Abstract
It is now well known that schizophrenia involves pervasive impairments in cognitive functions, with up to 98% of patients displaying significant cognitive decline relative to predicted premorbid levels [1]. Verbal memory is one of the most severely impaired and most functionally limiting domains in terms of real-world everyday living [2]. The magnitude and extent of memory impairment has been demonstrated by a meta-analysis of more than 70 studies [3], where deficits were independent of medication status, severity of psychopathology, duration of illness and frontal attentional dysfunction.
Cognitive deficits, including verbal memory, are also commonly present from the first episode of psychosis (FEP) [4], and have been found in individuals with a clinically defined at-risk mental state (ARMS) for psychosis [5,6,7]. The presence of childhood verbal memory deficits in the offspring of schizophrenia patients accurately identified 83% of individuals who later developed schizophrenia-related psychoses [8]. The Edinburgh High-Risk Study showed performance on the Rey Auditory Verbal Learning Test (RAVLT) was also a significant predictor of transition to schizophrenia [9]. Since then, accumulating evidence has led to the implication that verbal episodic memory deficits, present during the at-risk stage, are possible vulnerability markers for psychosis [6,10].
The pattern of memory impairment in schizophrenia has been noted to be similar to that found in patients with medial temporal lobe (MTL) lesions [11]. MTL structural abnormalities in schizophrenia are well documented [12], specifically with reduced hippocampal volume now being recognized as one of the most common neuroanatomical abnormalities in this disorder [13]. In addition, positron emission tomography (PET) and functional magnetic resonance imaging (fMRI) studies have shown verbal episodic memory to be specifically linked to abnormal hippocampal activation, via word list and paired associate learning tasks [14–16]. Mounting evidence of impaired encoding and contextual binding memory processes, and more severe explicit rather than implicit memory deficits further supports the link between hippocampal dysfunction and schizophrenia pathophysiology [17].
Hippocampal volume reductions have also been observed in FEP patients [18], although findings are less consistent with some evidence of volumes being similar to that of controls [19,20]. This may be due to several factors. Degree of structural change may be dependent on stage of psychosis, as Velakoulis and colleagues [21] found FEP patients to demonstrate reduced left hippocampal volumes, whereas chronic patients typically have bilateral reduction. In comparison, individuals with ARMS (who later converted to psychosis) had similar hippocampal volumes to healthy controls. When these patients were grouped based on diagnosis, volume reductions were restricted to the first episode schizophrenia (FES) group in comparison to the schizophreniform psychosis or other psychoses groups. Findings suggest hippocampal abnormalities may be a distinguishing feature of FES, or potentially appear during a more advanced stage of early psychosis, specifically considering schizophreniform disorder has an illness duration of less than 6 months.
The hippocampal formation has long been acknowledged as having an integral role in contextual binding and episodic memory [22]. Impairments in these processes have been implicated in schizophrenia with regard to hippocampal abnormalities [17]. However, deficits in the specific components of verbal memory in FEP are not well characterized. Relational memory is one component that has been suggested to require MTL structures [23,24], and involves the associative binding of the elements of episodic experience where representations of relations between the constituents of an experience are formed, leading to an enduring memory trace [25]. The aim of the current study was to investigate relational memory in FEP using a verbal paired associate (VPA) memory task, and to determine whether performance was dependent on phase of FEP (i.e. duration of illness less than or greater than 6 months). The task used in the current study is a variant of an experimental test shown to be sensitive to MTL dysfunction [26]. This VPA paradigm uses arbitrary word pairs as opposed to words that are semantically related, which has been previously shown to enhance the specificity for detecting MTL deficits [27,28]. Furthermore, it investigates the effect of interference on associative binding, by adding two interference trials after the initial 4-trial learning and cued recall phase (phase 1). For the interference trials (phase 2) the initial word from the previously learnt pairs in phase 1 (denoted AB pairs) was re-paired with a new word (denoted AC pairs) and learned and recalled, and finally phase 3 tested the impact of interference on initial learning with a single cued recall trial of the original AB pairings.
