Abstract
Antipsychotic drugs have been known for years to induce weight gain [1] and stimulate appetite [2]. In the last 15 years novel atypical antipsychotics have been developed that possess advantages over older typical agents in terms of both reduced side-effects (particularly reduced motoric extrapyramidal side-effects) and various enhanced therapeutic effects (greater efficacy against negative symptoms, reduction of suicidality and efficacy in some treatment-resistant patients who do not respond to older antipsychotic drugs) [3]. These advantageous properties have led to atypical antipsychotics being widely considered the drugs of first choice for the treatment of schizophrenia [4] and are now considered major therapeutic tools in the management of mood disorders [5]. In contrast, the relative effectiveness of second-generation antipsychotic drugs as compared with that of older agents have been addressed only recently in the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) study [6]. That large-scale study found that only olanzapine was the most effective in terms of rates of discontinuation, although this atypical antipsychotic drug was associated with greater weight gain and increases in measures of glucose and lipid metabolism [6]. Indeed, second-generation antipsychotics may induce marked weight gain associated with abdominal obesity [7] and enhanced adiposity [8], which causes expected increases in morbidity and mortality, as well as reduced quality of life [9], and interferes with medication compliance [10], leading to relapse [11]. Thus, weight gain and metabolic disturbance encountered with these agents have emerged as a major concern for clinicians nowadays [12–19], at the same time as we face an obesity epidemic.
The ability of antipsychotic drugs to induce weight gain is of particular significance in people with schizophrenia and mood disorders who are typically overweight [20, 21] and who have enhanced levels of visceral fat [22, 23]. Typically, weight gain seen with the newer antipsychotics increases over a number of months of treatment and ultimately reaches a plateau that may vary between drugs [24–27]. Although atypical antipsychotic agents are the most effective pharmacological tools in the management of many psychotic and affective disorders, the choice of such a medication should be individualized to each patient, with consideration of the risks and benefits of treatment [28–30]. In this respect, screening and monitoring of weight, diet, and exercise in patients with mood and psychotic disorders will help to limit or eliminate weight gain and obesity as potential added metabolic risk factors [31–34]. Indeed, patients with schizophrenia and mood disorders should be screened regularly, especially in the presence of other risk factors for type 2 diabetes [35].
While treating obesity in the general population has proven to be a challenge, the challenge may be even greater for those who have gained weight in relation to a mental disorder and its pharmacological treatment. It might be indeed extremely difficult for some chronically and severely mentally ill patients who require antipsychotic treatment to eat less and exercise more when their treatment increases appetite and produces fatigue and sedation, and their illnesses decrease motivation and limit social interactions and activities. Moreover, their typically strained financial resources and social circumstances often limit their access to low-calorie foods and preclude membership in health clubs, access to exercise equipment, and dietary counselling or education. Such general health-promoting activities are rarely provided in contemporary psychiatric clinical services or covered by health insurance programmes for psychiatric illness, despite support for their integration into mental health services [36].
Accordingly, the present study aimed to document the effectiveness of a behavioural weight control programme including physical exercise in the prevention of antipsychotic-induced weight gain and associated comorbid conditions in outpatients with schizophrenia and mood disorders. Only a small number of studies have examined behavioural intervention programmes including both diet and exercise counselling in this patient population [37–41]. They provided the best evidence for small weight reductions among patients with schizophrenia over short time intervals [37] or considered the long-term success of these interventions [38–41]. However, such a conclusion is tempered by some design flaws and the small sample sizes used in those studies. Owing to the fact that antipsychotic drugs are associated with weight gain and metabolic dysregulation and because little is known about the management of this problem, we thus sought to examine the potential effectiveness of a behavioural weight control programme over an 18 month period by comparing its outcome to the status of control subjects who were also clinically supervised over the same period of time.
Methods
Subjects
Subjects fulfilling the following criteria were eligible for participation: ≥18 years of age; DSM-IV diagnosis of schizophrenia, schizoaffective or bipolar disorder; sedentary or moderately active (i.e. <3 h of physical activity/week); and in current treatment with an atypical antipsychotic drug alone or in combination with other psychotropic agents. Patients presenting medical contraindications to follow a physical activity programme or those having previously been enrolled in a similar clinical programme were excluded of the present study. A total number of 130 subjects were recruited through the outpatients followed in the Psychiatric Departments of Hôpital de l'Enfant-Jésus and Centre Hospitalier Robert-Giffard (Quebec, Canada). Among those subjects, 70 were included in the active group (experimental group) and 60 were considered as controls. The subjects were allocated to a 1:1 ratio after we decided to include a control group, explaining the different sample sizes. All of these subjects continued their habitual psychiatric follow up with their psychiatrist. The subjects included in the control group presented the same baseline characteristics and constituted the comparison group (Table 1). The effectiveness of this behavioural weight control programme was thereby evaluated by the addition of a clinical programme over an 18 month period and by comparing the outcomes obtained to the outcomes of habitual psychiatric outpatients who were not implicated in such a programme but who were supervised according to usual clinical practices. All subjects provided written informed consent after review and approval of the study by the Ethical Committee of the Centre Hospitalier Affilié Universitaire de Québec.
