Abstract
Clozapine is an antipsychotic drug which has been described as atypical because there is a much lower incidence of extrapyramidal side-effects associated with its use, and it is effective in treating up to 60% of patients who do not respond to other drugs. Determining the most suitable dose, however, is not straightforward. There appears to be little relationship between dose and therapeutic effect [1], so many clinicians use plasma-clozapine levels to help them determine the optimal dose that maximizes benefit without the development of adverse reactions. A moderate correlation between plasma levels and therapeutic effect has been reported [2–4], but plasma levels of clozapine after dosing can vary widely. Plasma concentrations 12 h after doses of 100 mg up to 500 mg range from 60 to 600 ng/mL [5, 6]. Many investigators have tried to determine a threshold plasma clozapine level [7]. No absolute threshold has been found, but the evidence suggests that if a patient is not responding to clozapine his or her dose should be increased, sideeffects permitting, to obtain plasma levels of atleast 370–420 ng/mL. Factors which affect plasma clozapine levels include differences in individual metabolism, induction or inhibition of cytochrome P450, drug interactions, smoking, age and gender [3], although the evidence for the last three is not compelling [2, 8, 9].
There are several side-effects associated with clozapine treatment, not all of which are dose related. The occurrence of life-threatening agranulocytosis, with an incidence of 0.8–0.9% [10–12], is not dose-dependent, nor are the less severe side-effects such as sialorrhoea, tachycardia, nausea, enuresis, diaphoresis, constipation and weight gain [13, 14]. Some side-effects, however, such as the degree of sedation [13] and severity of hypotension [6] are dose-dependent. The risk of developing seizures also increases with increasingdose [15, 16], but it has been postulated that this may be related to the rate of titration of clozapine dosing [17].
There are many reasons for attending psychiatrists wishing to know their patients' plasma clozapine levels. Measurement of plasma clozapine levels is of potential benefit when a patient fails to respond to what are usually adequate doses, or is responding but experiencing significant adverse reactions. Other instances of potential benefit involve circumstances where patients have altered pharmacokinetics, due to a disease process (especially hepatic pathology), concomitant administration of drugs affecting clozapine metabolism, advanced age, or as confirmation of a suspicion of poor compliance.
We are aware of 10 laboratories throughout Australia and New Zealand that perform these assays, but to date no data have been published by an independent body investigating intra- and interlaboratory variability. Variations in the techniques employed to determine plasma clozapine levels [16–18] make accurate analysis and comparison of data difficult. Current techniques utilize either highperformance liquid chromatography (HPLC) and ultraviolet detection or gas-liquid chromatography (GLC) and nitrogen detection to measure plasma clozapine concentrations.
A case cannot be made for using plasma clozapine levels in clinical evaluation unless the accuracy and reliability of measuring these levels are assured. Although there have been overseas reports of a high level of correlation in the measurement of split samples sent to reference, research and clinical laboratories for performance testing [19, 20], this is not universally the case [8]. The present study is the first that sets out primarily to determine the reliability of plasma clozapine assays by comparing the results of the 10 laboratories in Australia and New Zealand that perform clozapine assays against each other and against the concentration of samples as prepared.
Method
Preparation of samples
Phase I: initial determination of drug levels
A solution of clozapine (chloride salt MW = 326.8; Sigma, St Louis, MO, USA) was prepared by dissolving 100 mg of clozapine in 10 mL methanol. This was further diluted 1 in 100 in phosphate buffered solution (PBS) to produce a stock solution of concentration 10 μg/mL. The stock solution was used to prepare three samples of clozapine in PBS at concentrations of 140 ng/mL, 310 ng/mL and 580 ng/mL. These were marked sample A, sample B and sample C, respectively, and despatched to an arbitrarily selected laboratory to determine whether an assay would be returned corresponding to the prepared concentrations. The laboratory performed three separate assays in triplicate on each sample.
Phase II: assay of split sample
Following this, 100 mL volumes of samples A, B and C were prepared at the same concentrations as in phase I (140, 310and 580 ng/mL) in human plasma (obtained frozen from Australian Red Cross Blood Bank). These were divided into 10 × 10 mL aliquots, frozen at −20°C, packed in dry ice and despatched by courier tothe 10 laboratories in Australia and New Zealand.
As soon as the data were analysed, each centre was advised of the overall results and the relationship between that laboratory's results and the pooled data.
