Abstract
Parenting is a complex phenomenon including caretaking and attachment behaviours, emotional interaction, beliefs and a socioculturally influenced parental role. The parenting relationship has long been recognized as influencing the infant's psychological and emotional development, future relationships, and ability to adapt and respond to life's stress. Parenting disturbances, including child abuse and neglect, are of major public health concern with increasing numbers of infants and children reported to child protection authorities as being at risk of harm. Maltreatment in infancy is associated with a range of negative developmental consequences and is a key risk factor for physical, mental and psychosocial impairment [1,2]. Significantly, child abuse has been established as a transgenerational phenomenon in both human and animal models, influenced by an epigenetic mode of transmission via early maternal care [3,4] as well as social learning. However, discussions surrounding appropriate responses to, and interventions for, high-risk families remain ongoing.
While there are several theoretical models of parental disturbance (biological, psychoanalytical, and socioenvironmental) the attachment relationship is emerging as a central point of reference across disciplines. Bowlby [5] regarded the attachment relationship as an essential biological system that influences motivational and emotional processes pertinent to survival. Concepts embedded in attachment theory suggest that parents who experience early abuse develop core difficulties in recognizing and responding to their child's needs and are in turn at risk of repeating disturbed parenting behaviours [6–8]. Research has turned to the investigation of the neurobiological underpinnings of normal parenting behaviours with an aim to better understand parent–infant interactions. However, the neurobiology of disturbed human parenting has as yet received little attention. This paper will present a review of current knowledge surrounding the underpinnings of normal and abnormal parental behaviours and propose a transgenerational regulatory model of parenting disturbance. Finally, a clinical model for examining the neurobiology of disturbed parenting and strategies for examining its transmission are suggested.
The importance of infancy
Infancy is associated with periods of rapid neurological growth and organization that are thought to establish the foundation of later neurological functioning and reactivity, particularly that of the hypothalamic-pituitary-adrenal (HPA) axis and limbic regions associated with the regulation of affect and stress [9]. The experience-dependent nature of early brain development [10–12] and the significance of early infant–carer interactions as the context of development [12–17] are increasingly acknowledged in the literature. Positive and responsive early care is seen as vital for optimal neurodevelopment.
The care giver is widely theorized to serve as an external regulator of infant's affective states [9,15,18,19] a process based on reading and responding to the infant's emotional signals. In this role, care givers attenuate potentially maladaptive emotional responses and shape the infant's own capacity to self-regulate through an intuitive and primarily subconscious process of detecting, internalizing, and modelling an approximation of infant's emotional states [15]. Characteristic human care giving behaviours, such as mutual gaze, facial mirroring, touch and “motherese” vocalizations are considered fundamental to this role. In line with this, maternal sensitivity [20–22] and capacity for reflective functioning [23] (understanding the infant in psychological terms) have been associated with, and potentially mediate genetic influences on, the child's attachment status [24]. Such evidence has led to proposals that maternal regulatory capacities may impact significantly on the ability to effectively parent, and are a mechanism for the transgenerational transmission of stress reactivity, attachment and trauma in humans [6,25].
Parenting behaviour and infant development
Infant neurodevelopment occurs in a context of attachment experiences and emotional interaction with the carer. Recent neuroimaging evidence provides some support for the role of characteristic parent–infant interactions on early emotional and neurological development [26–28]. Infant-directed vocalizations have been associated with increased frontal brain activity in neonates in regions associated with early socio-affective experiences when compared to adult-directed speech patterns [26]. More broadly, the level of sensitivity of maternal care giving behaviours has been associated with the level of activity in frontal regions related to social behaviour and stress reactivity in infants [28].
