Abstract
At least twice the upper limit of normal troponin was found in 90% of cases, but 5 cases had C-reactive protein more than 100 mg/L and left ventricular impairment by echocardiography without a clinically significant rise in troponin.
The proposed monitoring protocol recommends obtaining baseline troponin I/T, C-reactive protein and echocardiography, and monitoring troponin and C-reactive protein on days 7, 14, 21 and 28. Mild elevation in troponin or C-reactive protein, persistent abnormally high heart rate or signs or symptoms consistent with infective illness should be followed by daily troponin and C-reactive protein investigation until features resolve. Cessation of clozapine is advised if troponin is more than twice the upper limit of normal or C-reactive protein is over 100 mg/L. Combining these two parameters has an estimated sensitivity for symptomatic clozapine-induced myocarditis of 100%. The sensitivity for asymptomatic disease is unknown.
The association between clozapine and myocarditis was first clearly demonstrated in 1999 by the article by Kilian et al. [1] using Australian data. Immediately following publication of that article, Novartis circulated in Australia cardiac monitoring guidelines designed to capture cases of myocarditis before they become serious.
The guidelines were developed based on the cases reported in the Kilian article [1], which were cases reported to the Australian Therapeutic Goods Administration (the drug regulatory authority), published cases and other cases of which the company had been advised. Thus these guidelines were not based on data collected systematically for each case. The effectiveness of these guidelines has not been investigated, with respect either to prevention of death and prolonged injury from myocarditis or to discontinuation of clozapine unnecessarily in individuals with mild physical illness who may benefit from continuation of clozapine without cardiac consequences.
In Australia most individuals commence clozapine as inpatients, for the purposes of increased clinical surveillance. This strategy means that documentation of symptoms, vital signs and pathology results is more comprehensive than would be the case with patients initiated in the community. Using data from patient's medical records as our source, we have described a systematic review of cases of myocarditis, comparison of cases with control patients and development of a case definition [2]. Using these data and additional information collected from the documentation of further cases and controls, we propose here a monitoring protocol for myocarditis in patients starting clozapine.
Materials and methods
The methodology has been described elsewhere [2]. Briefly, suspected cases of myocarditis were documented from the patients’ medical records and data for each patient were reviewed by the study steering group for compliance with the case definition involving histological evidence or a combination of clinical and diagnostic criteria. Controls were selected from individuals starting clozapine at the same mental health service and at around the same time as a case. Controls were also documented from patients’ medical records and were required to have taken clozapine for at least 45 days with documented evidence sufficient to exclude myocarditis (at least one of: no tachycardia, no rise in troponin, normal echocardiogram) or to have taken clozapine for at least 6 months continuously without manifest cardiac disease. Cases and controls started clozapine between January 1994 and January 2009.
Approval for the study was obtained from the Human Research Ethics Committees of the following institutions and health services: Monash University, Austin Health, Barwon Health, Bayside Health, Bendigo Health, Department of Human Services, Department of Justice, Eastern Health, Mercy Health, North Western Mental Health, Peninsula Health, St Vincent's Health, Southern Health (all from Victoria); Northern Sydney Central Coast Health, Sydney South West Area Health Service (Royal Prince Alfred Hospital Zone) (from New South Wales); and Prince Charles Hospital (from Queensland). The approvals covered access to medical records without patient consent. In addition, an Access Agreement was signed with the Victorian Institute of Forensic Medicine for access to the National Coroners’ Information Service database and a Deed of Confidentiality and Conflict of Interest with the Therapeutic Goods Administration for access to original case reports.
Results
The case definition was met by 75 cases. Nine cases were fatal and for these the diagnosis was made by histological examination. The remainder met the clinical and diagnostic criteria for myocarditis [2]. The time to onset of myocarditis is presented graphically in Figure 1, where the duration of clozapine has been used as the marker for time to onset. Some 83% of cases occurred between days 14 and 21 inclusive, and three of five late-occurring cases were fatal. The requirements for controls were met by 94 individuals commencing clozapine. The demographic data for the cases and controls are presented in Table 1 and a comparison of selected clinical and diagnostic features of cases and controls is provided in Figure 2. Figure 3 presents the distribution of maximum recorded C reactive protein (CRP) and troponin I/T results for cases.