Questionnaire
In an attempt to account for any differences that might occur a questionnaire was sent with the samples to each laboratory to determine the procedures used, the state of the samples on arrival, the methods of analysis and other relevant data.
Phase III: follow-up study
Six months after the participating laboratories had been advised of the results of phase II, a second set of samples was despatched to each laboratory. The concentrations of these samples were: sample A = 160 ng/mL; sample B = 380 ng/mL and sample C = 640 ng/mL of clozapine in human plasma. To investigate the stability of clozapine we prepared samples made up in purified water at the same concentrations. These were designated samples D (160 ng/mL), E (380 ng/mL) and F (640 ng/mL). A fresh stock solution of clozapine was used. All the samples were frozen immediately after being prepared and were despatched in dry ice as was done in phase II.
Lyophilized samples
To determine if clozapine would be more stable if dry, one set of samples was freeze-dried overnight on the night of preparation and despatched in dry ice to one of the participating laboratories for analysis.
Stock from phase II
We also wished to investigate the stability of clozapine solutions over a period of time. Samples of clozapine 640 ng/mL, one in human plasma and one in purified water, were made up from the stock solution prepared in phase II 6 months earlier. These were also despatched in dry ice to one of the participating laboratories for analysis.
Analysis of results
The results were subject to analysis of variance (ANOVA)with ‘sample concentration’ and ‘laboratory’ as two fixed factors. Laboratory was treated as a fixed rather than a random factor because the laboratories involved constitute the population of the laboratories of interest. Post-hoc comparisons were undertaken using the conservative Schiffé method. Confidence limits of the median of 95% were determined using the method described by Marascuilo and McSweeney [21].
Results
Phase I
The results returned from the initial assay were: solution A (140 ng/mL) = 120 ± 60 ng/mL; solution B (310 ng/mL) = 300 ± 40 ng/mL and solution C (580 ng/mL) = 490 ± 90 ng/mL (the figures in parentheses are the concentrations of the prepared solutions). The concentrations of the prepared solutions were embraced by the 95% confidence limits of the returned data for each sample.
Phase II
Laboratory A performed only one run of each sample in triplicate. Laboratory C performed three runs on each sample with a single assay on each run. This is taken into account in the analysis of the results. All but one of the laboratories received the samples in a frozen state.
Comparison with nominal concentrations of samples
Figure 1 shows the median value for each centre as well as the overall median with 95% confidence limits for each sample. The median plasma level determined for each sample was below the nominal concentration of the prepared solutions. The coefficient of variation for each sample for each centre was less than 0.2 in each case, and less than 0.1 in most cases. The lowest coefficient of variation was 0.01 and the highest 0.15.