Methods
Participants
Forty-seven FEP patients were recruited from the Early Psychosis Prevention and Intervention Centre at Orygen Youth Health, an outpatient service in Melbourne (see Table 1 for demographic details), and participated in one of two randomized double-blind clinical trials [29,30]. FEP inclusion criteria were an age at onset of 15–25 years, and current psychosis as reflected by the presence of at least one of (i) delusions, (ii) hallucinations, (iii) disordered thinking/speech, and (iv) disorganized, bizarre or markedly inappropriate behaviour. DSM-IV diagnoses were obtained using the Structured Clinical Interview for DSM-IV [31]. Diagnoses were: schizophrenia (N = 11), schizoaffective disorder (N = 5) and schizophreniform psychosis (N = 31). Sixteen patients were neuroleptic-naïve, but the distribution of cases was not significantly different between the three patient groups (see Table 1). The majority of treated patients were on quetiapine (N = 23), while the remaining were on risperidone (N = 5) or olanzapine (N = 3). The median cumulative chlorpromazine (CPZ) equivalent dose at testing was 376 mg (range 34–3600 mg). We have chosen to report the median here as it is more reflective of the group data, as one participant in the schizophrenia group had received 8 days treatment with quetiapine, resulting in the maximum CPZ equivalent exposure (i.e. 3600 mg), which was more than double that of the next highest cumulative dose (1650 mg). However, medication is unlikely to have affected cognition given treatment was over a limited time period and recent findings suggest quetiapine has minimal effects on cognitive performance even after 6 months treatment [32].
Demographics
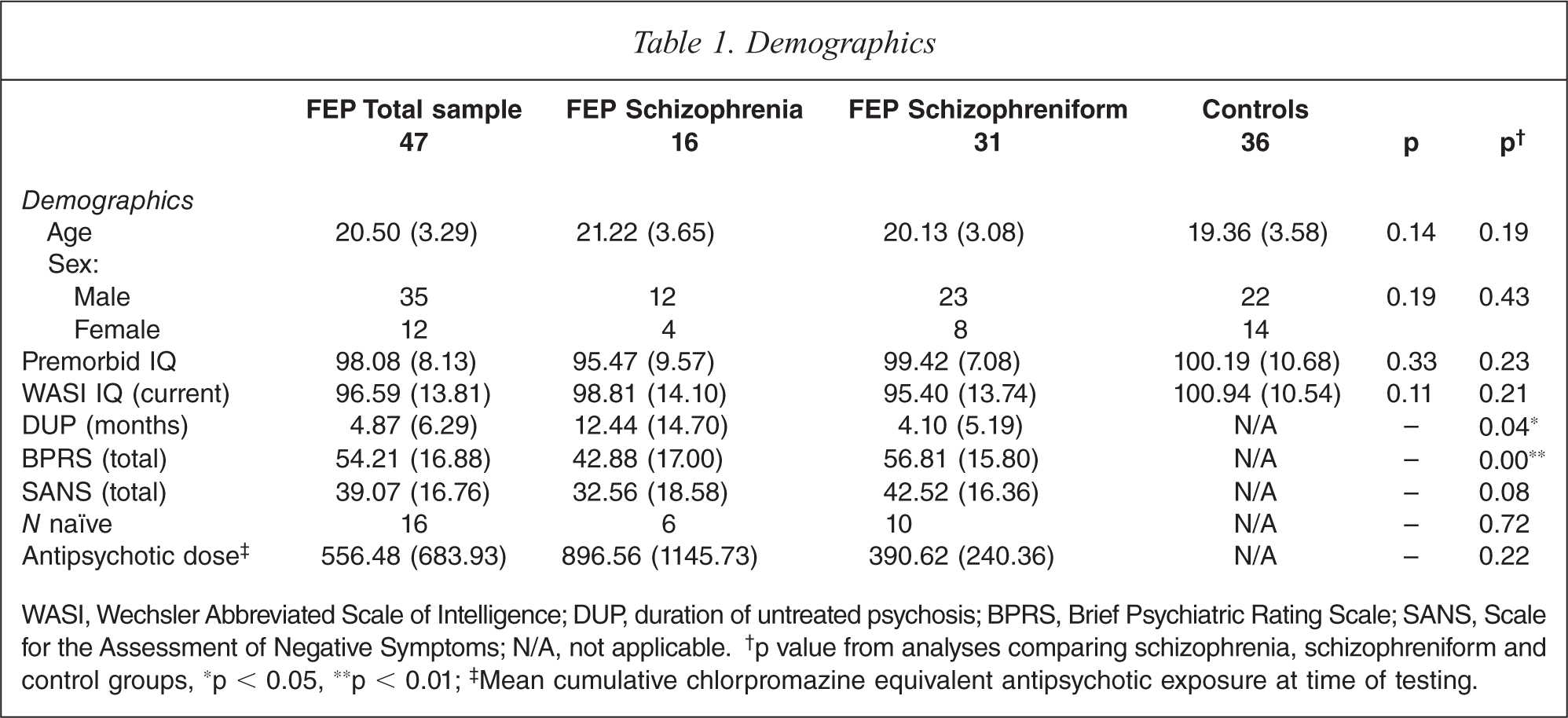
WASI, Wechsler Abbreviated Scale of Intelligence; DUP, duration of untreated psychosis; BPRS, Brief Psychiatric Rating Scale; SANS, Scale for the Assessment of Negative Symptoms; N/A, not applicable.† p value from analyses comparing schizophrenia, schizophreniform and control groups, ∗p < 0.05, ∗∗p < 0.01;‡ Mean cumulative chlorpromazine equivalent antipsychotic exposure at time of testing.