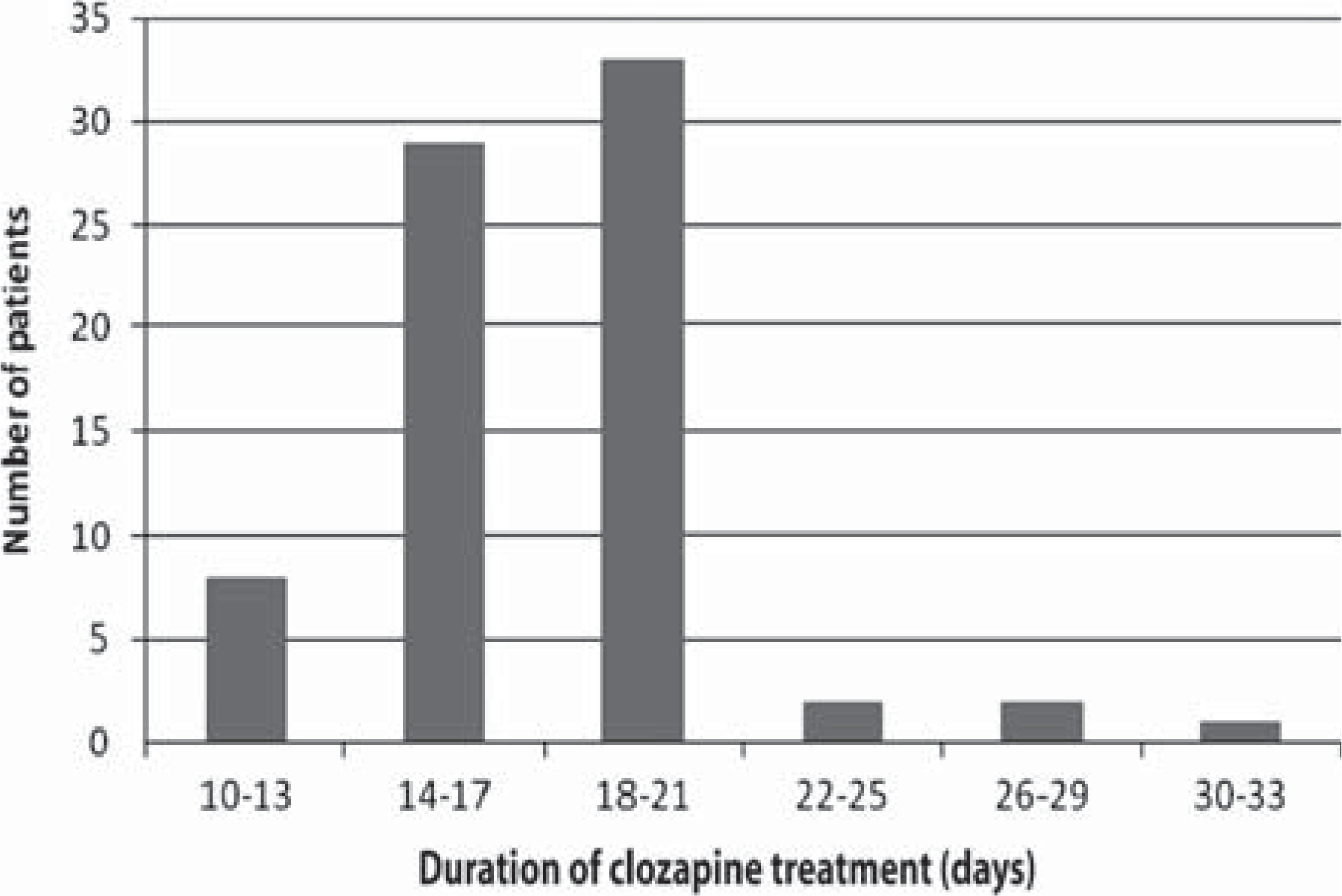
Bar graph indicating the distribution in time to onset of myocarditis for cases, as measured by the duration of clozapine.
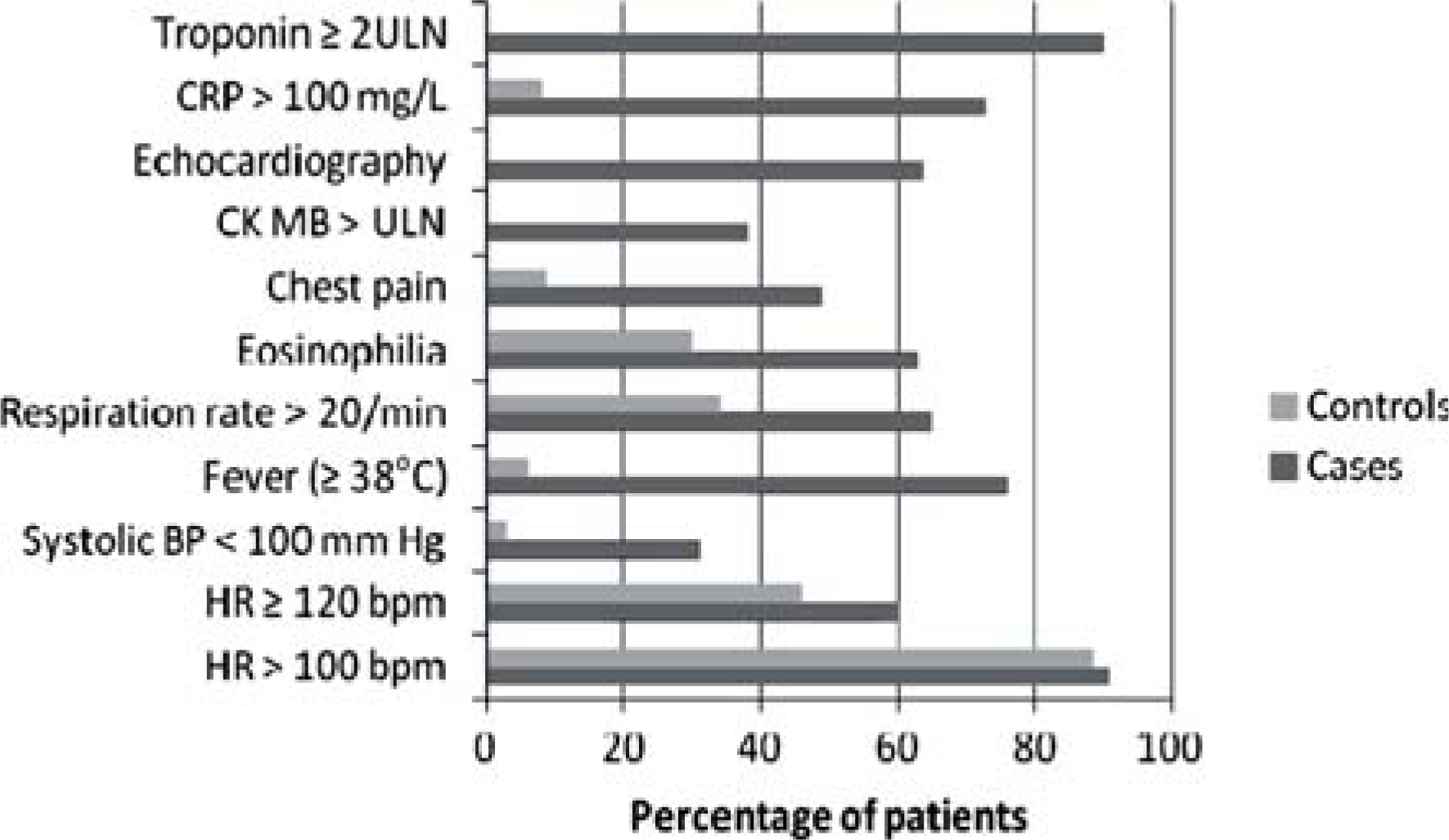
Comparison of cases and controls indicating discriminatory features. Percentages have been calculated using the total number of individuals in whom the feature was investigated as the denominator. BP, blood pressure; bpm, beats per minute; CK MB, creatine kinase muscle and brain; CRP, C-reactive protein; HR, heart rate; ULN, upper limit of normal.