Histograms showing the median value of clozapine concentration in plasma for each centre and the overall (OA) median with 95% confidence limits for sample A (a), sample B (b) and sample C (c) in phase II. The solid line indicates the nominal concentration of each sample.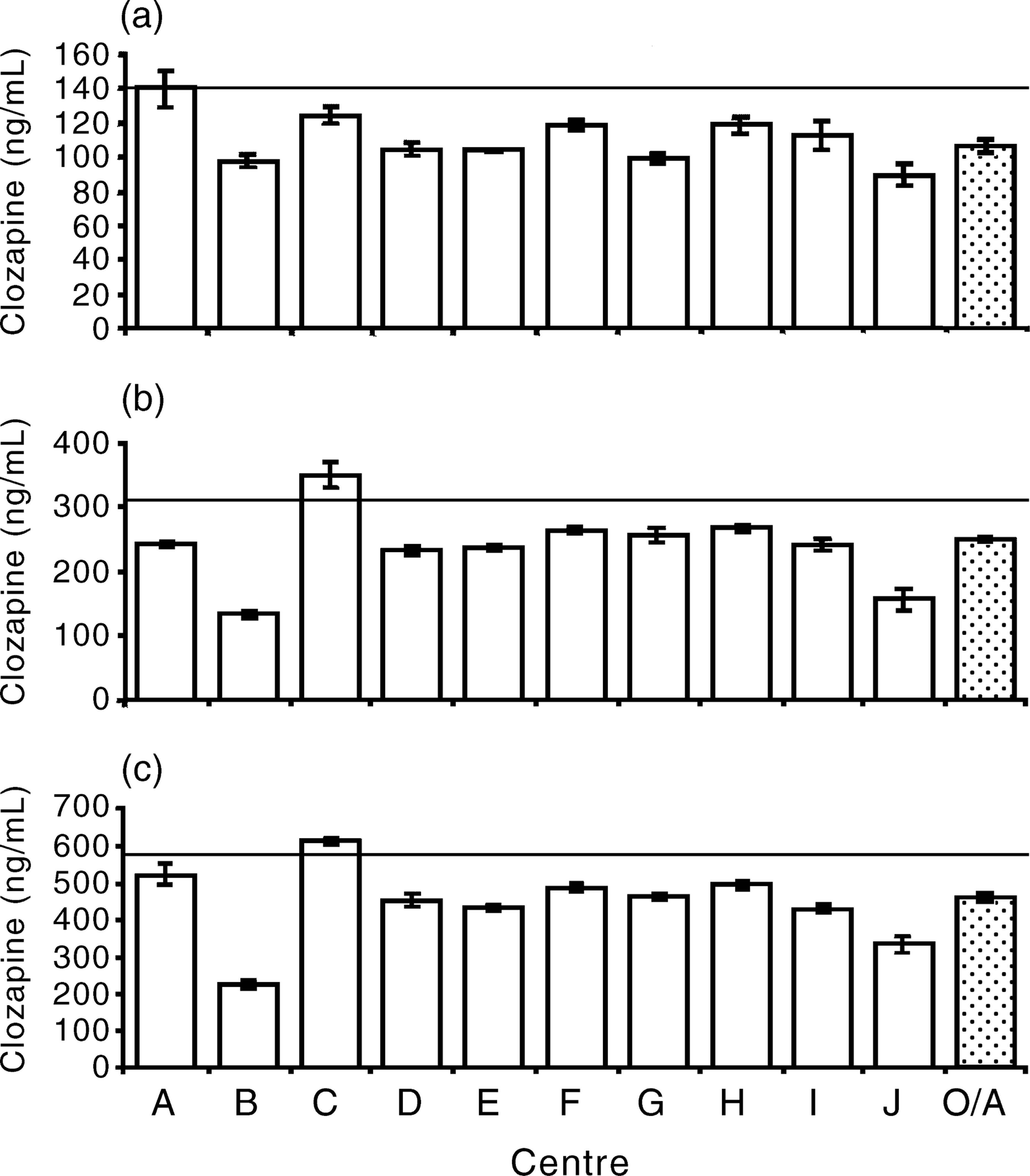
Comparison with other laboratories
Post-hoc comparisons were undertaken between sites using the conservative Schiffé method. The results of these revealed three outlying centres and a scattered pattern of relatively small differences. Centres B and J have significantly and substantially greater errors than any other centre. Centre C was the only one to overestimate the plasma concentration, although the magnitude of the error was substantially less than any other site.
Questionnaire
All the laboratories used high performance liquid chromatography (HPLC) and UV detection except laboratory B, which used gas chromatography and thermonic selective detection. The choice of wavelength for UV detection varied between laboratories. Laboratory E was the only one to receive the samples in an unfrozen state. Each laboratory used a different solvent process and a different internal standard.
Phase III
Centres B and I performed only one run in triplicate for each sample while centre C performed two runs in triplicate. Centre E did not participate in phase III and centre J assayed the samples dissolved in human plasma (samples A, B and C), but not those made up in purified water (samples D, E and F).
Clozapine in human plasma
Figure 2 shows the median value for each centre as well the overall median with 95% confidence limits for each sample. Centre A returned results for sample A that encompassed the nominal concentration of the prepared solution. One laboratory's results (D) were significantly higher than the nominal concentration and all the other results were significantly lower. For sample B all the results were significantly lower than the nominal concentration, as were all the results for sample C. The median values for all the samples were significantly lower than the nominal concentrations. There was no difference in accuracy between centres in determining the concentration of clozapine in human plasma over the range of concentrations, although there were some individual differences for each concentration. Post-hoc comparisons were undertaken between centres using the conservative Schiffé method. There were no differences between any of the centres. The coefficients of variation for each sample for each centre were below 0.1 in all but two cases for the plasma samples, with a rangeof 0.01–0.16. Less accuracy was shown for samples made up in water, with coefficients of variation ranging from 0.01 to 0.26.
