Abstract
Schizophrenia is usually characterized by a lack of insight into the illness and the need for treatment. This often results in poor adherence to medication, a situation exacerbated by the range of unpleasant side effects of antipsychotic drugs [1]. Currently there are no measures to identify those who will respond to treatment. Developing such measures would help to rationalize treatment and improve compliance. Pharmacogenomic approaches have not yet yielded clinically useful measures, and are also potentially expensive and time consuming. The investigation of physiological indices related to drug response that are simple to measure in the clinical setting is therefore a priority, particularly when they may also be measured in laboratory animals, in which relevant biological, pharmacological and genetic studies may be made.
An approach that has been investigated in animal studies is the measurement of thermoregulatory cutaneous blood flow, i.e. vasoconstriction of cutaneous vascular beds regulating heat loss to the environment, a process mediated by the sympathetic nervous system. Rat and rabbit animal models demonstrate that a number of atypical antipsychotics, including clozapine, potently inhibit vasoconstriction of the thermoregulatory cutaneous bed [2–8]. This sympathoinhibitory effect is mediated by central nervous system neurotransmitter receptor interactions similar to those thought to underlie the therapeutic effects of antipsychotic medications, including blockade of 5-HT2A receptors and stimulation of 5-HT1A receptors. Sympathetic outflow to brown adipose tissue (BAT) is controlled by similar brain neurotransmitter mechanisms and BAT metabolism has major effects on thermoregulatory heat production and basal metabolic rate [9,10]. Thus, animal results suggest that antipsychotics could have an acute, robust inhibitory effect on resting thermoregulatory sympathetic outflow in humans. This effect may potentially be related to the therapeutic effects of these agents, and may also have consequences upon basal metabolic rate.
In humans, sympathetic outflow to the cutaneous vascular bed in the hand is determined by thermoregulatory requirements, and is also acutely subject to affective disturbance [11], hence we go ‘pale with fright’. Previous clinical observations support the idea that increased cutaneous vasoconstriction may be a feature of schizophrenia. Kraeplin and Bleuler described cyanosis of the hands and feet in patients with schizophrenia [12,13], and subsequent investigators confirmed significantly lower finger temperatures in these patients, compared with controls [14,15]. In addition, a related measure of sympathetic outflow, namely electrodermal activity, is tonically elevated in the majority of schizophrenia patients, and correlates with symptomatology [16,17]. Furthermore, this abnormality is normalized by both atypical and typical antipsychotics [18].
We therefore aimed to determine whether atypical antipsychotics might acutely increase cutaneous hand blood flow in patients, just as they do in the corresponding thermoregulatory cutaneous vascular beds in experimental animals. Cutaneous blood flow is a major determinant of hand temperature [19] and infrared thermography is a robust, non-invasive and reliable means of measurement. In the present study, an infrared camera was used to measure hand temperature in patients with schizophrenia or schizoaffective disorder. We tested the hypothesis that atypical antipsychotics, by inhibiting cutaneous sympathetic outflow, increase blood flow to the hand. We investigated the acute effects of clozapine and clozapine-like antipsychotics on hand temperature, and determined whether this effect correlates with disease symptomatology.
Methods
Setting
Patients involved in this study were admitted to the Adult Mental Health Rehabilitation Unit (AMHRU) at Sunshine Hospital, a tertiary referral service that treats patients with severe psychotic disorders that have proved resistant to treatment. Many of these patients are treated with the antipsychotic medication clozapine, a drug with unique clinical efficacy in patients with resistant schizophrenia [20]. Inclusion criteria were subjects aged 18–64 with major psychotic disorders of at least 2 years duration receiving antipsychotic medication. Exclusion criteria were history of head injury; medical conditions affecting peripheral blood flow, including peripheral vascular disease, connective tissue disease, diabetes mellitus, thyroid disease, significant hepatic or renal impairment, or any condition affecting hypothalamic function; use of medication affecting peripheral blood flow; history of alcohol dependence or intoxication within the last 48 hours; history of illicit drug abuse or dependence or illicit drug use within the preceding 24 hours; raised core temperature, or raised inflammatory markers. The majority (> 70%) of participants were smokers. This variable was not controlled for in the analysis.