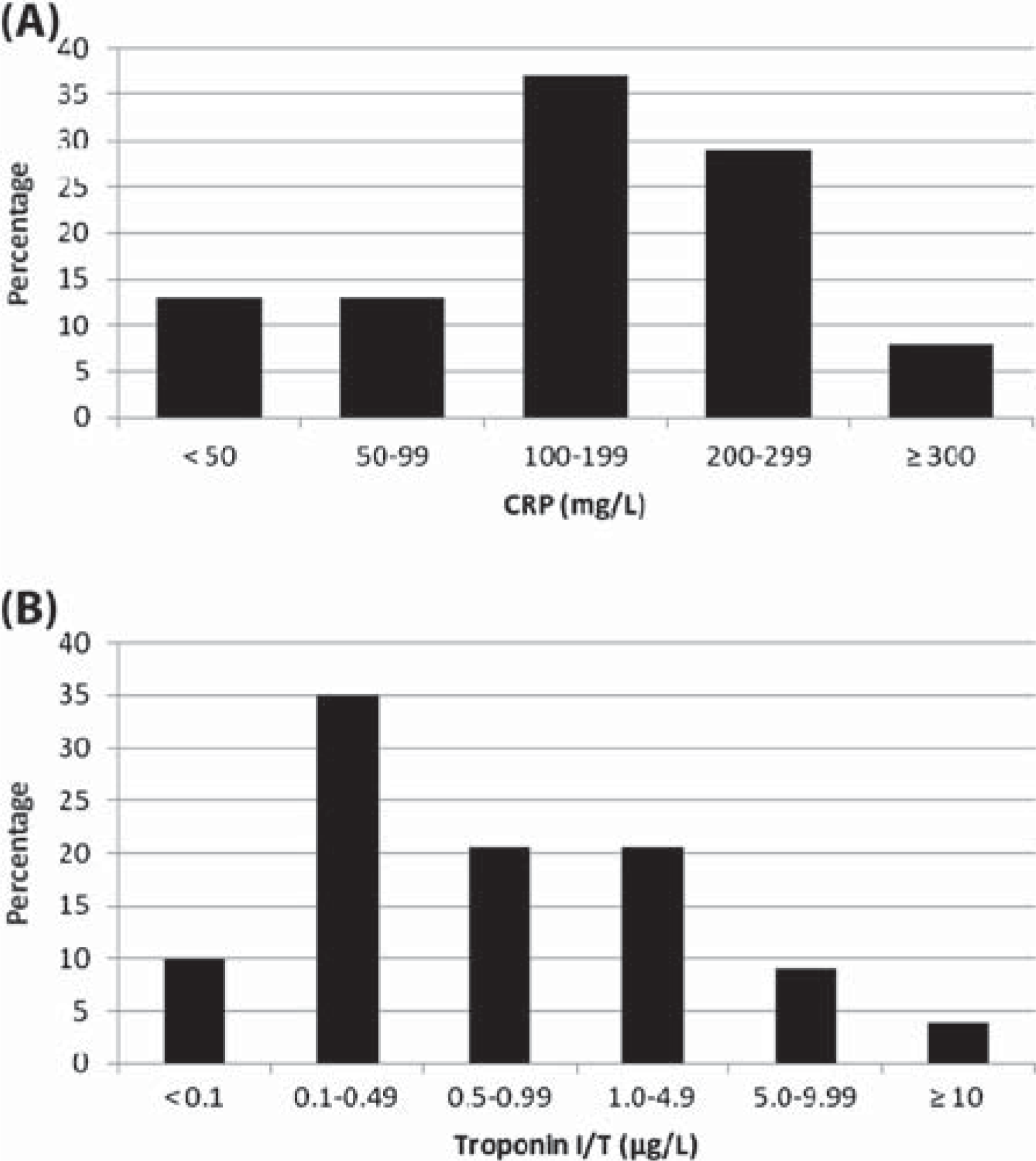
Bar graphs of maximum values recorded in cases for (A) C-reactive protein (CRP) (n = 52) and (B) troponin I/T (n = 68). Upper limits of normal for troponin I/T were 0.03–0.6μg/L and for CRP were 5–10 mg/L.