Histograms showing the median value of clozapine concentration in plasma for each centre and the overall (OA) median with 95% confidence limits for sample A (a), sample B (b) and sample C (c) in phase III. The solid line indicates the nominal concentration of each sample.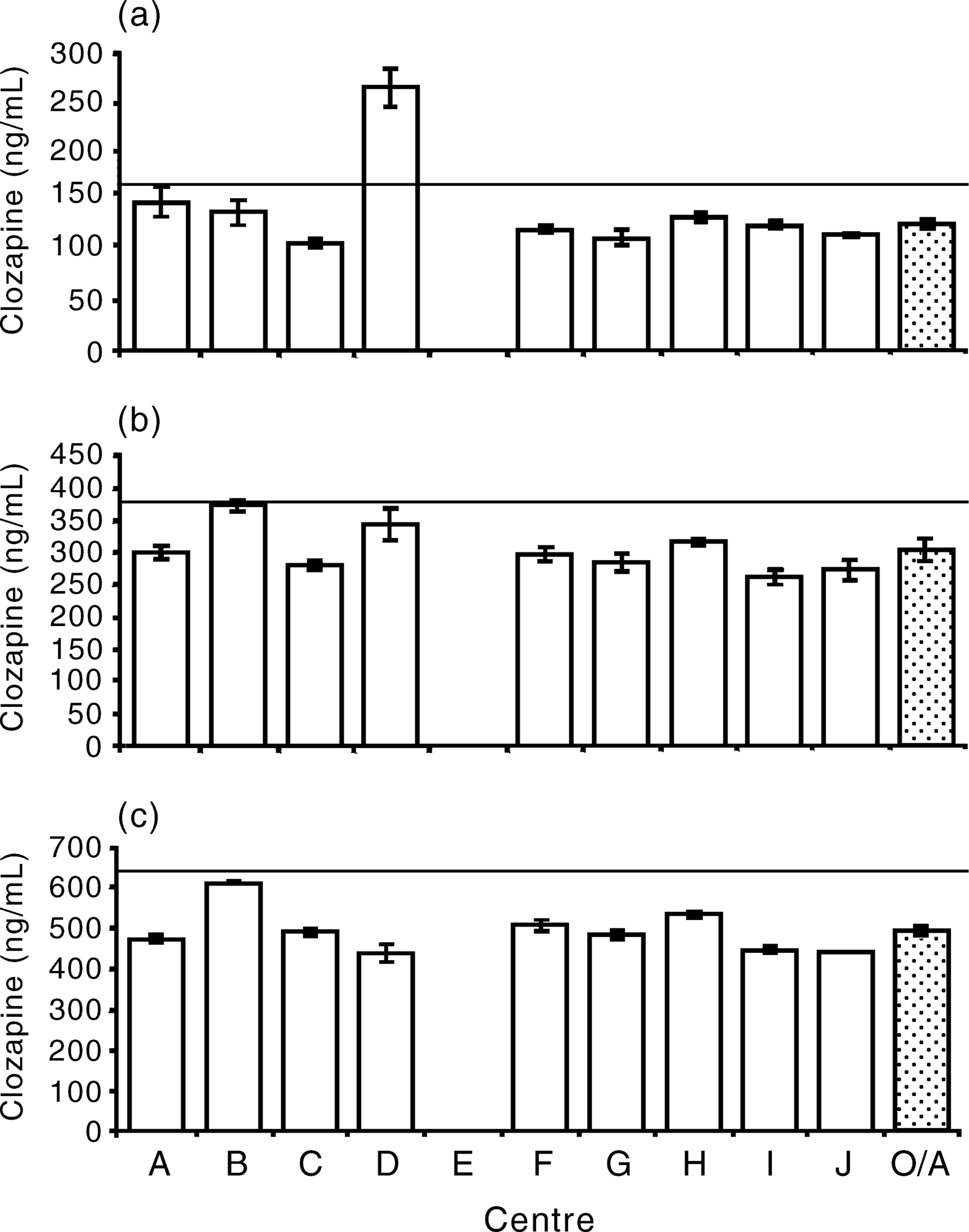
Clozapine in purified water
All the results returned for the three samples of clozapine dissolved in water (samples D, E and F, Fig. 3) were significantly lower than the nominal concentrations. The medians of the pooled results for each sample were substantially lower than for samples A, B and C.
