Abstract
Keywords
Since the early twentieth century it has been noted that co-occurrence of obsessive–compulsive symptoms (OCS) and obsessive–compulsive disorder (OCD) is frequently seen in patients with schizophrenia and related disorders [1] Recent studies reveal high co-morbidity rates for OCS in schizophrenia patients varying from 7.8% to 46.6% [2,3,4]. Prevalence rates for OCD reported in schizophrenia populations vary from 7.8% to 26% [2,5–10]. Estimated prevalence figures for OCS in the general population are similar (21–24%), but clearly lower for OCD (2–3%) [11]. The wide range of reported co-occurrence rates of OCS in schizophrenia patients is probably due to differences in definitions of obsessive–compulsive features and sample characteristics (i.e. age of subjects, inpatient-outpatient, stage of illness). An important challenge facing investigators and clinicians is differentiating an obsession from a delusion. Obsessions in OCD have traditionally been distinguished from delusions in psychotic disorders based on the individual recognition of the compulsions or obsessions as ego-dystonic phenomena, implying the presence of insight. Earlier studies concluded that patients with schizophrenia can develop OCS as a sign of emerging reality testing of psychotic symptoms and stated that the presence of OCS could be an indicator of good prognosis [12,13]. However, subsequent studies reported poorer outcome and higher levels of positive and negative symptoms in schizophrenia patients with OCS [3,4,14].
Recently, several hypotheses concerning the association of OCS in schizophrenia have been proposed. First, OCS in schizophrenia might be considered as a subtype of schizophrenia, with early onset obsessive–compulsive symptoms representing first manifestations of the psychiatric disorder [3]. This subtype can be seen as a separate category of schizophrenia, by some authors referred to as schizo-obsessive disorder [15,16]. Second, the occurrence of OCS in schizophrenia patients may be associated with the treatment with second generation antipsychotic medications [17]. And finally both disorders could be considered as two separate disorders with a high co-morbidity and both with an onset in adolescence [3,14].
OCS can either begin prior, during or after the first episode of psychosis (FEP). If OCS typically occurs prior to FEP, it would be likely that OCS together with schizophrenia can be seen as a schizophrenia-subtype with OCS as early symptoms of the psychotic disorder. Since the mean age at onset of OCS in patients with first-episode schizophrenia reported by Poyurovsky et al. [6] is 16.6 years (SD = 8.7) and the mean age at onset of psychosis is 23.4 (SD = 5.9) years, this would also support the hypothesis that onset of OCS occurs prior to the onset of psychotic symptoms. Another possible supportive argument for the hypothesis that OCS is a subtype of schizophrenia, is the high incidence of OCS in patients at ultra high risk (UHR) of developing psychosis [18], suggesting that OCS could be a part of the prodromal phase of schizophrenia.
However, if OCS occurs mostly after the first psychotic episode, the hypothesis that OCS in schizophrenia might be associated with antipsychotic medication is more likely. Supporting this argument, Byerly et al. [19] report in their study that OCS predominantly concurs with, or develops after the onset of the psychotic disorder in patients with schizophrenia or a schizoaffective disorder.
To our knowledge there has only been one earlier study that specifically addresses the time of onset of FEP in relation to the emergence of OCS [19]. Byerly and colleagues included only patients that had OCS at the time of assessment, their study population had a relatively high age (mean age 47.3 years) and subsequently a long delay between mean onset of schizophrenia (22.3 years) and assessment. More recently Devulapalli and colleagues performed a meta-analysis on studies concerning the temporal sequence of clinical manifestations in schizophrenia with co-morbid OCD and reported that the onset of OCD precedes schizophrenia; however, this finding was at a trend level (p = 0.066) [20]. To the best of our knowledge no earlier publication has assessed onset of OCS and/or OCD in a FEP population.
In the present study, we sought to assess the prevalence of OCS and OCD among all referrals with a FEP or with a potential UHR to develop psychosis to a specialized early psychosis department. When OCS or OCD was present, we determined the time of onset of OCS related to the time of onset of FEP: prior, during or after first-episode psychosis. Referrals to our specialized early psychosis department were not exclusively from Amsterdam, and demographic characteristics might differ between referrals from Amsterdam and from outside this catchment area. The Amsterdam population is known for its multiethnic background. Because of possible differences we will assess, gender, ethnicity and referral status between subjects with OCS and subjects without OCS.