The North Western Mental Health Research and Ethics Committee approved the study. Eligible subjects under the clinical care of L.K. and C.P. were approached for consent by an independent researcher (R.A.), and participating subjects provided written informed consent. Biochemical results including full blood exam, liver function tests, kidney function tests, thyroid and lipid profile, random blood sugar, and inflammatory markers were collected for each patient. Body mass index (BMI) was also recorded.
Process
Hand temperature was measured in an isolated temperature controlled (22–24°C) room. Subjects were asked to place their right palm on a polystyrene support 50 cm below a portable FLIR A40M internally calibrated infrared camera, and were requested to keep their hand still. The accuracy of the camera was <1°K, with a sampling rate of 1 Hz. Images were acquired over 1 min and imported into Igor Pro (Wavemetrics, Tigard, Oregon, USA). The image-processing feature was used to generate the colour–temperature maps as shown below in Figure 1. A calibrated image of each patient's hand, from which an average temperature value was calculated, was obtained for each of three experimental conditions. These included 30 min prior to medication (baseline), then 30 and 60 min post-medication. Time intervals were based on results from animal studies in which clozapine significantly increased cutaneous blood flow within minutes following subcutaneous injection [21]. Measurements were made before and after patients’ nocte medication, so baseline measurements were undertaken between 7.30–8.30 p.m. This process was carried out on two consecutive days, providing six measurements for each subject. Five subjects were also receiving fortnightly intramuscular depot injections, including risperidone and zuclopenthixol, in addition to their daily oral medications.
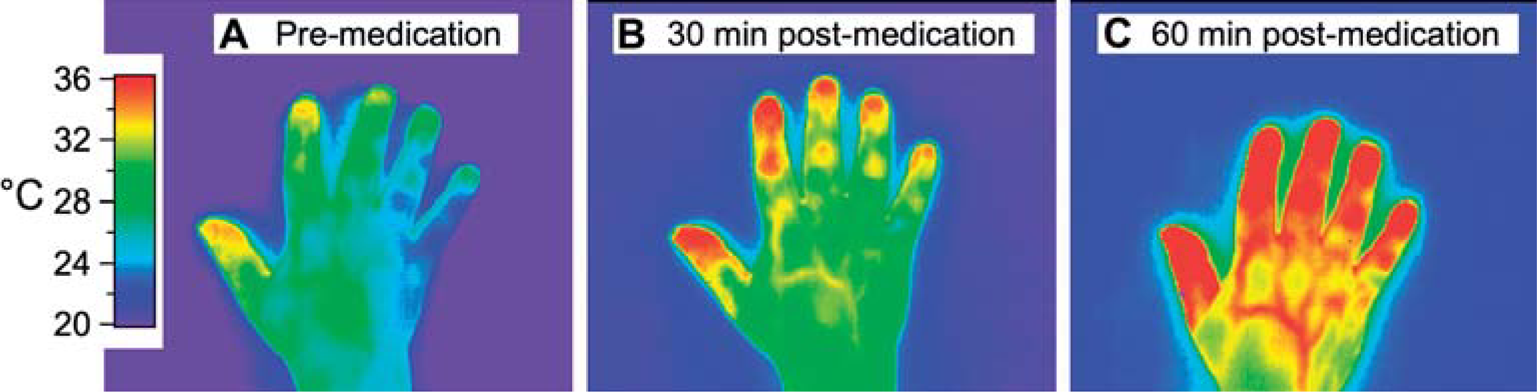
Infrared images showing the temperature gradient across the surface of the hand before medication (A) and 30 and 60 min after medication (B and C). The bar at the left of the figure shows the temperature scale derived from the internal calibration of the FLIR A40M infrared camera.
Each subject was assessed using the Brief Psychiatric Rating Scale [22]. Clinical assessments were completed by one of us (LK) with most assessments undertaken on the same day or in the same week as the finger blood flow measurements. Statistical comparisons were performed using regression and repeated measures analysis of variance (ANOVA).