Histograms showing the median value of clozapine concentration in plasma for each centre and the overall (OA) median with 95% confidence limits for sample D (a), sample E (b) and sample F (c) in phase III.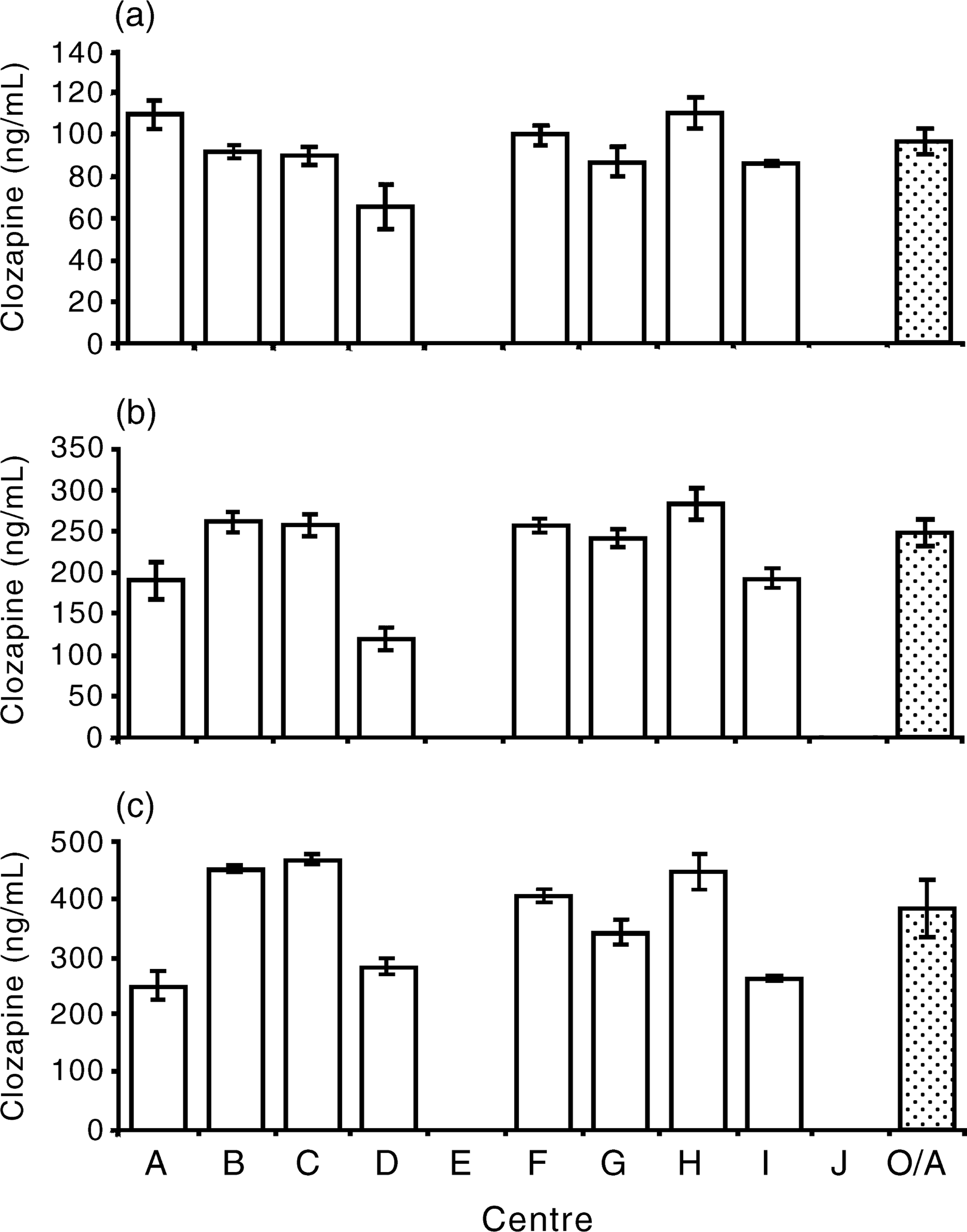
Freeze-dried samples
The results of the assays of the freeze-dried samples were not significantly different from the medians of the unprocessed samples. Only one assay was performed on each sample (results not shown).
Stock solutions made in phase II
The results for the samples prepared from the stock solutions made for phase II were 939 ± 376 ng/mL (mean ± 2 SD, n = 9) for plasma and 659 ± 106 ng/mL (mean ± 2 SD, n = 8) for water (nominalconcentration = 640 ng/mL for each sample). These results are significantly higher than the means of the results returned from the same laboratory for the samples made from freshly prepared stock (results not shown).