With a consecutively diagnosed young study-sample with UHR subjects and subjects with recent onset of FEP and by using a strict definition of OCS (excluding repetitive psychotic experiences), our study design is appropriate to assess prevalence and disentangle the association between psychosis and OCS onset.
Materials and methods
Participants
This study was conducted in a specialized early psychosis department for treatment and research of subjects with FEP and subjects who are at ultra high risk of developing psychosis, located in the Academic Medical Centre Amsterdam, the Netherlands. The study was prospective and included all patients who were consecutively referred to the department or to one of our outreaching early intervention for psychosis teams in Amsterdam from 1 July 2006 until 1 July 2008. Patients from Amsterdam represented a treated incidence sample of FEP patients, while patients who were referred to our department came also from regions surrounding Amsterdam. Separate analyses for these two groups of patients will be presented in the Results section.
Patients gave their consent that anonymized clinical data could be used for the purpose of study assessment and were given the choice to ‘opt out,’ in which case their data were not used.
Instruments and procedure
For all patients who entered the study we collected demographic and psychiatric variables (age, gender, ethnicity and urbanicity for determining living in Amsterdam or outside of Amsterdam, diagnosis). During the diagnostic procedure, patients were interviewed by a psychiatrist or a psychologist with extensive clinical experience. In this approximately 2-h face-to-face interview, subjects were asked about their premorbid history of complaints, psychotic symptoms, OCS and general characteristics. Diagnosis of psychotic disorders was established using the Comprehensive Assessment of Symptoms and History (CASH) schedule. All residents and researchers were trained in performing the CASH interview and supervised by senior psychiatrists of our department. All cases were discussed in a weekly meeting with three senior psychiatrists and all residents and researchers involved in our diagnostic facility. Difficulties in assessing symptom status or diagnostic criteria were discussed and resolved by reaching agreement between all professionals involved. In 19 cases there was insufficient information to reach consent on symptom or diagnostic status. In these cases an extra diagnostic interview was held, sometimes including extra neuropsychological testing. In referrals to the diagnostic facility of our clinic with no evident psychosis, the Structured Interview for Prodromal Syndromes (SIPS) [21] was administered. This semi-structured interview was used to determine the presence, severity and type of UHR symptoms. The Scale of Prodromal Symptoms (SOPS), the rating scale of the SIPS, has four SIPS subscales that include five positive symptom items, six negative symptom items, four disorganization symptoms items and four general symptom items. All symptoms were rated on a 7-point rating scale rating from 0 (never, absent) to 6 (severe/extreme and psychotic for the positive items). The diagnosis of a UHR status is based on the score on the positive symptoms items. Scores in the 3 to 5 range were considered to be indicative of the UHR phase. Each SIPS interviewer received a two-day training workshop by T.J. Miller, one of the SIPS authors, including a reliability check after approximately six months. The pair-wise inter-rater reliability concordance of the SIPS was 77% and deemed acceptable by the training team. Patients were also classified as being UHR when they had a genetic risk and reduced functioning or brief limited intermittent psychotic symptoms [22]. Simultaneously, in a separate interview the parents or guardians were asked about the premorbid emergence and presence of symptoms and signs of the patient. We used information of both interviews to assess the presence of OCS in schizophrenia, schizophrenia-related disorders and UHR patients.
We used the strict definition of OCS according to the Structured Clinical Interview for DSM-III-R, patient version, as persistent, repetitive, intrusive, and distressful thoughts, obsessions unrelated to the patient's delusions, or repetitive goal-directed rituals, compulsions, clinically distinguishable from schizophrenic mannerisms or posturing [23,24]. An example of OCS unrelated to psychotic symptoms: fears of contamination associated with repetitive hand washing or compulsive behaviours such as counting, organizing or checking rituals in a patient with acoustic hallucinations with unrelated content. In contrast; an example of obsessive–compulsive-like symptoms related to psychotic symptoms: compulsive checking behaviours associated with paranoid fears, such as a need to compulsively check the house for camera or recording devices [18]. Consequently, patients whose obsessional thoughts or compulsions are related exclusively to psychotic content of thoughts are not included in the OCS or OCD group.
We screened all patients for the presence of OCS and if present, we determined the time of onset of OCS. If only a year was known, we noted the first of July of that year as a date and when there was a month and year known, we noted the 15th of that month and year. Additionally, for the patients with OCS who met the DSM-IV criteria for schizophrenia, schizophreniform or schizoaffective disorder we determined the age of onset of OCS and assessed whether onset of OCS was prior, during or after the onset of FEP.