Demographic characteristics of 75 cases and 94 controls
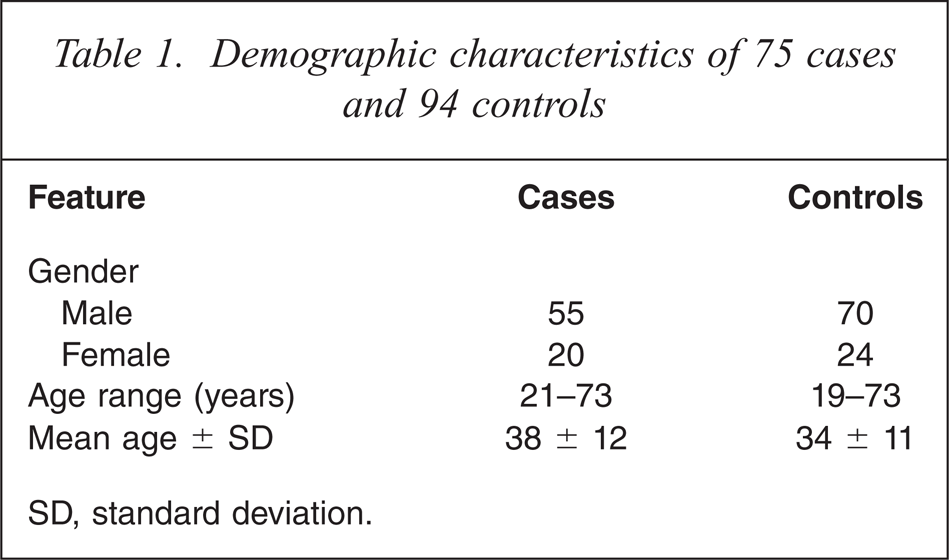
SD, standard deviation.
The typical clinical course of myocarditis
The typical evolution of clozapine-induced myocarditis is presented in Figure 4. The first indications of the onset of myocarditis are non-specific symptoms of illness such as fever associated with features in common with influenza, but symptoms may include severe diarrhoea and vomiting or dysuria. These occur at point 2 in Figure 4. CRP usually begins to increase around this time.
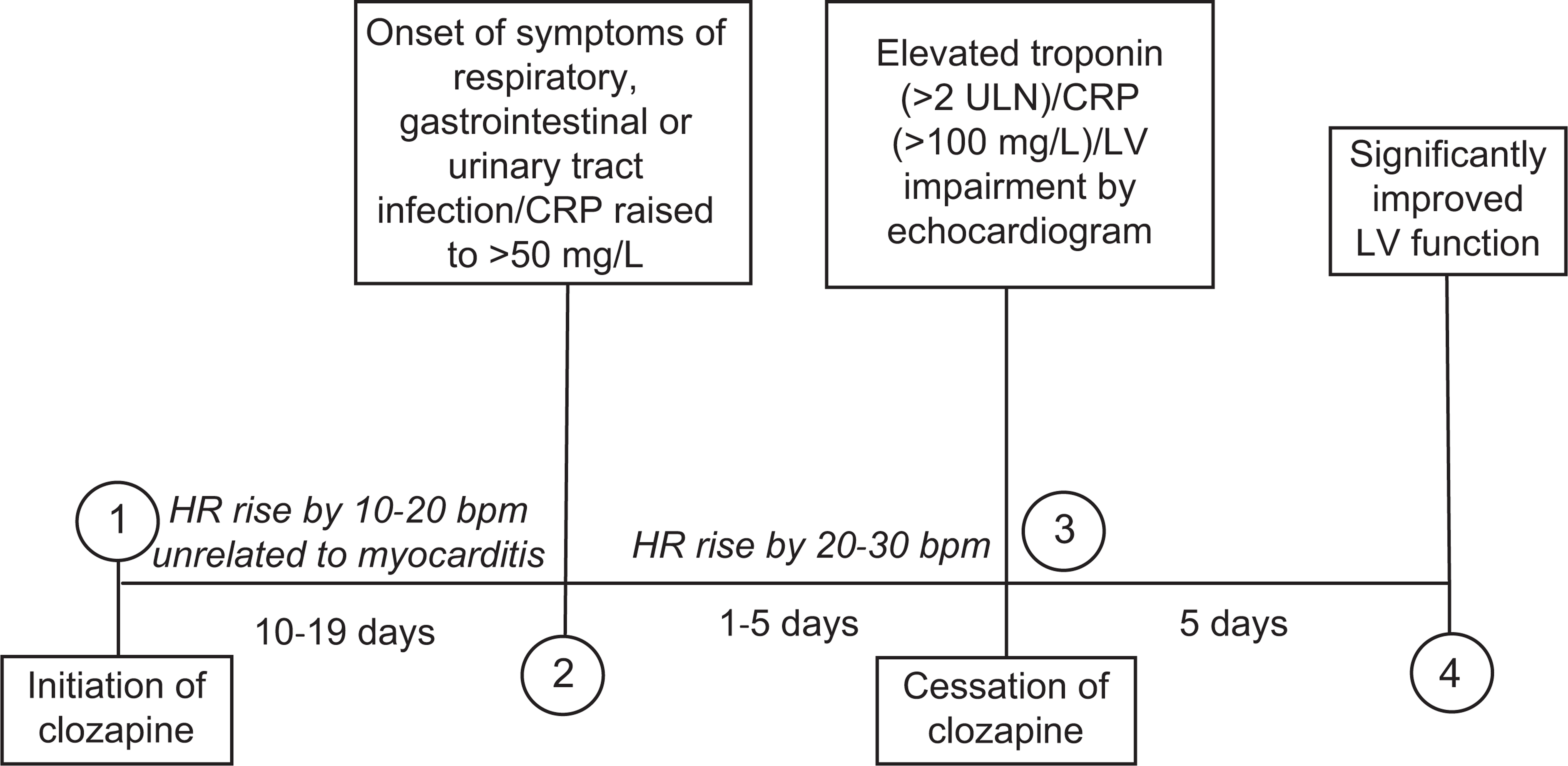
The typical evolution of clozapine-induced myocarditis. bpm, beats per minute; CRP, C-reactive protein; HR, heart rate; LV, left ventricular; ULN, upper limit of normal.