Results
Hand temperature was measured on two consecutive days in 12 patients with diagnoses of schizophrenia (n = 8) or schizoaffective disorder (n = 4). All patients were taking one or two types of atypical antipsychotic (Table 1). There was a substantial correlation between the hand temperature increases observed for day one and the increases observed for day two (r = 0.68; p < 0.05). Infrared images of one patient (day 1) before and after medication are shown in Figure 1. The hand temperature increase 60 min after medication for this patient was 4.5°C. Group data from all patients is shown in Figure 2. Hand temperature increased significantly following medication (repeated measures ANOVA f(2,44) = 44.96) so that 30 min post-treatment values were significantly greater than pre-treatment values (p < 0.001), and 60 min post-treatment values were greater than both the pre-treatment (p < 0.01), and 30 min post-treatment values (p < 0.02). An increase in hand temperature following medication was observed in all patients, with a mean increase of 4.1 ± 2.4°C, and a range of 0.8–10.8°C.
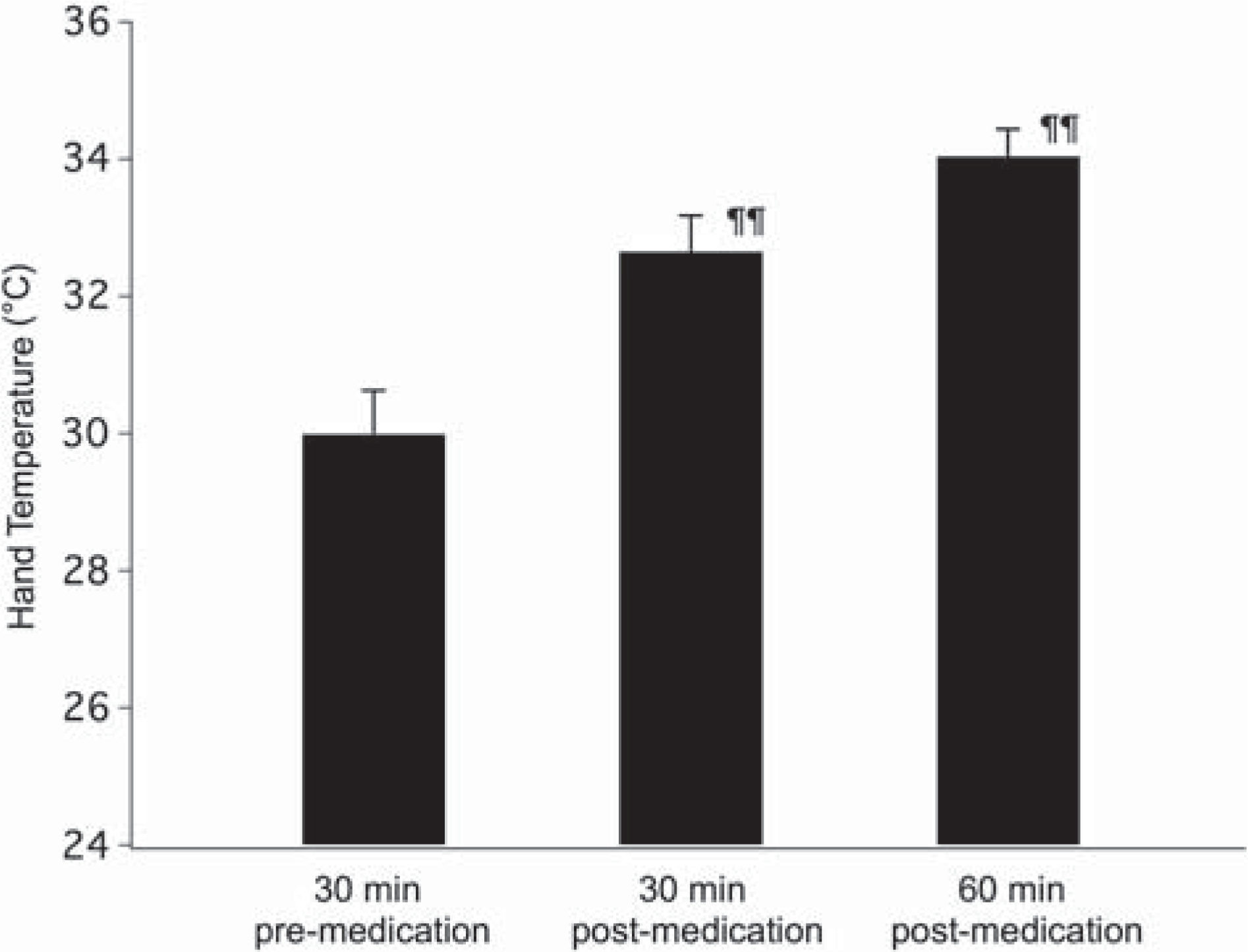
Effect of medication on hand temperature. Mean ± SE of hand temperature measurements for 30 min prior, and 30 and 60 min after medication. Values for the two treatment days were combined. ¶¶Significantly different from pre-treatment value, p<0.001, and different from each other p < 0.02 (repeated measures ANOVA, f(2,44) = 44.96, Fisher's protected least significant difference).