Baseline patient characteristics
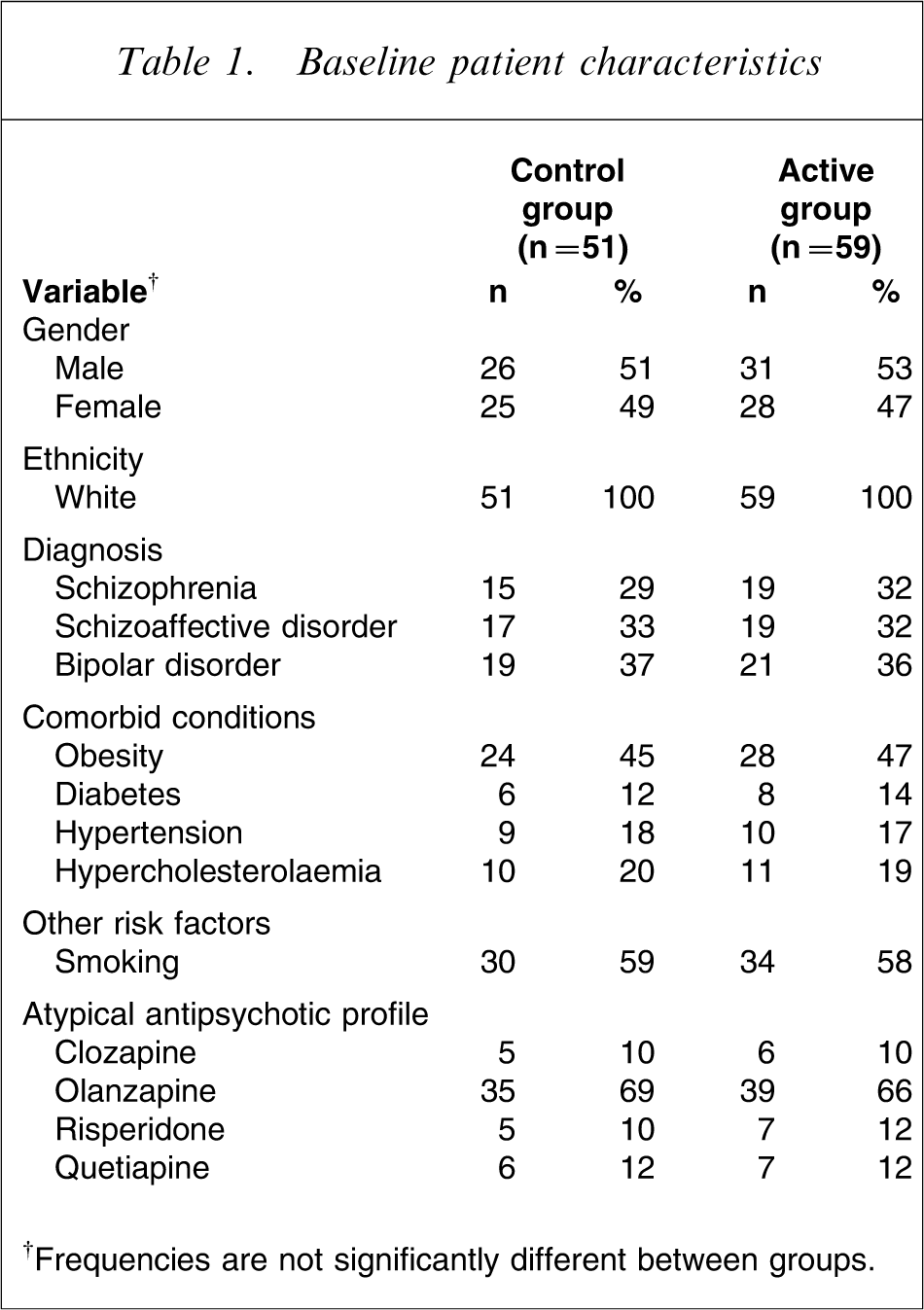
†Frequencies are not significantly different between groups.
Project overview
This prospective, comparative, open and naturalistic study aimed to determine the potential effectiveness of a behavioural weight control programme in the prevention of antipsychotic-induced weight gain and associated comorbid conditions in outpatients with schizophrenia and mood disorders. The patients included in the active group (nutritional recommendations and physical exercise) were compared to those who were not included in this clinical programme. In this regard, the programme offered to the experimental group included a small group educative activity about dietary and physical activity counselling. This educative activity of 90 min length was given once at the beginning of the study for the active group and was delivered by a nutritionist and a psychiatric nurse. The focus here was that the patient understands the role of nutrition and exercise as a vital therapeutic goal in preventing many risk factors. The Canada's food guide to healthy eating was a useful tool for this nutrition education counselling [42]. In addition, the active group was involved in a structured, supervised, facility-based exercise programme in a small gymnasium of the Hôpital de l'Enfant-Jésus. A kinesiologist supervised small groups (8–10 subjects) who were devoted to exercise sessions including cardiovascular workouts on treadmills and stationary cycles. Strength-training exercises included use of free weights and resistance bands as well as flexibility and balance drills. Music was used to maintain enthusiasm for exercise sessions. Rates of perceived exertion and heart rate were closely monitored. Special attention was given to subjects with medication-related impaired balance and coordination. This individualized fitness training was performed for 60 min twice a week over the 18 month period.
Assessments
Patients were evaluated at 12 study visits: screening visit for eligibility, baseline visit within 1 month of the initial screening visit, and at months 1, 2, 3, 4, 5, 6, 8, 10, 12 and 18. Screening procedures included physical and mental examination, 12-lead electrocardiogram and medical history. During the other visits the measurements described in the following section were performed in each subject.
Anthropometric measurements
Bodyweight, height, and waist circumference (WC) were measured according to standardized procedures recommended at the Airlie Conference [43]. Body mass index (BMI) was calculated as bodyweight divided by height squared (kg m−2).
Biochemistry
Blood samples were collected from an antecubital vein into vacutainer tubes containing ethylenediamine tetra-acetic acid (EDTA) after a 12 h overnight fast for the determination of plasma lipids and lipoproteins. Plasma was separated immediately after blood collection by centrifugation at 850 g for 10 min at 4°C. Plasma cholesterol and triglyceride concentrations were determined enzymatically in plasma lipoprotein fractions using a Technicon RA-500 automated analyzer (Bayer, Tarrytown, NY, USA) and enzymatic reagents were obtained from Randox (Crumlin, UK). Plasma lipoprotein fractions (low-density and high-density lipoprotein; LDL and HDL) were isolated using previously described procedures [44]. In addition, serum concentrations of prolactin, thyrotropin-stimulating hormone (TSH), and glycated haemoglobin (HbA1c) were performed by standardized procedures routinely used at the Centre Hospitalier Robert-Giffard (only at baseline and at months 6, 12, and 18 for these three variables). Finally, fasting plasma glucose concentrations were enzymatically determined using the technique of Richterich and Dauwalder [45].