It has further been proposed that such early mother–infant interactions have a longitudinal influence on the infant's neurological activity. Early infancy is considered a critical period for the development of the limbic system and reactivity of the HPA axis [29]. These biological systems are crucial to the appraisal of, and psychobiological response to demanding stressors, and have been implicated in the interpretation of social cues and regulation of emotion. In line with this knowledge, sensitive and responsive care giving has been associated with the onset of a HPA axis hyporesponsivity phase in infants around three months of age, while exposure to non/low responsive care giving in infancy has been associated with a heightened HPA response to stress [9,18,30,31]. Prolonged exposure to stress-related hormones in infant development may have a significant damaging effect on neurodevelopment, inhibiting neurogenesis and myelination [9,32]. Findings from primate studies suggest that unregulated stress exposure may result in hippocampal cell death [32]. In humans, child neglect has been associated with reduced volume of the corpus callosum [33] as well as abnormal dopamine, vasopressin-oxytocin and HPA responses to psychosocial stress in conjunction with attenuation of left neocortex, hippocampus and amygdala development [34]. Associations between early life stress and HPA reactivity have also been demonstrated in retrospective studies involving human children [35,36] and postpartum women [37], suggesting that the regulatory capacities of the care giver may have a long-term impact on the reactivity of the infant stress response system.
The regulation of infant stress may also play a key role in the development of the neurochemically driven dopaminergic and oxytocinergic reward circuits critical to the human social experience. In a longitudinal cross-fostering study, Maestripieri et al. [38] found that rhesus monkeys that had been exposed to high levels of rejection during early infancy possessed significantly lower cerebrospinal fluid levels of serotonin and dopamine metabolites. Importantly, these blunted serotonin levels remained consistent in monkeys across the three-year study period. Further, those who had experienced trauma in infancy and became abusive mothers displayed lower cerebrospinal fluid levels of the serotonin metabolite 5-hydroxyindoleacetic acid, suggesting that these early effects on hormonal system may have long-term significance for interactional disturbance in some monkeys.
In the human literature, changes in the stress-related corticotropin releasing hormone (CRH) have been connected with the binding of oxytocin (OXT) and argine vasopressin (AVP), hormones associated with social interaction, stress and the capacity for care giving behaviours, as will be reviewed in the next section. Abnormal care giving experiences during infancy have been associated with lower basal levels of AVP and lack of OXT response following social contact in later childhood as compared to children with ‘normal’ (raised from birth in own home) experiences [39]. This attenuation of hormonal responses to positive social contact in the absence of attentive parental care during infancy suggests that early caretaking experiences have a long-lasting impact on neurotransmitter systems associated with social affiliation [39]. Taken together, these findings suggest that abusive and neglectful parenting practices may disrupt key neurological processes involved in mother–infant regulating interactions, promoting emotional dysregulation and predisposing the infant to later psychopathology [6,18,40,41]. Such dysregulation of the stress response systems in infancy may also provide insight into the long-term and transgenerational negative influence of poor parenting.
Neurobiology of normal parenting
The existence of a ‘social brain’, a specialized network of neurological systems and structures facilitating the processing of and response to socially relevant stimuli, has been widely theorized [42]. Within Bowlby's [5] framework of the attachment relationship, the neural substrate of normal parenting must involve reward and motivation, affiliation and emotional processing pathways. Observed links between abnormal parenting and poor infant socio-emotional outcomes have led to the increased consideration of the neurobiological substrates of parenting in both the human and animal literatures. By examining the neurobiological correlates of normal parenting responses, researchers hope to identify areas that could be targeted in the remediation of abnormal, and those at risk of abnormal, parenting.
An extensive series of neural connections and signals are associated with the perception of affective stimuli. Positron emission tomography (PET) studies suggest the amygdala, widely thought to play a pivotal role in the perception of facial affect, receives significant influence from the thalamus, hippocampus, and fusiform gyrus [43,44]. The fusiform gyrus itself, heavily implicated in the processing of complex visual stimuli such as facial affect, has further been hypothesized to play a key role in strengthening the perception of emotional stimuli [45]. Oxytocin is also thought to play a role in modulating facial affect perception in humans through its effects on amygdalic responses to emotional stimuli [46]. It follows that these networks may play a pivotal role in early parenting behaviour and the ability to respond appropriately to infant cues.