Medications taken by participants
The subjects in whom the greatest medication-induced hand temperature increases were observed were those with low baseline temperatures (Figure 3). Hand temperature increase was not correlated with BMI or with patient age.
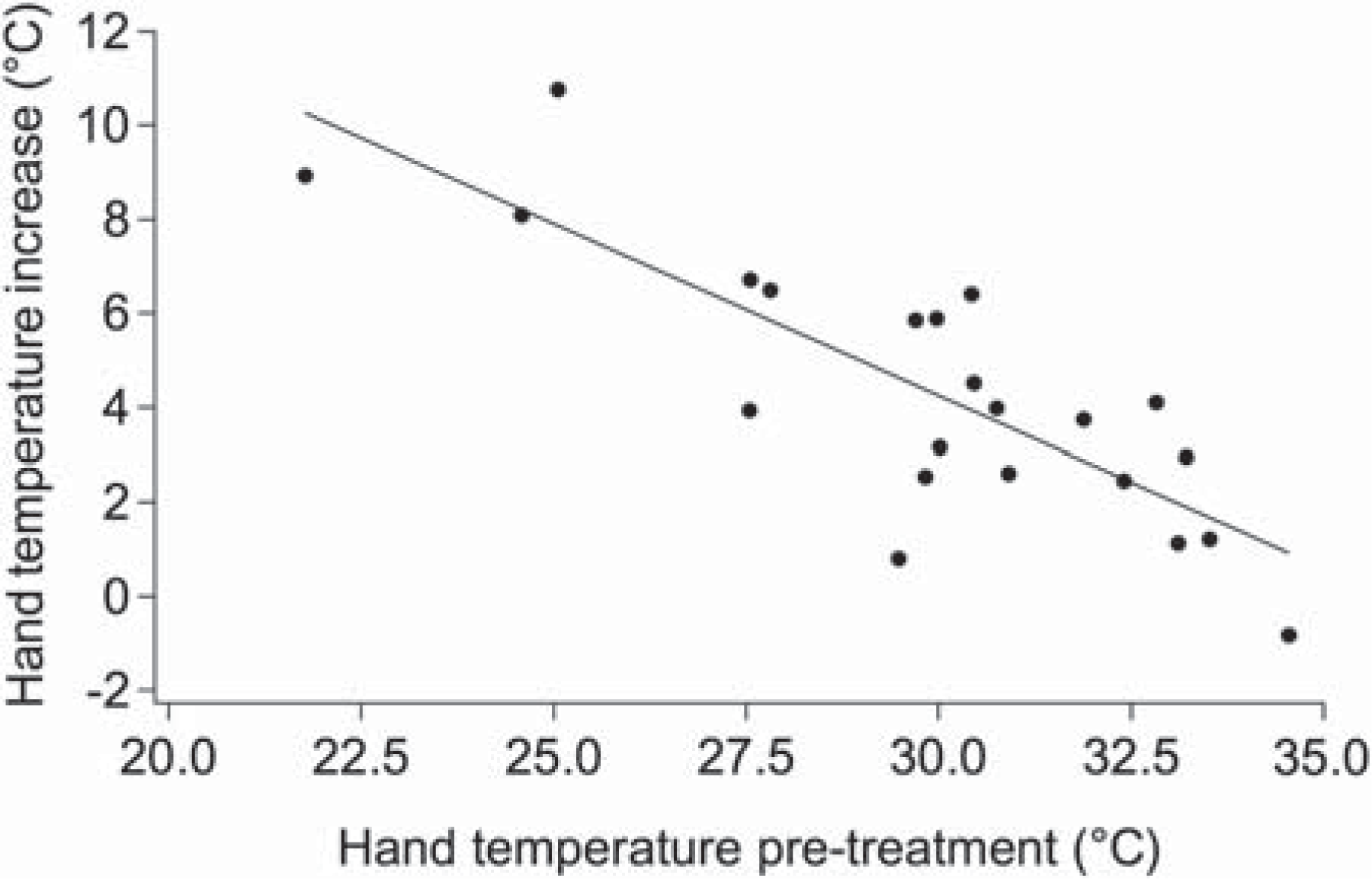
Relationship between baseline hand temperature and the increase observed 60 min after medication. Data for both treatment days are shown. Linear regression is shown (r = 0.76; p<0.005).