After the diagnostic procedure the presence of OCS was assessed independently by two of the authors (B.S., K.L.), taking longitudinal, clinical, and heteroanamnestic information into account. They agreed upon the presence of OCS in (95%) of the cases. In case of uncertainty (5%), the final decision on presence of OCS/OCD was reached in a discussion with a senior psychiatrist (L.H.). The 5% of cases in which the presence of OCS/OCD was uncertain mainly comprised cases were there was some doubt about the relation with psychotic content. We used a minimal uncertainty model, so if there was the slightest doubt it was discussed with the senior psychiatrist. We only assessed OCD when DSM-IV criteria were fulfilled.
To determine ethnicity we used the classification used by the Dutch Central Bureau of Statistics [25]. If a patient or one of his or her parents was born in another country, that patient was assigned to the ethnic group that fitted that foreign country. If the parents were born in different countries, the country of birth of the mother was used.
Statistical analysis
All statistical analyses were performed using SPSS 16.02 software. By using chi-square tests and logistic regression we compared different variables (gender, ethnicity, living in Amsterdam or not) in patients with OCS to patients without OCS.
Results
Three hundred and sixty-five consecutively referred patients were included in this study. No patients were rejected and none decided that their anonymized clinical data could not be used for study purposes. Of these 365 included patients, 297 (81.3%) were male and the mean age at diagnostic assessment was 21.7 years, (SD 4.5); 197 (54%) patients lived in the catchment area Amsterdam, 168 (46%) were referred from other parts of the Netherlands. Regarding ethnic origin, 194 (53.2%) patients were of Dutch origin and 171 (46.8%) had another background, e.g. Surinamese (13.1%), Moroccan (9.9%) or Turkish (3.3%). Of 21 (5.8%) patients we were not able to identify the ethnic background because there were not enough reliable data available to determine the ethnicity. Of the cases included, 154 met DSM-IV criteria for schizophrenia (120 paranoid type, 21 undifferentiated, 12 disorganized and 1 catatonic), while 20 met DSM-IV criteria for schizoaffective disorder and 20 met DSM-IV criteria for schizophreniform disorder; 76 subjects were diagnosed with other DSM-IV psychotic disorders (five substance-induced psychotic disorder, one psychotic disorder due to general medical condition, four mood disorder with psychotic features, eight brief psychotic disorder, 58 psychotic disorder not otherwise specified); 29 patients met UHR criteria. The remaining 66 participants were referred to our clinic but did not have a psychotic disorder nor met UHR criteria.
OCS was assessed in 42 of the 365 subjects (11.5%); 18 patients within the group of schizophrenia and related disorders had OCS (9.3%), 11 patients in the ‘other psychotic disorders’ group, 14.5%), six in the UHR group (20.7%) and seen within the group of patients assessed at our clinic but who did not have a psychotic disorder, nor met UHR criteria (10.6%). Three patients with schizophrenia, schizophreniform or schizoaffective disorder also met the DSM-IV criteria for OCD (3/194, 1.5%). One patient was diagnosed with OCD in the group ‘other psychotic disorders’ (1/76, 1.3%) and also one patient with OCD was found in the UHR group (1/29, 3.4%). Three patients without a psychotic disorder, nor fulfilling UHR criteria had OCD (3/66, 4,5%) (see Table 1).