Animal research has further implicated a range of cortical structures and circuits in the expression of normal parenting behaviours including the medial preoptic area (MPOA) of the hypothalamus [47,48,49] and its interaction with mesolimbic dopamine activity [50], as well as the ventral part of the bed nucleus of the stria terminalis and lateral septum [51,52], areas associated with the release of oxytocin. Lesions to the MPOA have been shown to abolish many parenting behaviours [53–56] particularly those associated with habitual caretaking practices such as nursing, breastfeeding and nesting. Electrolytic lesions to the MPOA and basolateral amygdala in rats have been shown to disrupt maternal behaviour and impair pup-directed reward reinforcement pathways [57]. These studies support a neurological basis of parenting behaviour and confirm the importance of known social perception and reward networks in its expression.
The hormonal systems underpinning normal parenting responsivity to infant cues have also gained increasing attention. The neuropeptides OT and vasopressin, discussed previously in the context of infant development, have been extensively implicated in mammalian formation of the mother–infant bond, social memories, maternal behaviour, social approach behaviours as well as the mammalian stress response [58,59,60]. Further, experimental human research has found intranasal administration of OT to be associated with superior detection of subtle social cues as well as increased feelings of trust and generosity [61]. In line with such observations, OT has been observed to have a significant binding effect within limbic structures [62–64] and there is increasing evidence of an anxiolytic effect of OT on structures involved in stress responsivity and the assessment of socially relevant information in humans [65,66] such as the amygdala [62,67] and hypothalamic paraventricular nucleus [67] that may facilitate social approach.
Some animal models implicate OT in the onset, but not the maintenance, of maternal behaviours [68]. Both lesions to paraventricular nuclei and intracerebroventricular administration of an OT antagonist have been found to result in delayed onset of maternal behaviour [69,70] while intracerebroventricular administration of OT in nulliparous female rats has been shown to induce maternal behaviour [71]. These results suggest a role of OT in the salience and motivational value of infant stimuli to the mother.
In humans there is some preliminary evidence of an association of peripheral OT levels, both during pregnancy and in the early post partum, and expression of sensitive maternal responses to infants [72]. In one study intranasal OT administration was associated with an improved the ability to infer the mental states of others from subtle facial cues [73]. Further, functional magnetic resonance imaging (fMRI) research suggests OT attenuates the normal responses of right amygdala and several frontal, temporal and brain stem region when viewing emotional expressions [46]. These results suggest OT may facilitate deeper processing of complex social information by reducing arousal in general [46]. This influence of OT on stress and emotion processing may be modulated by individual's stress responsivity, with evidence that OT receptor expression in the rat amygdala may be modulated by dopamine [62]. On the whole, this research provides preliminary evidence that maternal sensitivity is related to OT system interactions with stress and emotional regulatory systems; however, the efficacy of this system may be modulated by individuals basal stress levels.
In line with such models, Slattery and Neumann [74] detail the occurrence of a HPA-hyporesponsivity period in both animal and human mothers that extends from mid-pregnancy into the postpartum period and is thought to be related to increased activation of OT and prolactin systems. These systems act to attenuate HPA activity as well as dampening opioid and noradrenergic systems which serve as excitatory inputs of the HPA axis. Researchers have proposed this attenuation of the stress response is a normal and adaptive stage of pregnancy that allows mothers to begin to respond to infants in a calm and sensitive manner, perhaps through the regulation of maternal arousal [74]. While such a period may be conducive to the formation of adaptive patterns of parent–infant interaction and a secure attachment relationship, the effects of stress and affiliation system dysregulation on the occurrence of this period of in humans are as yet unknown.
In the animal literature, dysregulation of the dopaminergic reward system has been identified as a risk factor for parenting disturbance in naturally neglectful MaD1 mice [75], an effect accompanied by altered neuronal functioning in key dopaminergic brain regions, including the ventral bed nucleus of the stria terminalis. Maternal care has been shown to be significantly disrupted by the injection of dopamine receptor antagonists in normal rats [76,77].