Psychological assessments
The Clinical Global Impression scale (CGI) was used to describe the patient's overall clinical state as a global impression by the psychiatrist [46]. Both the CGI-Severity (CGI-S) and the CGI-Global Improvement (CGI-I) scales were used in the present study. The CGI-S assesses the clinician's impression of the patient's current illness state and gives a score ranging from 1 (disease-free) to 7 (greatest possible severity), whereas the CGI-I assesses the patient's improvement or worsening since the start of the study and gives a score ranging from 1 (very much improved) to 7 (very much worse). In addition, the psychopathology state of subjects was determined with the Brief Psychiatric Rating Scale (BPRS) [47]. The BPRS is a Likert-type scale composed of 18 items that are evaluated by the interviewer, in which each item gives a score ranging from 0 (absence of symptom) to 7 (extreme severity). Finally, the Short Form (SF)-36 Health Survey was used as a generic measure of health status [48]. It is a multipurpose, short-form health survey that contains 36 questions and yields an eight-scale profile of scores as well as a summary of physical and mental measures. High scores are associated with a good health status. Both the BPRS and the SF-36 Health Survey were used only at baseline and at months 12 and 18. Furthermore, it is to be noted that the clinician ratings of psychopathology were blind to control or intervention status.
Statistical analysis
Mean, standard deviation, and frequency were calculated. A χ2 test was used for comparison of frequencies between the control and active group. An analysis of variance (ANOVA) for repeated measures was performed to assess the effect of sex as well as the sex × treatment (control vs physical activity programme) interactions on all dependent variables. Because there was no sex effect or sex × treatment interaction, we combined the data for both sexes in order to improve clarity and statistical power. An ANOVA for repeated measures was used to assess the differences over time. A Tukey's honestly significant difference post-hoc test was then performed in order to contrast mean differences. Adjustment for type of drug, dose of drug or duration of drug therapy had no meaningful effect on the outcomes. Data are given as mean ± SD unless otherwise noted. Statistical significance was set at p < 0.05. All statistical analyses were performed using JMP version 3.2.2 (SAS Institute, Cary, NC, USA).
Results
Participants