OCS and OCD within four diagnostic categories
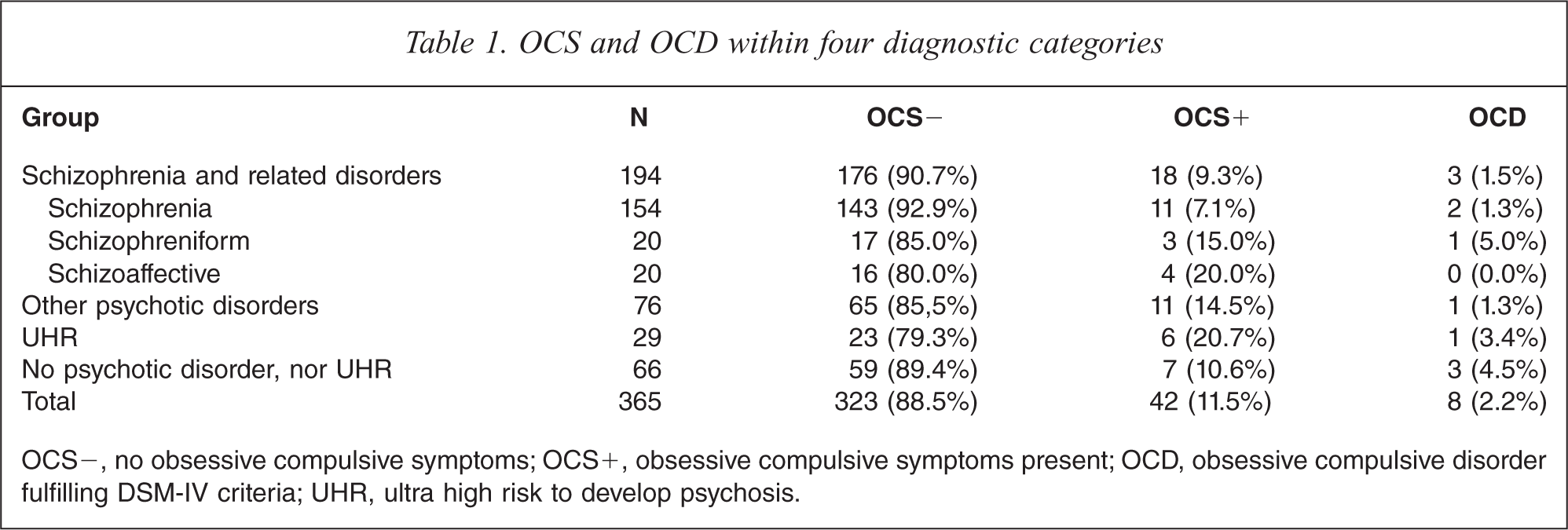
OCS−, no obsessive compulsive symptoms; OCS+, obsessive compulsive symptoms present; OCD, obsessive compulsive disorder fulfilling DSM-IV criteria; UHR, ultra high risk to develop psychosis.
For further analyses we focused on the group with schizophrenia and related disorders and the UHR group. We compared the patients with OCS to the patients without OCS.
Characteristics of patients with schizophrenia or a related disorder and OCS versus non-OCS
Of the 18 patients with schizophrenia or a related disorder with OCS, 13 (72.2%) were male, 13 (72.2%) had a Dutch ethnicity and 12 (66.7%) did not live in Amsterdam. The mean age at diagnostic assessment was 21.9 years (SD = 3.4). The mean age at onset of FEP was 19.9 years (SD = 3.7). The mean age at onset of OCS was 18.5 years (SD = 5.6). Of the 176 patients with schizophrenia or a related disorder without OCS, 148 (84.1%) were male, 75 (42.6%) were Dutch and 63 (35.8%) did not live in Amsterdam. The mean age at diagnostic assessment was 22.3 years (SD = 4.1). By using chi-square tests, we found that having a Dutch ethnicity or living outside of Amsterdam was related to prevalence of OCS. However, when we fitted these two variables in a logistic regression model, only living outside of Amsterdam seemed to predict OCS (odds ratio 3.6, CI 1.2–10.0, p-value 0.01), whereas adding ethnicity did not improve the model (see Table 2).