Figure 4 shows baseline hand temperature (Figure 4A) and medication-induced hand temperature increase (Figure 4B) as a function of BPRS score. There was a significant negative correlation between the BPRS score (r = −0.60, p < 0.05) and baseline hand temperature. Thus, the more severe the symptomatology, the lower the baseline hand temperature. There was also a significant positive correlation between the BPRS score and medication-induced hand temperature increase (r =0.71, p < 0.01), implying that atypical antipsychotics produce a greater increase in hand temperature in patients with more severe psychiatric symptoms.
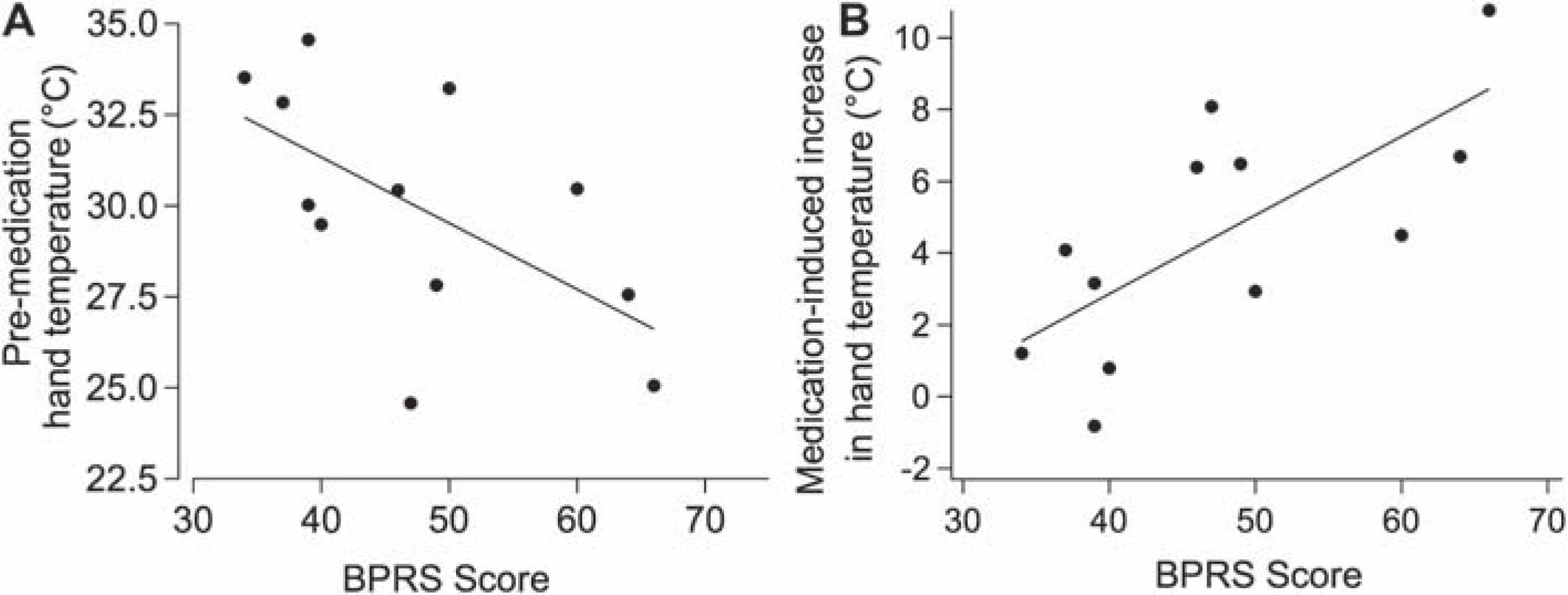
Relationship between BPRS score and hand temperature. A: pre-medication baseline hand temperature as a function of BPRS score, significant linear regression (r = −0.60; p<0.05). B: Medication-induced hand temperature increase, significant linear regression (r = 0.60; p<0.01).