Thirty-six healthy volunteers were selected from a larger sample recruited from similar socio-demographic areas as the patients, and were matched as closely as possible for age and sex. Exclusion criteria for all subjects were: a history of significant head injury, seizures, neurological disease, impaired thyroid function, steroid use or DSM-IV diagnosis of alcohol or substance dependence. Control participants with a personal or family history of psychiatric illness were excluded. This protocol was approved by the Royal Melbourne Hospital Mental Health Research Ethics Committee, and each participant provided written informed consent to take part.
Materials
The VPA learning task was a spoken version adapted from a previous study of temporal lobe epilepsy [26]. Each trial included eight word pairs: four using concrete words (i.e. with high imageability, e.g. beach-wheel) and four using abstract words (i.e. with low imageability, e.g. usual-exist). Imageability has been shown to be an effective variable in assessing verbal learning, where more durable memories are formed for highly imageable words in both healthy controls and neurological patients [26,33]. Concrete and abstract pairs were presented in different series of fixed random orders.
Psychopathology was assessed using the Brief Psychiatric Rating Scale (BPRS) [34] and Scale for the Assessment of Negative Symptoms (SANS) [35]. Premorbid IQ was estimated using the National Adult Reading Test (NART) [36] and baseline IQ was obtained with the four subtests from the Wechsler Abbreviated Scale of Intelligence (WASI) [37].
Procedure
The mode of presentation of the relational memory task was similar to that used in the VPA subtest of the Wechsler Memory Scale – third edition (WMS-III) [38]. Initially, practice examples were given until the participant understood the task. The task comprised three phases. During each trial of phase 1 (and 2), word pairs were read aloud by the experimenter at a rate of one pair every 3 s. After a 5-s break recall was cued by presentation of the initial word of each pair (e.g. ‘Do you remember the word that went with beach?’). If the correct response was not recalled within 5 s, the complete pairing was provided (e.g. ‘It was beach-wheel’). The cues were presented in a different order from the learning trials. In phase 2 (interference), two trials of presentation/cued recall were administered, where the first word of the phase 1 pairs (denoted AB pairs) was paired with a completely new second word (denoted AC pairs). In phase 3 (post-interference cued recall) the cue words were read aloud and the participant was asked to recall the original word that it was paired with in phase 1 (i.e. AB pairs). No feedback was given in phase 3.
Statistical analyses
Demographic data were analysed using χ2 statistics (sex, treatment naïve status). One-way analyses of variance (ANOVAs) with Tukey post-hoc comparisons were performed for the variables age and premorbid IQ, and (for patient groups only) independent samples t-tests were performed for duration of untreated psychosis (DUP), total BPRS, total SANS, and chlorpromazine equivalent antipsychotic dose.
Two sets of analyses were performed on the relational memory data, aimed at testing two experimental questions: (i) are FEP patients impaired on a relational memory task which is suggested to index MTL functionality, and (ii) is impaired performance specific to the schizophrenia subgroup who are in a later illness phase than schizophreniform patients? Therefore, Analysis 1 involved the between-subjects factor ‘group’ to define participants as either FEP or controls. Analysis 2 included the between-subjects factor group to define participants in terms of a diagnosis of schizophrenia (including schizoaffective disorder), schizophreniform psychosis, or the control group. All analyses consisted of mixed-design ANOVAs with Greenhouse-Geisser corrections: trial and imageability were within-subject factors. Follow-up analyses were performed for all significant main effects and interactions in which the factor group was involved.