Demographics and characteristics of patients with schizophrenia or a related disorder with OCS versus non-OCS
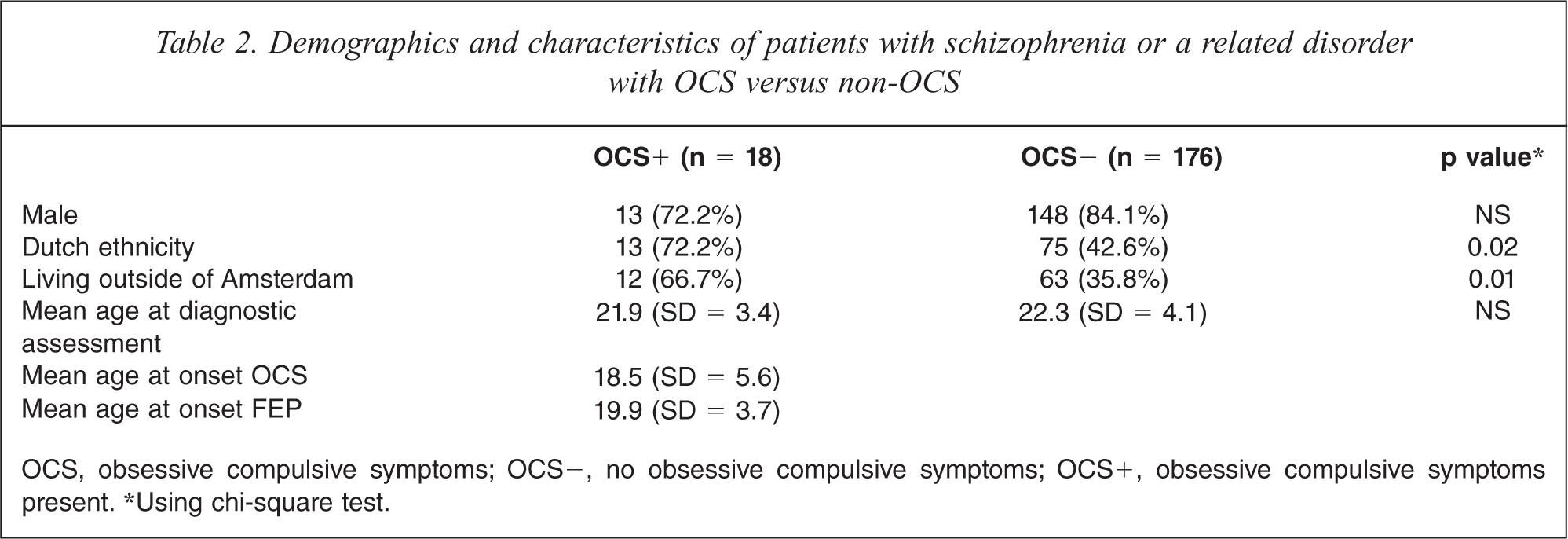
OCS, obsessive compulsive symptoms; OCS−, no obsessive compulsive symptoms; OCS+, obsessive compulsive symptoms present. ∗Using chi-square test.
The onset of OCS occurred before, concurrent with, and after the onset of first-episode psychosis in the following proportion of patients: 38.9% (7/18), 16.7% (3/18) 44.4% (8/18) (see Table 3). OCS onset would therefore be as likely to occur before as after the onset of schizophrenia or related disorders.
Time of onset of OCS prior, during or after time of onset of FEP

OCS, obsessive compulsive symptoms; FEP, first episode psychosis.
Characteristics of UHR patients
Of the 29 patients fulfilling UHR criteria, 26 (89.7%) were male. Their mean age at diagnostic assessment was 19.1 years (SD = 3.4). Eleven (37.9%) patients lived in Amsterdam and 18 (62.1%) were referred from other parts of the Netherlands 20 (69%) had a Dutch background; the remaining nine (31%) had an ethnic background other than Dutch.
Characteristics of UHR patients with OCS versus non-OCS
Of the six patients who fulfilled UHR criteria, with OCS, three (50%) were male, four (66.7%) had a Dutch ethnicity and three (50%) lived in Amsterdam. The mean age at intake was 18.3 years (SD = 2.5). The mean age at onset of OCS was 13.9 years (SD = 3.7). Of the 23 patients who met UHR criteria without OCS, 23 (100%) were male, 16 (69.6%) were Dutch and eight (34.8%) lived in Amsterdam. The mean age at intake was 19.3 years (SD = 3.6).
Discussion
In this study 9.3% of the patients with recent onset schizophrenia, schizophreniform or schizoaffective disorder had OCS, when a strict definition of OCS used. 1.5% also met the criteria for OCD. These prevalence figures for OCS and OCD are not as high as reported prevalence rates in other recent studies that examined OCS and OCD in schizophrenia and related disorders [2–10]. In contrast with the relatively low prevalence rates for OCS and OCD in schizophrenia and related disorders in our sample, we did find relatively high rates of OCS (20.7%) and OCD (3.4%) in UHR patients, although this finding should be interpreted with caution, since the sample size of this UHR group was reasonably small (N = 29) and predominantly male.
The onset of OCS occurred before, concurrent with, and after the onset of first-episode psychosis. Unlike Byerly et al. [19], we found no significant difference between the time of onset of OCS prior to or after the time of onset of first-episode psychosis. Also we did not confirm the finding that OCD precedes the onset of schizophrenia in co-morbid patients as found at a trend level in the meta-analysis by Devulapalli and colleagues [20]. An explanation of these differences might be the heterogeneity and possible selection bias (i.e. chronically ill and older age at assessment) in the study samples included in this meta-analysis. We specifically studied a young, recent-onset population.