Discussion
The three areas of interest to this study are (i) the precision of each laboratory; (ii) the interlaboratory variation; and (iii) the results returned by each centre against the nominal concentration of the prepared samples. The last is most relevant for sample B as this concentration represents most closely the therapeutic range of plasma clozapine levels.
Precision
The greatest individual variation for samples made up in plasma was ± 10% (95% confidence limits), with the average variation being around 5%. The narrow rangeof 95% confidence limits is evidence that each laboratory gives consistent results over a period of time and that different operators do not generate different results.
Interlaboratory variation
The narrow range for the overall 95% confidence limits (about 4% for each sample) indicates that there is little interlaboratory variation. In phase II, centres B, D and J were significantly less precise than the other centres over the three samples. In phase III, however, the only result that was significantly different from the others was centre D, sample A. While this appears to be an anomalous result, it shows that using only one reading from one laboratory is not necessarily accurate, albeit that the result in this case was for a concentration lower than generally accepted therapeutic levels.
Overall, however, it would appear that a plasma sample from a given patient being treated with clozapine will produce a similar result at any of the centres in Australia and New Zealand that perform plasma clozapine assays.
Accuracy
In both phases II and III the median result for each sample was substantially lower than the nominal concentration of the prepared sample. The high interlaboratory concordance suggests that this is not due to inaccurate reading of the sample which is injected into the chromatograph at each centre. Discussion with each laboratory indicates that their internal standards are chosen carefully and in-house quality assessment procedures are carried out regularly. Possible explanations are (i) the clozapine in plasma was degraded between preparation and assay; (ii) the drug is adsorbed on to the surface of the container; (iii) errors were made in preparing the samples; or (iv) variations in extraction or chromatograph technique.
To examine the possibility of degradation in transit two variations were made to the procedure. One was to make up the samples in purified water instead of human plasma. This was done to avoid possible metabolism of the drug by enzymes present in the plasma. The other was to freeze-dry the samples and have the centre reconstitute them immediately before assay. The results for the samples prepared in purified water were universally lower than for those made up in plasma. Clozapine is more stable in plasma than in water, indicating a fairly rapid hydrolysis as the samples were frozen for most of the time between preparation and assay. Alternatively, the different results may be due to the different speeds at which the liquid phase passes through the column or a difference in binding to the column. Whatever the reason, it appears that clozapine is not degraded in frozen human plasma. The results of the assays of the freeze-dried samples were not significantly different from those for the unprocessed samples. Although this assay was performed by only one centre it suggests that clozapine does not degrade frozen in human plasma over a few days. This is corroborated by other researchers who have stored plasma containing clozapine frozen or refrigerated over several weeks and report no degradation [5, 14, 17].
Extreme care was taken in preparation of all the solutions. While it is possible that in spite of this an error may have been made in preparing one sample, it is unlikely that an error could have been made in preparing all of them. It is noteworthy that the samples prepared from the stock solution made for phase II yielded results much higher than those prepared from fresh stock. In fact, the sample prepared in human plasma was assayed as being significantly higher than the nominal concentration. This is further evidence against the possible degradation of clozapine. The possibility that clozapine degrades into a substance that gives a false reading on a HPLC, however, cannot be ignored. It has been reported to us anecdotally that clozapine is absorbed by certain plastics, but we have found no published information to support this. The tubes we used were made of polystyrene. In the next programme each centre will receive samples in containers of various materials to test this hypothesis.
The assay itself can also provide undetermined sources of error. The extraction process is about 85–95%efficient [22]. One centre (name withheld) rechecked recoveries compared with its unprocessed neat standards and found them to be about 70%.
General considerations
We have previously reported that there is a case for increasing the dose of clozapine in non-responsive patients to levels above 350–420 ng/mL, within the maximum allowable dose of 900 mg daily [7]. In using plasma clozapine levels many factors must be considered. Levels can vary due to drug-related cytochrome P450 induction or inhibition, smoking, drug interaction and age [7]. It is also important to allow steady-state conditions to be established, which takes 5–7 days [2, 18], and adequate time for tissue distribution, usually 12 h after the last dose [2, 8, 9] must be allowed to avoid inappropriately high readings.
Conclusion
Plasma clozapine levels can be useful in determining the optimal dose for each patient. Clinicians in Australia and New Zealand who wish to know their patients' plasma clozapine levels can be confident that the result of the assay is unlikely to vary with their choice of centre or the operator who performs the assay.