Results
Demographics
There were no significant group differences for the variables age, sex, premorbid IQ and WASI IQ at the time of assessment (see Table 1 for demographic details). DUP (t(16.95) = 2.2, p < 0.05) was significantly longer for the schizophrenia group in comparison to the schizophreniform group, as expected. Total BPRS score (t(45) = 2.79, p < 0.01) was higher for the schizophreniform group when compared to the schizophrenia group.
Learning and memory (phase 1)
Analysis 1
A 4 (trial; 1–4) by 2 (imageability; concrete, abstract) by 2 (group; FEP, controls) mixed ANOVA showed a significant main effect for trial (F(2.6,210.16) = 202.77, p < 0.001), where participants performed better over consecutive trials. Additionally, a significant main effect was seen for imageability (F(1,81) = 148.11, p < 0.001), where, as expected, participants performed better with concrete items. There was no main effect or interactions for group (see Figure 1 and Table 2 for means and standard deviations of all relational memory parameters).
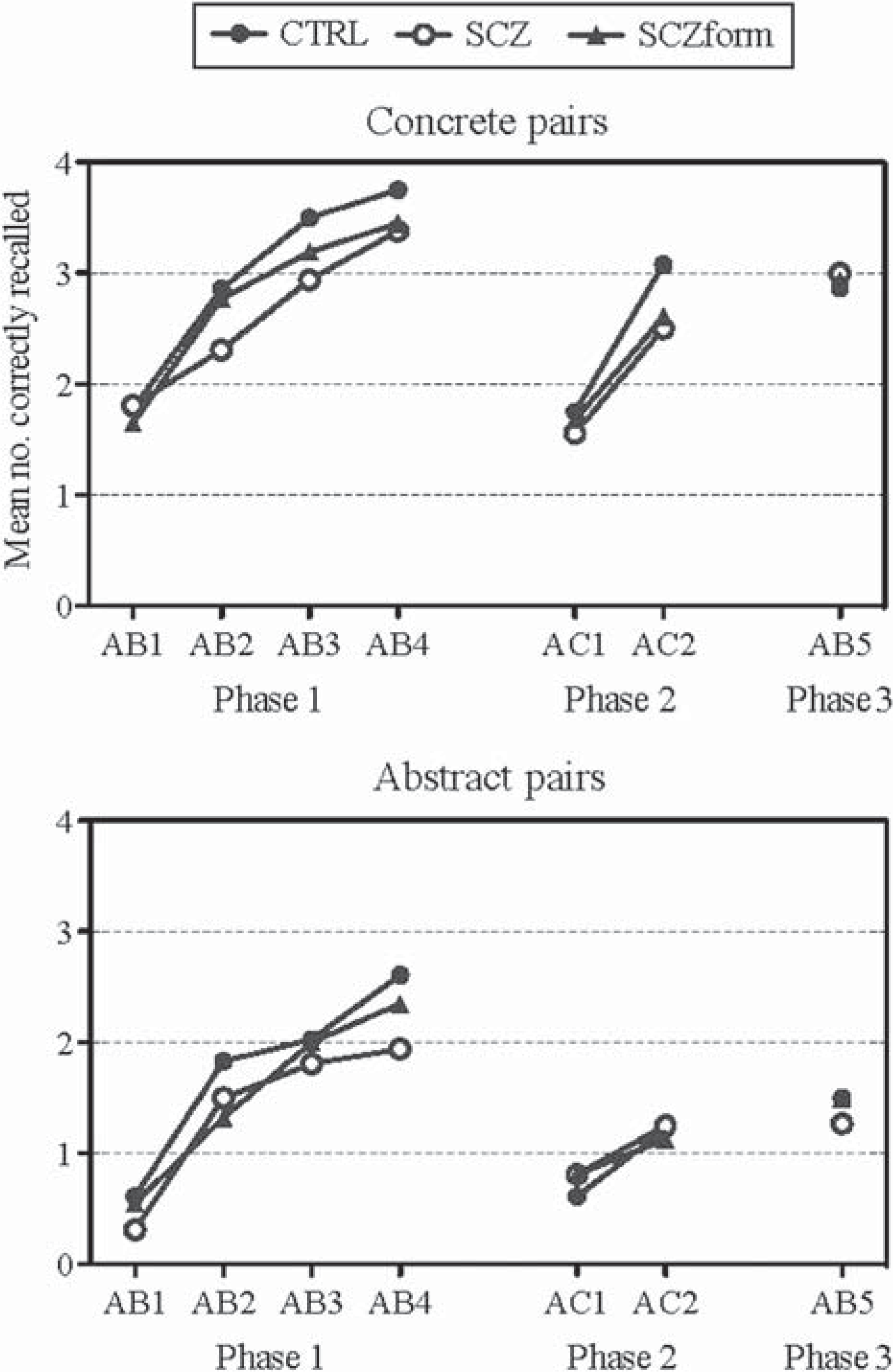
Learning curves (phase 1 and 2) and post-interference retention (phase 3) for concrete and abstract pairings of the relational memory task for the control (CTRL), schizophrenia (SCZ) and schizophreniform (SCZform) groups.