Troponin I or T typically increases (point 3, Figure 4) with a delay of as much as 5 days after both the onset of symptoms and commencement in the rise of CRP. A sudden drop in systolic blood pressure may occur around this time and the patient may report chest pain. The first appearance of non-specific electrocardiogram (ECG) changes also occurs at this point. Basal crepitations, third heart sounds, peripheral oedema and raised jugular venous pressure also may develop at point 3.
Approximately two thirds of cases have left ventricular impairment by echocardiography. Left ventricular function recovers rapidly on withdrawal of clozapine and significant improvement is usually observed within 5 days in even severe cases (point 4, Figure 4).
Heart rate typically increases a few days following initiation of clozapine in all patients including those not developing myocarditis. It may increase again with the onset of fever and elevation in CRP (point 2, Figure 4), or it may suddenly increase with the first development of high troponin (point 3, Figure 4).
Atypical cases of myocarditis
In six instances myocarditis developed without accompanying symptoms. Three of these cases were fatal. For only two of these patients was a CRP result available and in both instances the result taken at point 3 (Figure 4) was less than 30 mg/L.
The three non-fatal asymptomatic cases had increases in heart rate exceeding 20 beats per minute (bpm) occurring at or before point 2 (Figure 4) with a further increment of about 10 bpm around point 3. However, one of the asymptomatic fatal cases had a pulse rate of 90 bpm, which was not greatly increased from baseline, on two occasions on the day before he died overnight in his sleep.
Continuation of clozapine with mild illness
Five clozapine recipients documented as part of this study had transient incremental rises in troponin I or T with or without clinical illness, but continued clozapine without interruption, with reduction in dose or with withholding of 1 or 2 doses only. None sustained any cardiac injury as determined by echocardiography, even though three met the criteria for cases (heart rate >100 bpm and troponin ≥ twice upper limit of normal − ULN).