The equal distribution of the time of onset of OCS related to the time of onset of first-episode psychosis suggests that OCS and schizophrenia are two separate disorders which have their onset in adolescence. However, it could still be that both hypotheses, OCS as a schizophrenia-subtype and OCS induced by antipsychotic medication, are true. It is not ruled out that maybe a small number of the patients with OCS occurrence prior to FEP suffer from a ‘schizo-obsessive’ disorder, and possibly another subgroup of patients with OCS occurrence after FEP, developed OCS associated with the effects of antipsychotic medication.
We found prevalence rates of OCS and OCD in patients with schizophrenia that are almost the same, or even lower, as the rates of OCS and OCD found in the general population. More importantly, these findings show lower prevalence rates of OCS and OCD in schizophrenia than many other studies. This difference might be caused by several reasons:
Our criteria for defining OCS were strict: obsessive–compulsive symptoms needed to be unrelated to the psychotic content. By narrowing our definition of OCS, we aimed at reducing the risk of over-reporting OCS.
The symptoms of schizophrenia or related disorders in our patients were probably not as severe as chronic schizophrenia patients as described in other studies.
Most patients were not using antipsychotic medication yet or had only recently started antipsychotic medication. Therefore, the suggested inducing effect of antipsychotic medication could have not yet been in force. There are indications that OCS might develop during long-term use of antipsychotic medication [26,27]. The patients in our sample generally had a short duration of treatment with antipsychotic medication; this may have reduced the occurrence of OCS and OCD in our sample.
It has been suggested by other authors that the use of low dose of antipsychotic medication may reduce OCS in schizophrenia and schizophrenia related disorders [26]. Because giving low doses of antipsychotic medication is common in recent-onset psychotic disorders this may also have been a factor related to the relatively low prevalence of OCS in our sample.
Some limitations to the present study need to be acknowledged. First, although we assessed the largest group to date of recent-onset schizophrenia and related disorder patients in respect to OCS occurrence, the sample size might be still insufficient to comprehensively evaluate the prevalence of OCS and OCD and the association of occurring OCS with the onset of first-episode psychosis. Hence, replication with a larger group and a strict catchment area is required to substantiate our findings. By including all patients who were consecutively referred to the department and all patients who were referred to one of our outreaching early intervention for psychosis teams in Amsterdam, we do not think we have missed FEP patients who are willing to participate in a diagnostic interview or willing to receive treatment in Amsterdam during the study period. However, we miss information on FEP patients not in contact with mental health care professionals. Included subjects with an UHR were help seeking. Many subjects with UHR may not have come to our attention, because they were not help seeking or were not referred to our diagnostic facility. All referrals with UHR were given a second chance if they missed their first appointment. However, 26 subjects (from the 121 potential UHR subjects referred) did not present themselves at the second appointment and information on their clinical characteristics is missed. Second, there might be a reporting bias: although psychiatrists and psychologists involved with the diagnostic procedure were instructed to focus on obsessive–compulsive symptoms, our strict OCS definition may have induced underreporting of OCS. However, since patients were also asked to fill in questionnaires about OCS we suppose that OCS with a certain severity is detected in our study. Third, on the other hand, OCS might have been over reported due to the possible presence of referral bias: patients referred to our specialized clinic from outside Amsterdam might suffer from a more severe disorder, thus may have more symptoms, including obsessive–compulsive symptoms. This might have affected our findings in this group. Fourth, although we suppose that we were able to include almost all FEP in contact with mental health care in Amsterdam, we probably have missed several cases. However, a systematic bias in occurrence or severity of OCS, between patients included and those not, is not likely. Fifth, the relatively small sample of UHR subjects precludes more definitive conclusions to be drawn.
In conclusion, we found a relatively low prevalence of OCS and OCD in a large consecutively diagnosed cohort of patients with a first episode of schizophrenia or related disorder. OCS either developed prior, during, or after the onset of FEP, which may have implications for understanding the relationship of the two conditions and their association with each other. We found a relatively high incidence of OCS in subjects who met UHR criteria, possibly prodromal for FEP, suggesting that OCS might be a part of the prodromal phase of first-episode psychosis. The predictive validity of OCS for transition to psychosis in UHR subjects warrants further investigation. Prospective, long-term follow-up studies are needed to fully characterize the occurrence, development and impact of OCS in subjects at UHR for psychosis and patients with recent onset schizophrenia, schizophreniform or schizoaffective disorder.
Footnotes
Acknowledgements