Of 70 enrolled subjects in the active group, 11 dropped out for many reasons (patient request, n = 4; loss to follow up, n = 3; difficulties with scheduling, n = 3; or mortality, n = 1). Similarly, the control group was first constituted of 60 subjects, of whom nine dropped out for the same reasons (patient request, n = 3; loss to follow up, n = 2; difficulties with scheduling, n = 3; or mortality, n = 1). In this regard the adherence rate of patients was 85%, both in the active and in the control group. In addition, the subjects included in the exercise programme were in general compliant with this clinical intervention and particularly by their capacity to reach a minimal commitment of six sessions every 4 weeks. The baseline characteristics of participants were not significantly different between the two groups, as shown in Table 1. The mean age of subjects was 36.1±6.1 years and 35.3±5.2 years in the active and control group, respectively. Moreover, the patients did not present significant mean difference between groups regarding the duration of drug therapy before study entry. They had taken the current primary antipsychotic agent for an average of 2.63±1.68 years and 2.68±1.61 years for the active and control group, respectively. The duration of secondary psychotropic medications before study entry was similar (2.42±2.34 years vs 2.36±2.82 years for the active and control group, respectively).
Changes in anthropometric variables
Figure 1 presents changes in bodyweight over the study in the active and control groups, whereas Table 2 shows values over time and changes from baseline for anthropometric measurements. Whereas the control group experienced a significant increase in bodyweight (4.1%), BMI (5.5%) and WC (4.2%), the active group reduced significantly its bodyweight (−3.5%), BMI (−4.4%), and WC (−4.6%) by the study end-point. Therefore, the changes observed at the study end-point for these three anthropometric variables differed substantially between the two groups: bodyweight (difference of 6.7 kg, p < 0.01), BMI (difference of 3.2 kg m−2, p < 0.01) and WC (difference of 9.3 cm, p < 0.01).
Mean changes in bodyweight over the study in the control (n = 51) and active (n = 59) groups. Vertical bars, standard errors. ∗∗p < 0.01.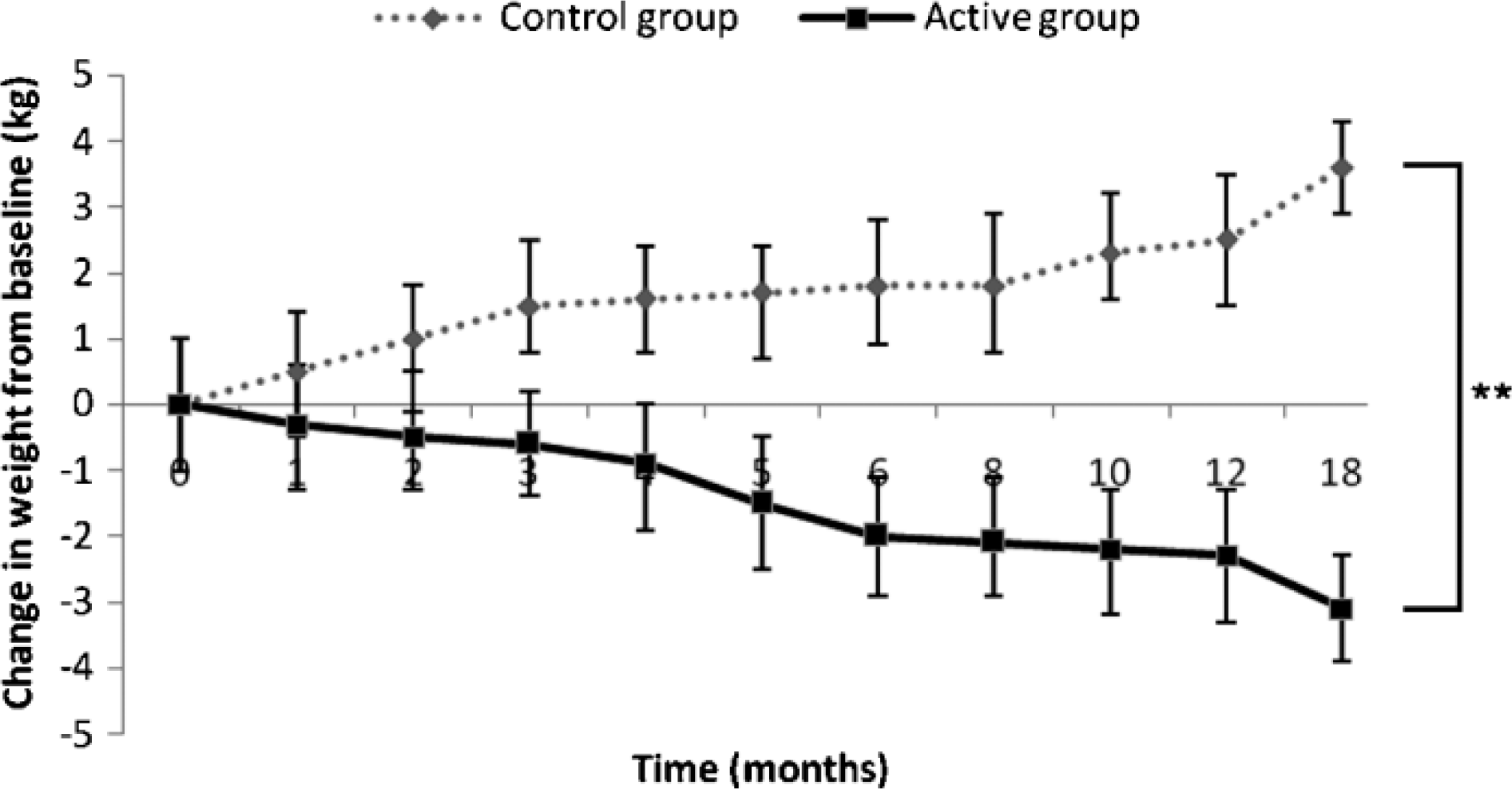
Anthropometric measurements
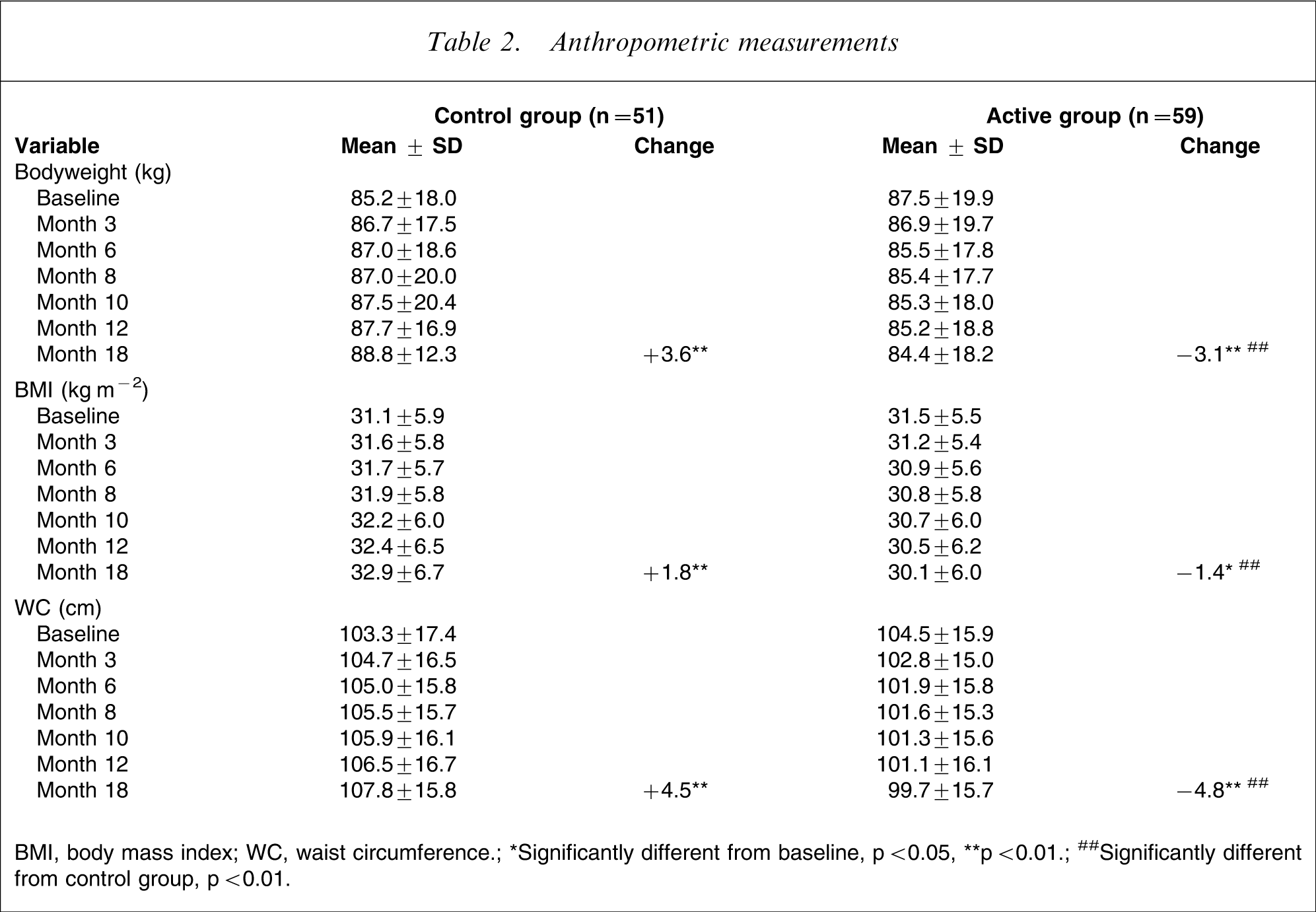
BMI, body mass index; WC, waist circumference; ∗Significantly different from baseline, p < 0.05, ∗∗p < 0.01; ##Significantly different from control group, p < 0.01.