Proposed monitoring protocol
The proposed monitoring protocol in Figure 5 recommends obtaining baseline troponin I or T, CRP and echocardiography results. Subsequently CRP and troponin should be monitored weekly for the first four weeks of treatment and vital signs observed at least every alternate day taking into account reported symptoms of illness. In the presence of relevant symptoms, an abnormally increased heart rate or raised CRP (>50 mg/L), it is recommended that troponin and CRP be measured daily and the patient monitored for developing illness.
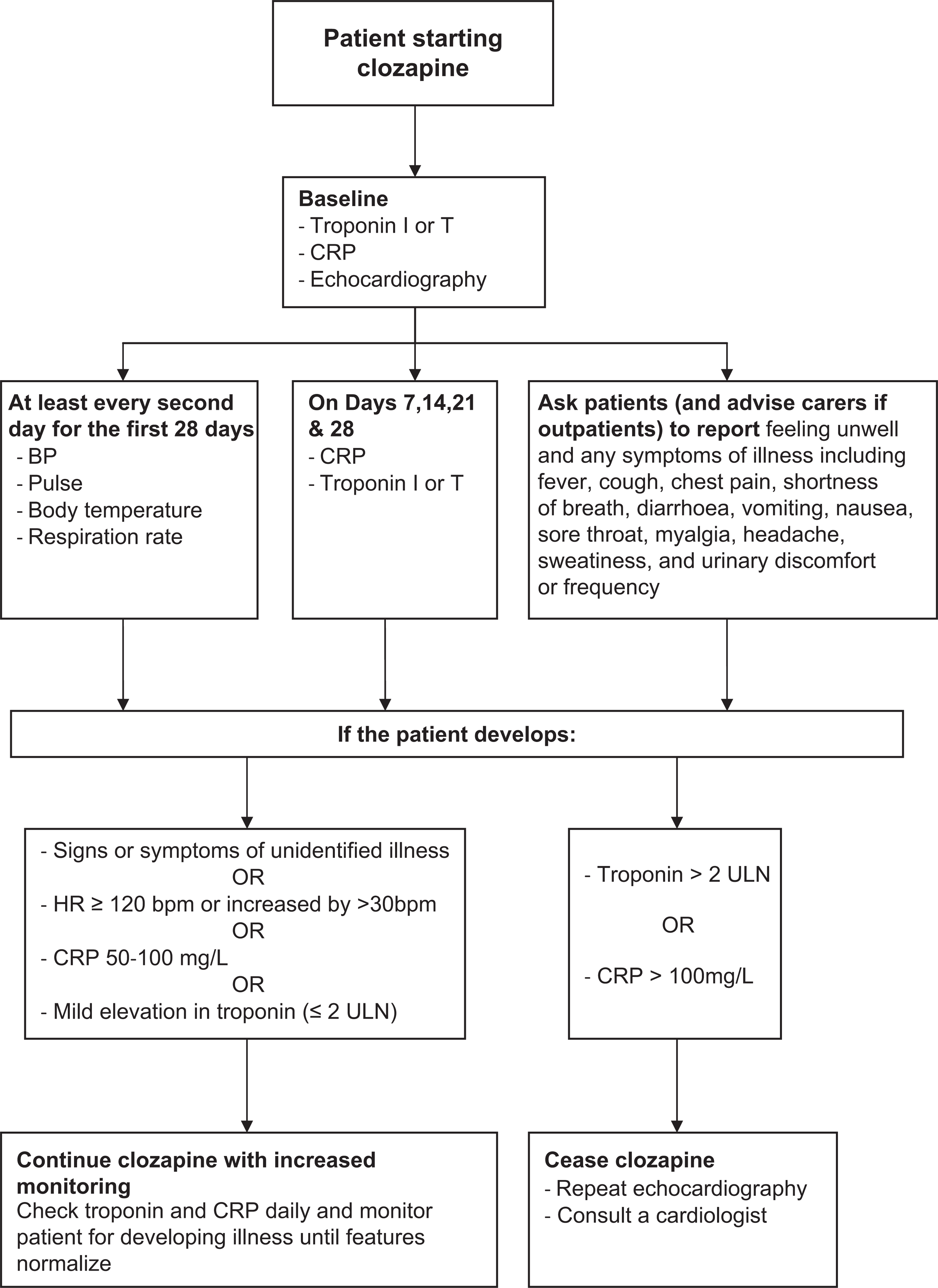
Proposed protocol for monitoring patients commenced on clozapine for clozapine-induced myocarditis. BP, blood pressure; bpm, beats per minute; CRP, C-reactive protein; HR, heart rate; ULN, upper limit of normal.
If troponin levels are only slightly raised (less than twice the upper limit of normal) and CRP remains less than 100 mg/L, clozapine may be continued. Discontinuation of clozapine and investigation by echocardiography is advised if either troponin is in excess of twice the normal maximum or CRP is more than 100 mg/L.
While 90% of cases had troponin results at least twice the upper limit of normal (Figure 2), five cases had CRP >100 mg/L and left ventricular impairment by echocardiography without a clinically significant rise in troponin. Combining troponin (>2 ULN) and CRP (>100mg/L) gives a sensitivity of 100% for the symptomatic cases, but the sensitivity for asymptomatic cases is unknown.
Table 2 outlines the current cardiac monitoring guidelines circulated in Australia by Novartis Pharmaceuticals Australia and Hospira Australia, and Table 3 compares these guidelines with the new monitoring protocol proposed here.
Clozapine cardiac monitoring guidelines circulated by Novartis Pharmaceuticals Australia and Hospira Australia
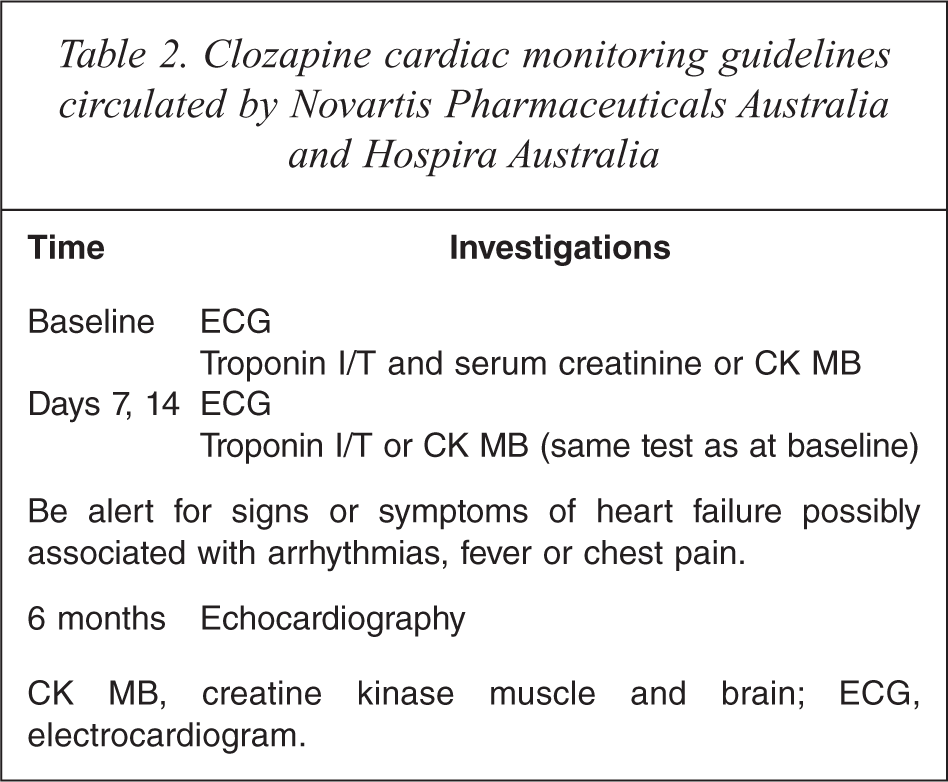
CK MB, creatine kinase muscle and brain; ECG, electrocardiogram.