Means and standard deviations of relational memory performance measures
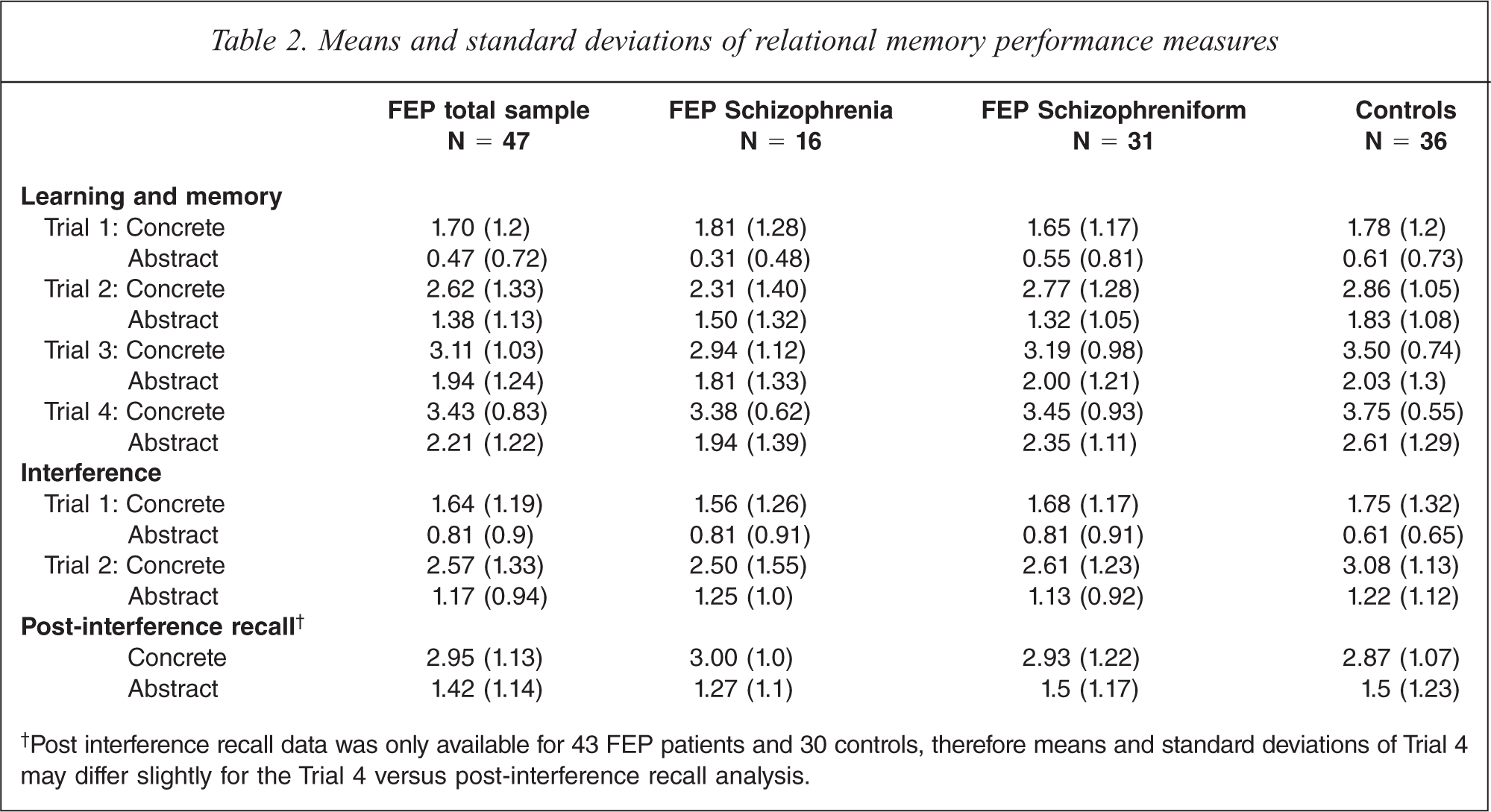
†Post interference recall data was only available for 43 FEP patients and 30 controls, therefore means and standard deviations of Trial 4 may differ slightly for the Trial 4 versus post-interference recall analysis.