Changes in biochemistry
As shown in Table 3, an opposite trend was observed between the two groups regarding the lipid–lipoprotein profile and fasting glucose concentrations over time as well as for changes from baseline. Indeed, a significant increase in LDL-cholesterol (14.8%) and in triglyceride concentrations (12.3%) was observed at month 18 for the control group. In contrast, HDL-cholesterol significantly increased (21.4%), and LDL cholesterol (−13.7%), triglycerides (−26.2%), total cholesterol (−12.1%), and fasting glucose concentrations (−12.0%) significantly decreased compared to baseline in the active group. In regard to the variations observed between the two groups at study end-point, significant mean differences were observed as follows: total cholesterol (difference of 1.00 mmol L−1, p < 0.01), LDL-cholesterol (difference of 1.02 mmol L−1, p < 0.01), HDL-cholesterol (difference of 0.33 mmol L−1, p < 0.05), triglycerides (difference of 0.88 mmol L−1, p < 0.05), and fasting glucose concentrations (difference of 0.79 mmol L−1, p < 0.05). No significant changes were observed regarding serum concentrations of prolactin and TSH over the study, whereas HbA1c significantly decreased (−11.4%) compared to baseline in the active group (data not shown).
Plasma lipid–lipoprotein profile and fasting glucose
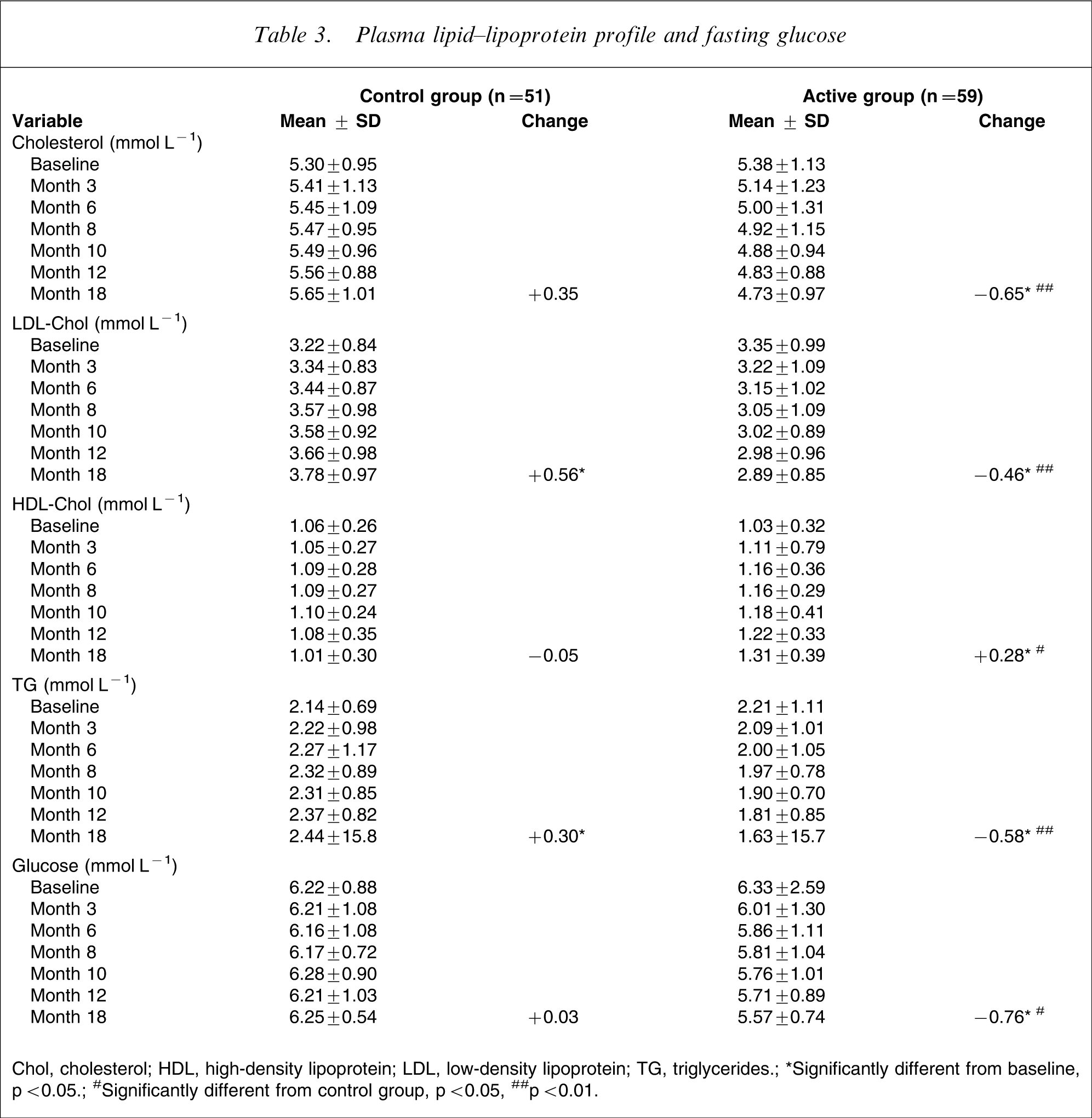
Chol, cholesterol; HDL, high-density lipoprotein; LDL, low-density lipoprotein; TG, triglycerides; ∗Significantly different from baseline, p < 0.05; #Significantly different from control group, p < 0.05, ##p < 0.01.
