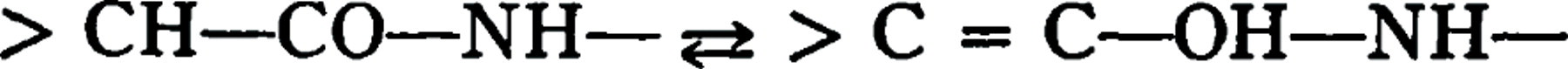 and that in the case of “racemized” casein this change must be complete for all the groups in the protein molecule, otherwise a point of attack for enzymes would be afforded and a partial splitting might occur. If the ketol-enol conversion be complete, the apparent discrepancy pointed out by Kober that all racemic peptides are attacked by erepsin would not necessarily be a real one, since the tautomeric change suggested by Dakin is not racemization in the ordinary meaning of the term. The discrepancy as to the number of end amino groups in casein, as indicated by the optically active amino acids found by Dakin, is as Kober
1
shows, fatal to Dakin's theory.
and that in the case of “racemized” casein this change must be complete for all the groups in the protein molecule, otherwise a point of attack for enzymes would be afforded and a partial splitting might occur. If the ketol-enol conversion be complete, the apparent discrepancy pointed out by Kober that all racemic peptides are attacked by erepsin would not necessarily be a real one, since the tautomeric change suggested by Dakin is not racemization in the ordinary meaning of the term. The discrepancy as to the number of end amino groups in casein, as indicated by the optically active amino acids found by Dakin, is as Kober
1
shows, fatal to Dakin's theory.
Anticipating the probability that a substance which is so changed as to be unaffected by enzymes and bacteria might also be changed in its antigenic properties, Ten Broeck, 2 at Dakin's suggestion, carried out experiments with “ racemized ” egg albumin and found that the substance was non-antigenic as shown by the anaphylaxis, precipitin and fixation tests. Underhill and Hendrix, 3 on the basis of fall of blood pressure and retardation of blood clotting, find that “racemized ” casein, egg-albumin and zein are non-toxic, but yield toxic products on partial hydrolysis with acid. The “racemic” proteoses, with the curious exception of zeose, are likewise toxic.
Get full access to this article
View all access options for this article.