Analysis 2
When we performed the same analysis using a 4 (trial; 1-4) by 2 (imageability; concrete, abstract) by 3 (group; schizophrenia, schizophreniform, controls) mixed ANOVA there were still no main effects or interactions for group.
Interference (phase 2)
Analysis 1
A 2 (phase; initial learning, interference) by 2 (trial; 1-2) by 2 (imageability; concrete, abstract) by 2 (group; FEP, controls) mixed ANOVA showed significant main effects for trial (F(1,81) = 200.62, p < 0.001) and imageability (F(1,81) = 201.28, p < 0.001), where both groups performed better on trial 2 compared to trial 1 and recalled more concrete than abstract words. There was a significant interaction between trial and group (F(1,81) = 4.64, p < 0.05), with controls showing slightly more improvement on trial 2 overall (see Figure 1).
Analysis 2
The 2 (phase; initial learning, interference) by 2 (trial; 1-2) by 2 (imageability; concrete, abstract) by 3 (group; schizophrenia, schizophreniform, controls) mixed ANOVA failed to find a significant main effect of group or group interactions.
Post-interference cued recall (phase 3)
Analysis 1
A 2 (imageability; concrete, abstract) by 2 (group; FEP, controls) mixed ANOVA showed a significant main effect of imageability (F(1,71) = 115.24, p < 0.001), where again concrete word pairs were recalled more frequently than abstract pairs. No effects of group were observed.
In order to assess the effect of interference on retention a 2 (trial; trial 4, post-interference cued recall) by 2 (imageability; concrete, abstract) by 2 (group; FEP, controls) mixed ANOVA was performed. There were no significant interactions found with the factor group.
Analysis 2
There were no significant group differences when the analyses were run using the two patient subgroups in comparison to controls.
Medication effects
Mixed ANOVAs were run for the patient group with treatment status (naïve, treated) as a between-subjects factor and relational memory parameters as independent variables. There were no significant effects of antipsychotic medication status on performance.
Discussion
First episode psychosis patients were generally unimpaired on the relational memory task. Although the FEP group showed less improvement on the second trial of learning, there was no pervasive impact of such a reduced benefit in terms of performance over the four initial learning trials, thus patients appear to have made up ground over repeated trials. In addition, relational memory performance was not dependent on phase of illness, that is, whether the individual had recently become psychotic (less than 6 months prior) or whether they had been ill for 6 months or longer.
The lack of a clear deficit in relational memory of our FEP sample, combined with research of chronic schizophrenia that consistently reports severe deficits, implies a progressive decline in verbal relational memory, and purportedly hippocampal functioning, over the course of the illness. This is contrary to a number of studies that found significant verbal episodic memory deficits in ARMS cohorts [5–7] and FEP patients [39–43] in comparison to controls. Similarly, our results contradict studies supporting a stable impairment across the lifespan of the disorder, where FEP patients were as impaired on verbal memory tasks as chronic schizophrenia patients [4, 44–46].
Variations in the types of verbal memory tasks employed and ensuing verbal memory processes assessed should be taken into consideration. Our previous research has found performance on the VPA subtest of the Wechsler Memory Scale – revised (WMS-R) to be similar between patients and controls, even when arbitrary and semantic pairs were analysed separately [44]. In contrast to this and our current findings, performance on a VPA task that assessed relational ‘recognition’ memory was found impaired in FEP patients [42,47]. Discrepancies are possibly explained by differences in task design, where the latter task may have been confounded by sustained attention deficits as it involved a recognition phase of 60–80 words, as opposed to eight used in the current study and the WMS-R. While the current study had 80% power to detect significant medium-sized differences between patients and controls (Cohen's d = 0.5), we had insufficient power to detect small effects that may exist. Albeit these effects would still be considerably smaller than that observed in chronic patients, which is consistent with the theory of progressive decline over the course of the illness [3]. It may also be that our patient group was too heterogeneous, as five patients in the schizophrenia group had schizoaffective disorder, and it is unknown as to whether the schizophreniform patients went on to develop a pure schizophrenia presentation or an affective psychosis. Despite this, one of the previous studies that did find relational recognition memory to be impaired also included schizoaffective, schizophreniform and bipolar disorder patients in their FEP sample [47], suggesting our findings are due to the specific nature of our relational memory task.