Changes in psychological measures
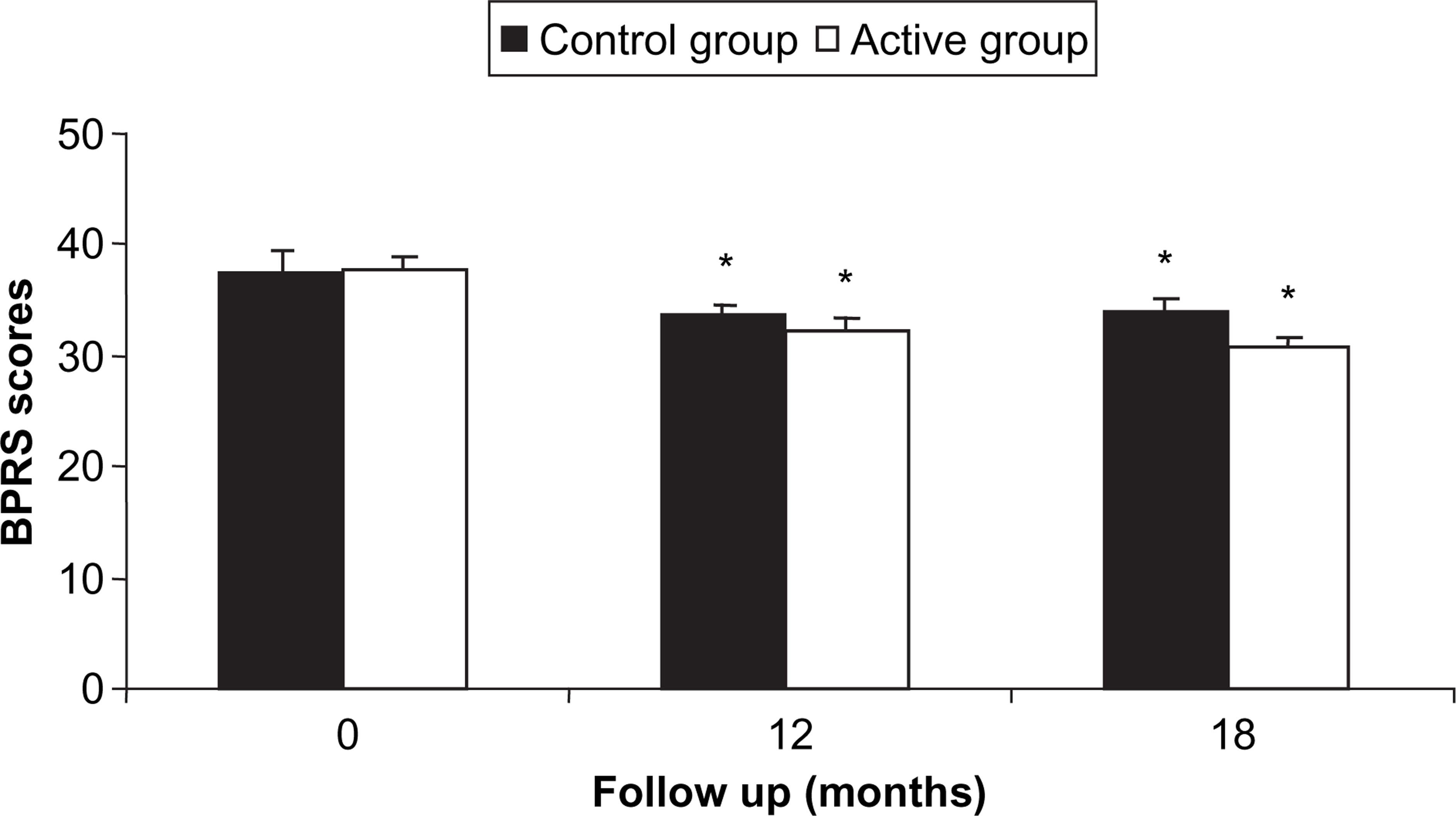
No between-group differences were seen in the clinical ratings of CGI-S and CGI-I during the study (Figure 2). Nevertheless, mean CGI-S scores were significantly lower for both groups at months 3, 6, 8, 10, 12 and 18 (p < 0.05) compared to baseline, and particularly for the active group at month 18 (p < 0.01). With respect to CGI-I scores, only the active group presented significantly lower mean scores at months 12 and 18 compared to baseline (p < 0.05). No significant difference was observed between the two groups (Figure 3) regarding the psychiatric status measured with the use of BPRS. However, both groups were significantly improved at months 12 and 18 (p < 0.05) compared to baseline.