Discussion
Our study is the first to report that atypical antipsychotic agents increase hand temperature in patients with schizophrenia and schizoaffective disorder. The sympathoinhibitory vasodilatory effects of atypical antipsychotics on the cutaneous vasculature in human patients are similar to those observed in animal models (see below). Patients with higher psychopathology scores on the BPRS have greater resting peripheral vasoconstriction and a greater medication-induced increase in hand blood flow. This is the first report of a relationship between a measure of symptom severity in schizophrenia, and the acute effects of atypical antipsychotic medications upon a physiological variable.
This study relates to physiological processes involved in stress-induced hyperthermia, a response to stressful stimuli that is evolutionarily conserved in mammals, including rodents, larger herbivores, wild cats, and primates and humans (for review see [30]). In animal models, the perception of stressful stimuli recruits brain coordination of sympathetic outflow to thermoregulatory end organs, including increased activity in brown adipose tissue (BAT), in which thermal uncoupling protein converts energy to heat, as well as sympathetic activation of cutaneous vasoconstriction, thereby decreasing heat loss, and together causing an acute increase in body temperature [6,9,23,24]. Brain pathways regulating these thermoregulatory end organs have been studied in animal models. The raphe nuclei in the medulla oblongata function as an integrative control centre in regulating sympathetic outflows from the spinal cord, and hypothalamic centres (dorsomedial hypothalamus and parvocellular paraventricular nuclei) are more rostrally situated control regions [25–27]. The link between the perception of salient events and increased cutaneous vasoconstriction requires the functional integrity of neurons in the amygdala [23,24,28,29].
Stress-induced hyperthermia occurs in normal human subjects, for review see [30]; however, underlying physiological mechanisms have not been established in humans. Cutaneous vasoconstriction of the hand in response to acute changes in emotional state has been documented extensively [12,19,31,32]. Until recently, significant BAT activity was thought not to occur in adult humans; however, positron emission tomography (PET) imaging has now demonstrated that a prolonged cold stimulus recruits heat-producing BAT activity in normal adult humans [33–35], implying that human basal metabolic rate is under active brain control via sympathetic outflow to BAT. Interestingly, overweight or obese subjects were found to have reduced BAT activity [35]. Whether BAT thermogenesis is activated by stressful stimuli in humans has not yet been investigated. The current results clearly demonstrate that cutaneous vasoconstriction in humans is subject to robust, acute inhibition by antipsychotic agents, and furthermore suggest that this effect may relate, via increased resting vasoconstriction, to disease activity in schizophrenia. As discussed below, results from animal models show that receptor mechanisms involved in mediating the sympathoinhibitory effect of antipsychotics may also be involved in both their therapeutic effect, as well as in their adverse effects of temperature dysregulation and weight gain. The question of whether increased resting vasoconstriction could feasibly be related to the disease process of schizophrenia is then discussed.
In animal models, antipsychotics affect thermoregulatory sympathetic outflow via receptor actions, namely blockade of 5-HT2A receptors (5-HT2A-Rs), and stimulation of 5-HT1A receptors (5-HT1A-R), thought to be involved in the therapeutic effect of these agents in humans [36–40]. Studies in rat and rabbit models show that cutaneous vasoconstriction in response to the perception of salient stimuli may be abolished via local intra-amygdala injection of a 5-HT2A-R antagonist [23,24,28,29]. Stimulation of 5-HT2A-Rs is an action of many psychosis-inducing compounds [41,42]. Systemically administered clozapine, chlorpromazine, olanzapine and risperidone (all of which block the 5-HT2A-R and/or stimulate the 5-HT1A-R) inhibit sympathetic cutaneous outflow, whereas haloperidol, lacking these actions, does not [6,21,43,44]. The 5-HT2A-R and 5-HT1A-Rs are also implicated in antipsychotic adverse effects. Clozapine causes hypothermia (reduced core temperature), both as a rare adverse clinical event [45] and as a consistent, mild effect [46,47]; 5-HT1A-R stimulation causes hypothermia [48] and 5-HT2A-R antagonists reverse hyperthermia elicited by 5-HT2A-R agonists. The current finding may also be relevant to a major adverse effect of antipsychotics, namely weight gain, which, curiously, is closely correlated with therapeutic effect [49,50]. Clozapine acutely inhibits BAT activity in rats, an effect that is blocked by both 5-HT2A-R blockade and 5-HT1A-R activation [9,10]. In addition, chronic administration of olanzapine has been shown to suppress BAT activity in rats, leading to weight gain at least partially independent of food-intake [51]. Thus suppression of BAT activity may significantly contribute to antipsychotic-induced weight gain, amongst other receptor interactions thought to mediate this effect; for review, see Kroeze et al. [52]. In light of the current result, future studies may determine whether antipsychotic-induced hand temperature increase and weight gain correlate over time, and whether this relates to therapeutic effect.