Other verbal memory measures that are not necessarily direct measures of relational memory have been more frequently used. Story recall (Logical Memory subtest; WMS-III or WMS-R) has repeatedly been found impaired in FEP and ARMS patients [4–7,43,44], although Addington and Addington [4] found FEP patients had improved on this subtest at 1-year follow up. Other measures that were previously found to be poorly performed include the RAVLT [40,41,43], California Verbal Learning Test (CVLT) [6,7,48,49], Hopkins Verbal Learning Test [39], Buschke Selective Reminding Task [50] and the Wide Range Assessment of Memory and Learning [51]. Worth noting is that a number of these studies analysed data using multivariate ANOVA statistics, thus obtaining a global indication of verbal learning and memory rather than a specific component. This implies broad verbal learning and memory deficits are present (implicating dysfunctional fronto-temporal networks), but that specific components of this cognitive domain may be less impaired in FEP, such as relational memory. This is supported by previous findings of intact visuospatial relational memory in a FEP sample while the chronic schizophrenia group displayed significant deficits, further implying progressive decline in relational memory sometime after illness onset [52].
Few studies have looked at verbal memory performance based on subdiagnoses in a FEP sample [43,48–51]. McClellan and colleagues [49] found subdiagnosis had no significant effect on performance of an extensive cognitive battery, which included the CVLT. However, no control group was used and their subgroups consisted only of schizophrenia, bipolar 1 and psychosis NOS, the latter of which was reportedly more representative of an ARMS group. Zabala and colleagues [48] also failed to find any difference in impairment in general cognition, nor on performance of the CVLT, between individuals with schizophrenia, bipolar disorder and other psychoses, but all were significantly impaired relative to controls. Fagerlund and colleagues [50] found similar results when comparing to a non-organic, non-affective psychosis subgroup. Conversely, other early psychosis researchers found verbal learning/memory deficits in particular were able to differentiate the schizophrenia patients from those with psychotic affective disorder [43] and those with psychosis NOS [51]. However, it can be argued that differentiation between these diagnoses and a diagnosis of schizophrenia are qualitative in nature, rather than related to phase or duration of illness, as in the current study. Given findings are mixed, and methodology varies greatly between studies, in terms of age range, verbal memory task used and division of subdiagnostic groups, further research is needed before definitive conclusions can be made.
Characterization of verbal memory performance in the FEP cohort is greatly needed to aid in the development of intervention strategies to optimize long-term social and occupational functioning. Recent research shows degree of cognitive impairment at the initial onset of FEP can be predictive of functional outcome at 1-year follow up [53]. In addition, verbal memory performance alone has been found to be more predictive of social functioning than severity of negative symptoms in a sample of UHR individuals [7]. This, along with evidence of potential improvement in verbal episodic memory after psychosis onset [4,54–56], suggests cognitive training may be beneficial to prevent decline and reverse impairment in certain cognitive domains at this stage of illness. To date, general cognitive remediation training, while proven to enhance performance on cognitive measures, has yielded mixed results in relation to their secondary impact on functional outcome. Nonetheless, several programmes such as cognitive adaptation training [57], the neuropsychological educational approach to remediation [58], cognitive enhancement therapy [59], and social cognition and interaction training [60], have been found to be beneficial for occupational and social functioning. More specific ‘auditory’ computerized training has also shown promising results, which significantly improved verbal memory in particular, with implications for neuroplasticity-based brain changes [61]. Future research should assess the impact of cognitive training on every-day living, and its utility in FEP cohorts.
In conclusion, our findings suggest that verbal relational memory deficits do not appear until some time after the first episode, implying preventative measures may be optimal at this point in the illness. However, it is common for free recall of verbal information to be significantly impaired even during the prodrome and in ARMS patients, suggesting dysexecutive retrieval deficits may be core targets for remediation during the at-risk/prodromal phase. Moreover, by enhancing our understanding of hippocampal functioning and brain changes related to memory impairment across the course of the illness, we will be better equipped to devise effective pharmacological treatment strategies for improving key cognitive processes in the future.
Footnotes
Acknowledgements