Mean scores for Clinical Global Impressions scales for (a) severity (CGI-S) and (b) global improvement (CGI-I). Vertical bars, standard errors. ∗significantly different from baseline, p < 0.05; ∗∗p < 0.01. Values within each mean score are not significantly different. Control group, n = 51; active group, n = 59. Mean scores for the Brief Psychiatric Rating Scale (BPRS). Vertical bars, standard errors. ∗significantly different from baseline, p < 0.05. Values within each mean score are not significantly different. Control group, n = 51; active group, n = 59.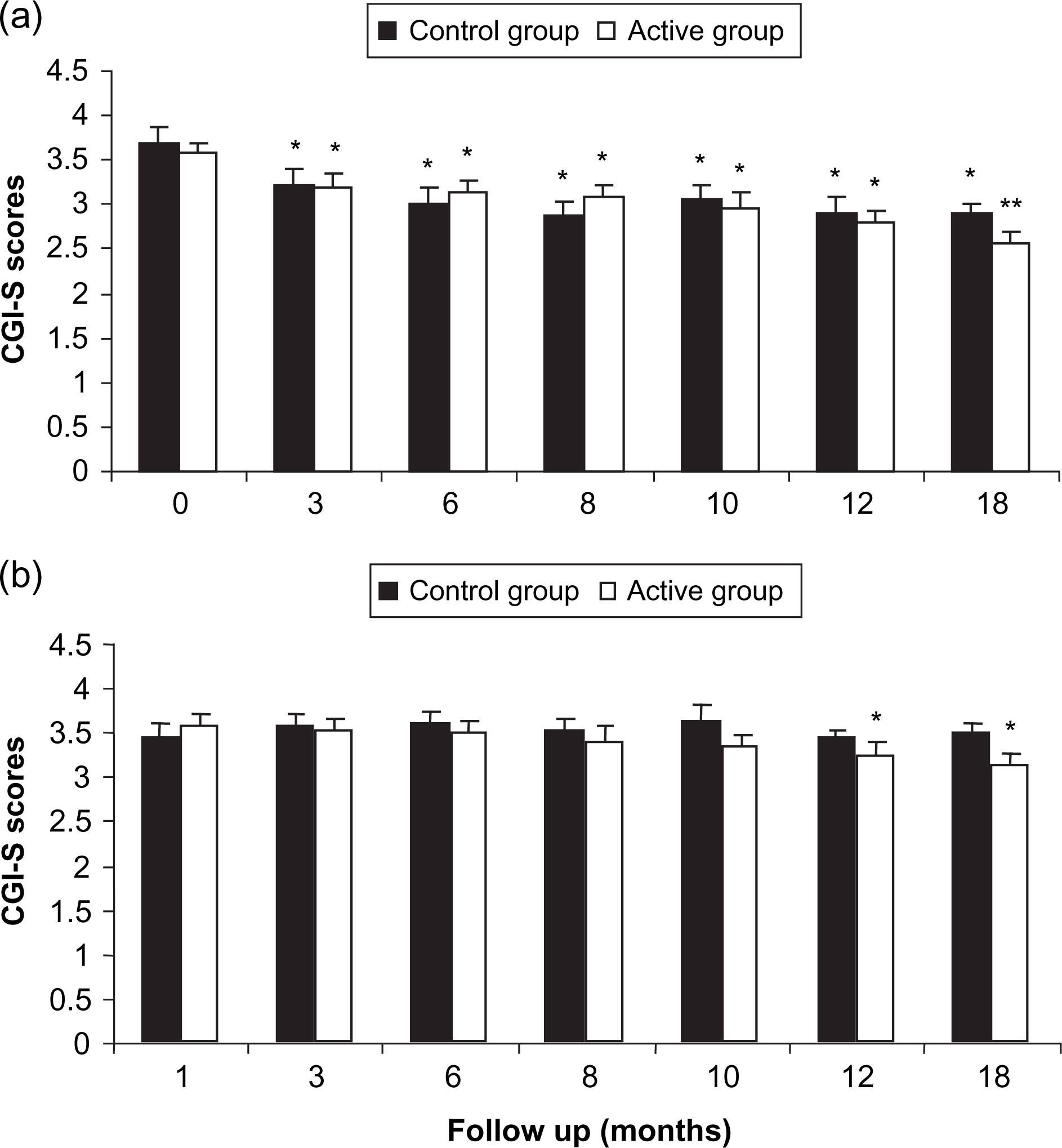
Finally, the SF-36 Health Survey was used as a tool for measuring health status (physical and mental health) and was filled out by the subjects. The results showed that physical health was improved only for subjects in the active group at months 12 and 18 compared to baseline (p < 0.05). In contrast, mental health was significantly improved for both groups at months 12 and 18 compared to baseline (p < 0.05; Figure 4).
Mean scores of the two summary measures (physical and mental health) of the Short Form (SF)-36 Health Survey. Vertical bars, standard errors. ∗significantly different from baseline, p < 0.05. Values within each mean score are not significantly different. Control group, n = 51; active group, n = 59.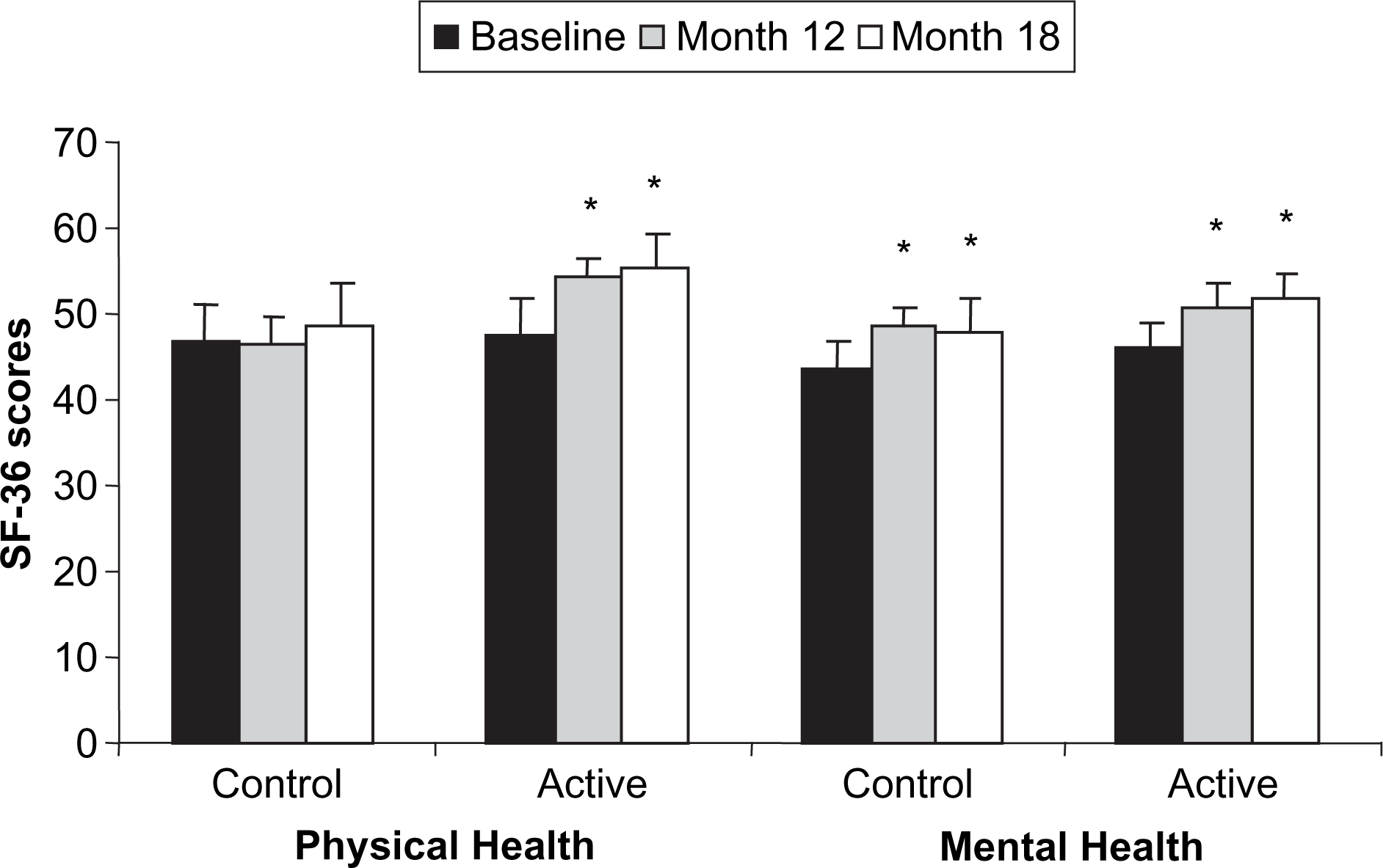
Discussion
The present study shows that bodyweight and metabolic risk profile in patients receiving atypical antipsychotics can be managed with a weight control programme including physical activity. Indeed, results obtained in the present study are of relevance and these findings add to the emerging view that weight gain is a major health problem associated with modern antipsychotic agents and that physical activity-based weight-control efforts in patients requiring antipsychotic treatment yield clinically promising benefits. Although the previous evidence [37–41] concurred with the results obtained here, those studies had several limitations that temper clear conclusions. In contrast, the present study included a relatively large sample size, the use of a control group and an 18 month follow up.