Recent research in the human literature has implicated genetic variants associated with dopamine system functioning in mediating parental sensitivity. Such studies have examined the possible mediating effects of genes associated with dopaminergic system functioning on parenting sensitivity when parents are exposed to environmental stressors [78]. Researchers have observed a strong association between daily stress levels and sensitive parenting in parents with a specific gene combination associated with less efficient dopaminergic system functioning. In these parents, high levels of daily stress were associated with less sensitivity and low levels of daily stress were associated with higher parental sensitivity. Upper levels of parental sensitivity observed for this group exceeded those observed in other groups, which displayed no association between daily hassles and parenting sensitivity. This suggests stress was associated with a decrease in parenting sensitivity depending on the genetic make-up of parents. In those who are susceptible, these patterns, perhaps represent a generally higher reactivity to environmental stimuli, may have either positive or negative outcomes. In a similar population, Bakermans-Kranenburg et al. [79] assessed the influence of oxytocin receptor and serotonergic transporter system genes on maternal responsivity, finding that potentially less efficient variants of these genes were associated with lower levels of sensitive maternal responsivity. These studies suggest an association between genes, stress and sensitive parenting and implicate the production of dopamine and oxytocin in differences in human parenting sensitivity.
Functional MRI studies have recently been used to further investigate the involvement of several inter-related mechanisms in maternal responses to infant cues: reward-motivation pathways [80,81], the processing of infant affective information [82,83], and the influence of a mother's own attachment system and related processing capacities [80,84]. In these studies, researchers have aimed to examine the systems underlying maternal responsivity to infant cues and whether differences in maternal responsivity are related to how mothers themselves were parented. Such research may provide evidence that on the basis of their own early parenting experiences, people with different attachment classifications have different neurobiological organization of relevant reward and affective related structures.
In an early fMRI study, Bartels and Zeki [85] proposed that a ‘push-pull’ mechanism facilitated the exhibition of maternal behaviour in humans, following observations of activation in reward-motivation pathways (rich in OT and vasopressin), and deactivation within areas linked to negative emotionality and social inhibition when processing attachment-related stimuli. Several reviews of functional neuroimaging research examining the neurological basis of human parent–infant interactions have since aimed to build upon this early model of parenting behaviour [81]. In their review of the literature at the time, Swain and colleagues [81], proposed that the processing of infant cues exists within a motivation and reward feedback system centred on the cingulate cortex, coupled with complex planning, memory processing (hippocampus, parahippocampus and amygdala) and contextual social response processing. Recently, Swain et al. [86] suggested that parental responses to infants involve an initial detection and organization of infant cues by the sensory cortices which then interact with subcortical memory and motivational structures for cue appraisal. When sufficient motivation is achieved, these structures are said to interact with a number of corticolimbic modules related to reflexive caring behaviours, emotion, alarm, preoccupation, and cognition and memory, in order to generate a coordinated response via activation of the motor cortex and hypothalamus.
Since these models were proposed, researchers have furthered their investigations into the nature of maternal affiliation and care by examining neurological regions associated with responses to mother's own infants and to infant cues of different valences. Strathearn, Li, Fonagy and Montague [82] observed that when mothers viewed their own compared to other infant expressions there was an increased activation of key dopamine-associated reward processing regions (i.e. midbrain, ventral tegmental area/substantia nigra region, striatum, prefontal cortex and primary motor area), and regions implicated in emotion processing (medial prefrontal, anterior cingulate and insula cortex) as well as the caudate, temporal gyri, thalamic nuclei, left lateral amygdala, and hypothalamus, suggesting that own infant cues were more rewarding than other infant cues and perhaps elicited deeper cue processing and empathy. Further, an association between infant affect and activation of nigrostriatal regions interconnected by dopaminergic neurons was observed most strongly for happy infant faces and diminished with decreasing infant expression positivity. These results suggest that infant faces may stimulate striatal dopamine release, an effect perhaps particularly true for positive infant expressions, which may in turn promote emotionally responsive maternal care.
These results have been replicated in a naturalistic paradigm by Noriuchi, Kikuchi and Senoo [83], wherein mothers viewed silent video clips of infant faces during mother–infant interactions (i.e. smiling together versus infant crying when separated). Researchers observed own infant viewing to preferentially activate pathways associated with arousal and reward. Further, mothers viewing own infant distress in this design preferentially activated areas associated with increased empathy and emotion, suggesting that negative emotionality may facilitate approach and care giving behaviours in anticipation of reward.