Comparison between the current cardiac monitoring guidelines and the proposed monitoring protocol
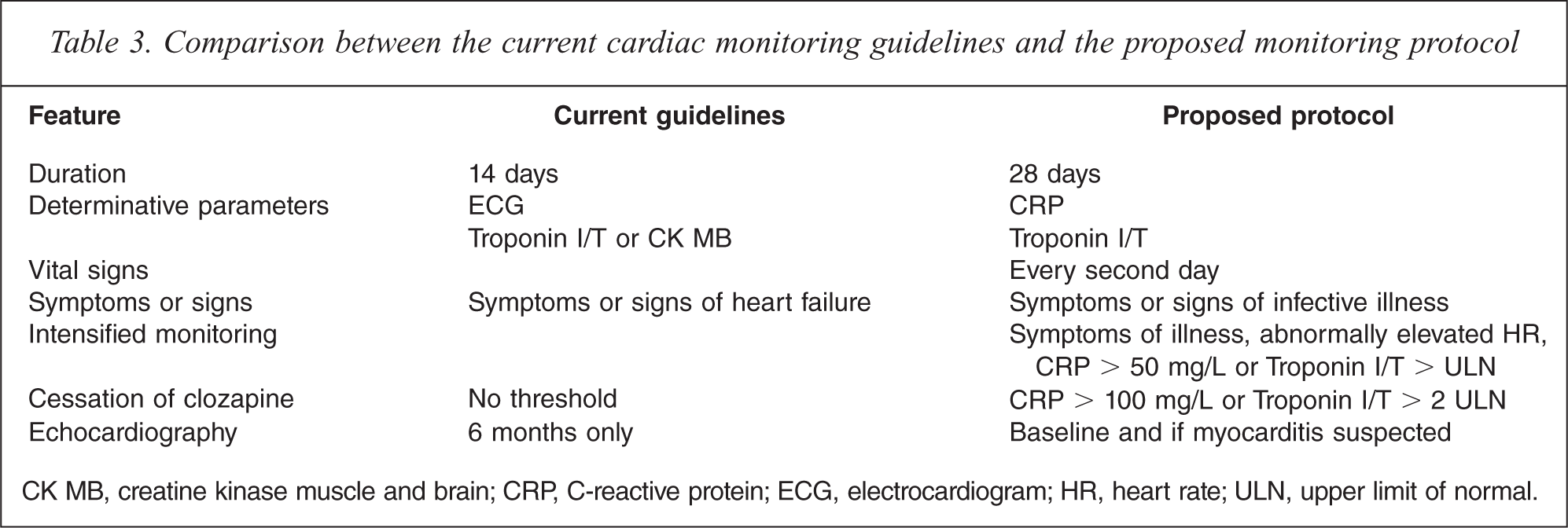
CK MB, creatine kinase muscle and brain; CRP, C-reactive protein; ECG, electrocardiogram; HR, heart rate; ULN, upper limit of normal.
Discussion
Duration of monitoring and protocol sensitivity
The proposed monitoring protocol recommends routine monitoring for myocarditis up to day 28, in comparison to the current guidelines which extend monitoring only to day 14 (Tables 2 and 3). Analysis of the present cases indicates that the mode for occurrence of clozapine-induced myocarditis is week 3, and that late occurring cases are frequently fatal. Hence, it is essential to extend the monitoring to day 28 in order to capture late developing cases early before they become serious and to prevent fatalities from myocarditis.
In Australia and New Zealand where routine weekly blood monitoring for the first 18 weeks of clozapine is mandatory, adding two extra parameters during the first four weeks to those already being investigated will contribute little inconvenience to patient and clinical staff. The added cost will be insignificant in comparison to the gain of preventing serious illness and fatality. It could be argued that given the high proportion of cases occurring in week 3, it would be worthwhile also to check troponin and CRP on day 17. In view of the limited data concerning the effect on CRP of developing asymptomatic myocarditis (occurring in 8% of the present cases), this may enhance the sensitivity of the monitoring programme. However, there are no data to indicate that a higher frequency of surveillance over this period would prevent serious illness and fatality. In part we have chosen to take a pragmatic course and recommend a monitoring programme that is likely to receive widespread clinical acceptance and not impose an excessive burden. While we would present no argument against someone wishing to add routine investigations on day 17, given that CRP appears to be an early indicator of the onset of myocarditis (at least in the symptomatic cases), and that the protocol recommends more intensive monitoring if CRP is raised, heart rate increases or symptoms of illness develop, we consider that the proposed monitoring regime will have sufficient sensitivity to pick up all symptomatic cases of myocarditis developing between days 14 and 21.
With regard to the fatal cases [3], the expectation is that death would have been prevented if clozapine had been stopped earlier and/or the patient had received adequate treatment such as therapy with an ACE-inhibitor or β-blocker or measures to support cardiac and respiratory function had been implemented. However, data to support this view are not available, and for some asymptomatic cases even assiduous monitoring may not provide sufficient warning of cardiac damage to prevent death.
Continuation of clozapine with mild disease
The proposed monitoring protocol suggests that continuation of clozapine may be contemplated if troponin I or T is no more than twice the upper limit of normal, provided CRP is less than 100 mg/L. This is based on the observation that five individuals with slightly raised troponin documented for this study continued clozapine without sustaining cardiac injury. Consistent with our data, Hill et al. [4], reporting 25 cases of clozapine-induced myocarditis occurring in New Zealand, indicated that two cases recovered despite ongoing clozapine.