It is now well recognized that weight loss, even modest, has many health-related benefits. Specifically, the main argument justifying weight-reducing programmes relies on the improvement of the metabolic profile. We now refer to the term ‘metabolic syndrome’ or ‘insulin resistance syndrome’ the cluster of risk factors increasing the relative risk of type 2 diabetes and cardiovascular diseases [49]. Among the five screening variables and cut-offs proposed by the National Cholesterol Education Program–Adult Treatment Panel III [50] and used to identify subjects with the metabolic syndrome, the majority of them were improved in the active group (waist circumference, circulating levels of triglycerides and HDL-cholesterol, and fasting glycaemia), meaning that the morbidity and mortality risk profile of patients has been potentially reduced [51]. Such an outcome is not trivial because the reduction in cardiovascular risk was estimated to be 75% higher in the active compared to the control group based on available algorithms [52].
The evidence supporting the need for interventions to promote physical activity among persons with serious mental illness was recently reviewed [53]. The psychological benefits for clinical populations comes from two meta-analyses of outcomes of depressed patients that showed that effects of exercise were similar to those of psychotherapeutic interventions [54, 55]. In addition, exercise can alleviate secondary symptoms such as low self-esteem and social withdrawal [56]. Research also suggests that exercise is well accepted by people with serious mental illness [57] and is often considered one of the most valued components of treatment [58]. Thus, because individuals with serious mental illness are significantly less active than the general population [59–61], physical activity as an adjunct treatment for individuals with serious mental illness is highly desirable. Moreover, physical activity interventions are a critical component of a biopsychological approach in recovery-oriented mental health services [62].
The subjects included in the active group benefited socially from their participation in the study and reported favouring the peer support and encouragement associated with the programme. It is known that the challenge to managing obesity may be greater for those who have gained weight largely due to the use of psychotropic medications [63]. Indeed, patients who require antipsychotic treatment present a decrease in motivation and social interactions and activities. In addition, with the weight gain and metabolic effects observed in association with olanzapine (most of the patients were on olanzapine in the present study), alternative atypical agents are now available that do not carry the cardiovascular risk that this drug carries. Thus, this emphasizes the necessity for a better management of antipsychotic-induced weight gain in psychiatric patients, particularly by targeting individuals prior to weight gain.
The fact that these patients were relatively stable on medications before entry into the study means that they may have already gained a lot of weight before entering the study. Indeed, weight gain seen with the newer antipsychotics increases over a number of months of treatment and ultimately reaches a plateau [24–26]. For example, clozapine has been reported to induce weight gain that persists for as long as 46 months [27], while olanzapine-induced weight gain reaches a plateau after 10 months of treatment [26]. Nevertheless, the control group continued to gain weight, that is, 3.6 kg over the 18 month programme. This progression of weight gain, which is counter to the proposed notion of plateau, is not really surprising when one considers the fact that bodyweight/fat gain represents an unfortunate solution/adaptation to restore body homeostasis [64]. Beyond the positive energy balance explained, among other factors, by physical inactivity, we cannot exclude the possibility that bodyweight/fat gain might confer salutary effects on psychological health.
In addition to the clinical benefits obtained in the active group regarding the metabolic risk profile, changes observed in psychological measures were also an outcome of interest in the present study. In this respect, even though the clinical ratings of CGI-S, CGI-I and BPRS did not really permit observation of a difference between the two groups, these mean scores were significantly lower for both groups study compared to baseline. In addition, the behavioural weight control programme seemed to have had an impact on physical health as measured with the use of SF-36 Health Survey, albeit mental health was significantly improved for both groups at months 12 and 18 compared to baseline. These results suggest that an improvement in mental health occurred in patients of both groups over the study and that a specific effect of such a weight control programme on psychological measures was not detectable on these psychiatric scales.
In conclusion, the present findings highlight the importance of ongoing weight management intervention including physical exercise designed specifically for patients on antipsychotic treatment. The present study shows that such a programme is feasible and effective and the results obtained here are encouraging with a high rate of adherence. Nevertheless, further studies are needed to confirm the long-term success of behavioural weight control programmes in psychiatric patients and to determine the optimal modalities of such programmes.
Footnotes
Acknowledgements
This study was funded by Eli Lilly Canada. Eli Lilly Canada was not involved in any part of the study except funding.