Lenzi et al. [80] recently attempted to explore not only the neurological correlates of the observation of infant cues but also the maternal act of mirroring infant facial expressions. This study provides indirect evidence for the existence of a mirror neuron system in humans, a system associated with understanding, empathy and imitation. Investigating activation in areas demonstrated to have ‘mirror-like’ properties (ventral premotor cortex, inferior frontal gyrus, and posterior parietal cortex), Lenzi assessed neuronal responses of 16 primiparious mothers when viewing and imitating their own and ‘other’ infant's facial expressions of joy, distress, ambiguity and neutrality. Areas postulated to be allied within this system were activated during both the observation and imitation of own and other infant faces, and activated to a greater extent in response to emotional, as opposed to neutral, infant expressions. Interestingly these researchers also observed a positive correlation between right anterior insula activation when viewing infant faces and maternal reflective functioning (as measured by the Adult Attachment Interview), suggesting an association between activation of components of the mirror neuron system (which have extensive connections to limbic structures) and maternal empathy. This research provides preliminary evidence that a mirror neuron system that interacts with the limbic system via the anterior insula may underlie face-to-face mother–infant regulatory interactions.
Finally, in a important initial step in the examination of the underpinnings of sub-optimal parenting, Strathearn et al. [84] compared the neurological and hormonal responses to infant cues in mothers with secure and insecure/dismissing patterns of attachment, as measured by the Adult Attachment Interview. In this study mothers with secure attachment patterns displayed higher peripheral oxytocin levels following mother–infant play interactions than mothers with insecure/dismissing attachment patterns. Further, when viewing infant facial cues while undergoing fMRI, securely attached mothers displayed greater activation in the oxytocin and dopamine related hypothalamus/pituitary regions than did insecure/dismissing mothers. These results suggest maternal attachment status, a classification related to mothers’ own early parenting experiences, may be related to differing neurobiological responses to infant cues. They further implicate oxytocin in mediating maternal attachment-related behaviours, perhaps heightening reward and motivation to engage in parent–child interactions. Reward processing regions were also activated when securely attached mothers viewed their own infants sad facial expressions; however, insecure/dismissing mothers showed increased anterior insula activation, a pattern previously associated with negative emotional states such as pain and disgust when viewing own infants sad expressions. This evidence may suggest that for securely attached mothers, infant cues, regardless of valence, serve to motivate responsive maternal care, but may lack this quality for mothers with insecure/dismissing patterns of attachment and may even lead to rejection and avoidance of negative infant cues [84].
The existing fMRI literature largely supports Swain et al.'s [81] model of an interaction between corticolimbic structures associated variously with affiliation and reward in the perception and assessment of infant stimuli. The role of neurohormones such as oxytocin, and dopamine in human maternal behaviour have only just begun to be explored; however, evidence suggests they are pivotal in the initiation and sensitivity of maternal behaviours and that their efficacy is mediated by genetic, existing stress and environmental factors. This evidence further provides us with a framework from which to explore the neurological and hormonal correlates of abnormal parenting behaviours.
Transmission of parenting disturbance
In humans as in animals [38,87–100], individuals exposed to early trauma are at an increased risk of parenting disturbances with their own children, which may involve direct repetition of patterns or types of abusive behaviours [3,101–104]. The transgenerational transmission of parenting disturbance appears to be complex, involving social learning (some individuals have poor or absent models of parenting), poor emotional processing abilities, as well as genetic and epigenetic effects. The transgenerational effect of disruptions of early caretaking experiences on infants have been known for some time. In a series of seminal social deprivation studies, Harlow et al. observed that female primates that had been isolated as infants displayed a high rate of abuse, neglect and infanticide as mothers [105,106]. Maestripieri and colleagues [38,98–100] built on this work to provide evidence for the perpetuation of infant abuse among first-degree offspring. In a longitudinal cross-fostering observational study, Maestripieri [99] found that over half (56%) of primates exposed to abusive parenting practices during infancy, displayed dysfunctional maternal behaviours in adulthood. In contrast, all those reared by non-abusive mothers displayed adequate or ‘good enough’ parenting styles. Similar dysfunctional maternal care patterns with respect to licking and grooming have been observed in rodent separation and cross-fostering studies [107–110]. Findings from non-human primate epidemiological studies suggest that aberrant parenting is a relatively stable characteristic, with similar inconsistent interactions repeated with subsequent infants [100,104,111]. Maestripieri et al. [38] further observed that the transgenerational effects of maternal abuse on stress regulation may be mediated by serotonergic mechanisms and that development of these systems may be negatively affected by exposure to neglectful parenting.