Considering the morbidity associated with inadequately controlled schizophrenia, and that those prescribed clozapine have been unsuccessfully treated with multiple other antipsychotics, defining a low level of possible myocarditis for which discontinuation of clozapine is not essential may offer long-term benefit to some patients. Added to this are the data indicating that clozapine is not only the most efficacious antipsychotic [5], but also the safest when measured using mortality and follow up over 11 years [6]. For these reasons every opportunity for continuation of clozapine should be grasped provided it can occur safely.
Heart rate as an indicator of myocarditis
The role of monitoring heart rate as a means to early identification of the onset of clozapine-induced myocarditis is difficult to define, since clozapine frequently causes benign tachycardia [7], and tachycardia may be a feature of the underlying illness, particularly when the patient is anxious or is a heavy smoker. The comparisons of the frequency of heart rates of more than 100 bpm and 120 bpm or more among cases and controls in Figure 2 illustrate these points. Nevertheless, monitoring heart rate on at least alternate days from baseline will mean that trends and tendencies for the individual patient can be identified and an abnormal increase associated with the onset of myocarditis is more likely to be correctly interpreted.
C-reactive protein in early diagnosis
The proposed monitoring protocol includes CRP along with troponin measurements in the routine monitoring for myocarditis. Data from the present study indicate that CRP may be an early marker for the onset of myocarditis. CRP is a non-specific marker of inflammation. Reviews of myocarditis by Brady et al. [8] and Batra and Lewis [9] list raised CRP as a diagnostic indicator for the presence of myocarditis and Lee et al. [10] and Kato et al. [11] associate higher values with a fulminant myocarditis. From our data, the use of CRP (>100 mg/L) in combination with troponin (> 2ULN) in a monitoring tool for clozapine-induced myocarditis yields a sensitivity of 100% for symptomatic cases. We propose that monitoring CRP may provide an early warning of the development of myocarditis caused by clozapine, where CRP of more than 50 mg/L foreshadows the onset. Nevertheless, the value of CRP in this role has not yet been fully elucidated. It is unknown how reliably it is raised to more than 50 mg/L before troponin increases in symptomatic cases and how many days notice of impending cardiac injury it gives. In addition, it may be that CRP in asymptomatic cases typically does not reach 50 mg/L.
Echocardiography and ECG
The monitoring guidelines do not recommend using ECG as a means of detecting the development of myocarditis. Cooper has described the sensitivity of ECG for myocarditis as low [12]. However, clinicians may choose to monitor heart rate by ECG and may find diagnostic benefit in monitoring the evolving ECG changes.
In order to use echocardiography as a diagnostic tool in suspected myocarditis, baseline investigation is advisable to exclude pre-existing dysfunction. Investigation for clozapine-induced impairment should be conducted promptly following withdrawal of clozapine when myocarditis is suspected. If no significant impairment of cardiac function is measured, no specific therapy apart from cessation of clozapine is required. However, where echocardiography reveals moderate or severe left ventricular impairment an assessment can be made of the need for drug or mechanical intervention.
Eosinophilia
Eosinophilia is typically regarded as a diagnostic feature of hypersensitivity myocarditis, such as that caused by clozapine [13,14]. However, in one report of three cases of myocarditis with suspected drug cause, two patients had eosinophil counts of zero [15]. We have found that for clozapine-induced myocarditis some cases have very low eosinophil counts (<0.1 × 109/L) and that where eosinophilia occurs the onset is usually late, after the acute assault on the heart as measured by the cardiac enzymes troponin I or T [2]. Hence, raised eosinophils should not be used to monitor for myocarditis occurring following clozapine initiation.
Conclusion
The monitoring protocol that we have proposed is based on an analysis of 75 cases, for which the data were collected systematically from the patient files. Comparison with systematically collected data for 94 control patients who took clozapine without developing myocarditis means that the relative diagnostic importance of some parameters could be determined.
The proposed evidence-based protocol for monitoring for clozapine-induced myocarditis involves active monitoring for four weeks and incorporates CRP along with troponin, vital signs and symptoms of illness. We suggest that continuation of clozapine may be contemplated if rises in troponin and CRP are mild, but that clozapine should be discontinued in the presence of indications of more severe illness (troponin I/T >2 ULN, CRP >100 mg/L). While observing this protocol will optimize the ascertainment of clozapine-induced myocarditis, as our data indicate, this illness is varied in its presentation and complete case-ascertainment cannot be guaranteed, especially not for asymptomatic cases. Nevertheless, in the context of mandatory weekly blood monitoring, application of this protocol will improve the safety of clozapine initiation without adding greatly to cost.
Footnotes
Acknowledgements
The assistance of Sally Wilson in documenting some of the control patients is gratefully acknowledged. In addition, the authors are grateful for the assistance of individuals at each of the following health services: from Victoria, Austin Health, Barwon Health, Bayside Health, Bendigo Health, Eastern Health, North West Mental Health, Peninsula Health, Southern Health, St Vincent's Health, Thomas Embling Hospital, and Werribee Mercy Mental Health Service; from New South Wales, Northern Sydney Central Coast Health, Sydney South West Area Health Service, Wandene Private Hospital and private psychiatrist Chris McDowell; and from Queensland, Prince Charles Hospital.