Evidence from the literature is consistent with the notion that schizophrenia is associated with abnormal thermoregulatory processes. Early investigators described cool, blue hands (acrocyanosis) in patients with schizophrenia as well as unusual thermoregulatory behaviour [13–15, 53–54]. Subnormal finger temperatures in unmedicated patients, and an increase with phenothiazine medication have also been documented [15,54,55], as have abnormalities in related variables including electrodermal activity (EDA) [16,17,56–61], and core temperature [62]. Notably, Dawson et al. found that EDA in schizophrenia patients was increased in the period prior to relapse of symptoms, as well as during the relapse, but not in remission [63]. Both atypical and some typical antipsychotics have been demonstrated to normalize elevated EDA [18]. Together with the current results, these previous reports support the hypothesis that increased resting sympathetic outflow accompanies disease activity in schizophrenia. This association suggests an underlying abnormality in brain mechanisms regulating the attribution of salience to perceptual events, such that the physiological consequences of salience are inappropriately recruited. Aberrant assignment of salience is recognized to be a central cognitive abnormality in schizophrenia [64,65], with documented diagnostic construct validity [66] and a positive relationship with delusions [67]. The current findings may also be relevant to the sensory gating theory of schizophrenia [68], in that inappropriate and persistent sympathetic outflow may result from a failure of brain mechanisms that normally mediate habituation to a novel (salient) stimulus.
Functional neuroimaging suggests possible links between brain areas implicated in psychosis and those regulating sympathetic outflow (electrodermal activity). In normal subjects, prefrontal cortical areas, including the medial prefrontal cortex (MPFC), modulate amygdala reactivity to salient stimuli [74], and neural activity in the amygdala has been shown to regulate salience-induced EDA [69–72]. Several studies have demonstrated schizophrenia patients to exhibit abnormal MPFC-amygdala connectivity in response to viewing emotive faces, for review see [79]. Reduced connectivity has been shown to correlate with increased EDA [72,77–78], and positive symptoms of schizophrenia, particularly suspiciousness and paranoia [72,73,77,78]. Thus, as suggested by several authors [71, 73], abnormal activity in the amygdala and regulatory cortical areas may be a key feature of schizophrenia, linking the pathophysiology of this disorder to our present observations of abnormal salience-induced cutaneous vasoconstriction.
Conclusion
Infrared measurement of hand blood flow via hand temperature is simple, non-invasive, rapid, and inexpensive, and results may be correlated with those obtained in laboratory animals. Our data obtained with this measure support the idea that patients with schizophrenia and schizoaffective disorder have excessive resting sympathetic cutaneous vasomotor tone that relates to symptomatology, and that this abnormality is alleviated by clozapine and clozapine-like antipsychotics. In the current study, only the acute effects of antipsychotics were measured. A longitudinal study in which hand temperature is measured on multiple occasions and correlated with a psychiatric assessment scale including both positive and negative symptoms would better determine the relationship between hand blood flow and symptomatology. Studies of patients with other psychiatric diagnoses, including anxiety disorders and mood disorders, will help establish whether increased resting sympathetic tone specifically characterizes schizophrenia. It may be that measurement of baseline hand temperature and the acute temperature response to antipsychotic medications will prove a useful guide to a clinically effective antipsychotic dose of particular agents in particular patients.
Footnotes
Acknowledgements