Clinically, mothers at risk of parenting disturbance in humans have reported feelings of anxiety, anger and estrangement with their infant from birth [6]. Additionally, these mothers have often been found to have difficulties in perceiving and attending to the emotional needs of their infant and as such tend to be inconsistent in their interactions, ranging from intensely intrusive to avoidant, thereby disrupting processes that promote attachment security [112–114]. Despite significant clinical and epidemiological support for a matrilineal transmission of abusive behaviours as well as poor neurodevelopmental outcomes for infants exposed to early deprivation of care, the majority of research focused on understanding the neurobiological underpinnings of abusive parenting has relied upon animal models for explanation [16,81].
Borderline personality disorder as a model of disturbed parenting
The study of parenting disturbance in individuals with borderline personality disorder (BPD) provides a potential model for understanding the impact of early maltreatment on human care-taking behaviours and their neurological substrates. Borderline personality disorder is a severe heterogeneous disorder that is primarily characterized by pervasive patterns of instability in interpersonal relationships, distorted self-representations, attachment disorganization [115,116], as well as deficits in emotional regulation and reflective capacity [117,118]. For mothers, a diagnosis of BPD is also associated with an increased incidence of disorganized attachment behaviours in Newman et al. [6,119], and ongoing interactional difficulties with their children [119]. However, there is a growing consensus suggesting that affect dysregulation, i.e. the inability to accurately perceive and process emotional cues, is a core feature of this disorder, and reflects developmental outcomes associated with trauma-related early interactional and attachment disturbances [120–124]. In particular, individuals with BPD often experience hyper-arousal to emotionally demanding internal and environmental stimuli, including high sensitivity, high intensity, and slow attenuation [45,124]. This inability to regulate affective responses may predispose individuals to various kinds of self-destructive and aggressive behaviour [125].
Neurobiology of BPD: evidence for frontolimic system disturbance
Several streams of research examine neurobiological deficits in BPD populations with documented early attachment-related trauma. While findings overall are difficult to interpret, due to both methodological implications and the heterogeneity of the disorder, the key phenomena that have been studied in these investigations include affect deregulation, impulsivity, and over-reactivity to emotional stimuli, particularly negatively valanced emotional stimuli. These areas have been identified as central to normal responses to infant stimuli. As discussed, BPD is a high risk parenting group, often with personal histories of childhood trauma. As such, parents with BPD may present a suitable group from which to examine the neurological correlates of early trauma that may be relevant to disturbed parenting behaviour.
To date, neurobiological research has implicated frontolimbic regions in BPD pathology. Studies examining neuro-structural disturbances in this population have identified decreases in structures involved in emotional regulation and evaluation of negative stimuli including smaller hippocampal and amygdala volumes in BPD-affected samples compared to controls (Table 1). Associations between these reductions and BPD symptomatology are in line with current understanding regarding the role and development of these key social brain structures. Reductions in amygdala volume have been associated with the expression of impulsivity, inappropriate anger, increased internalizing and externalizing behaviours, and depressive symptoms in BPD populations (Table 1). Similarly, reduced hippocampal volumes have been associated with duration and severity of experiences of childhood trauma, childhood neglect, current trauma-related symptomology, and lifetime histories of aggressive behaviours (Table 1). Such results provide some support for hippocampal damage and limbic dysregulation as a result of early trauma in a human model.
Studies examining structural disturbances of frontolimbic neurobiology in BPD
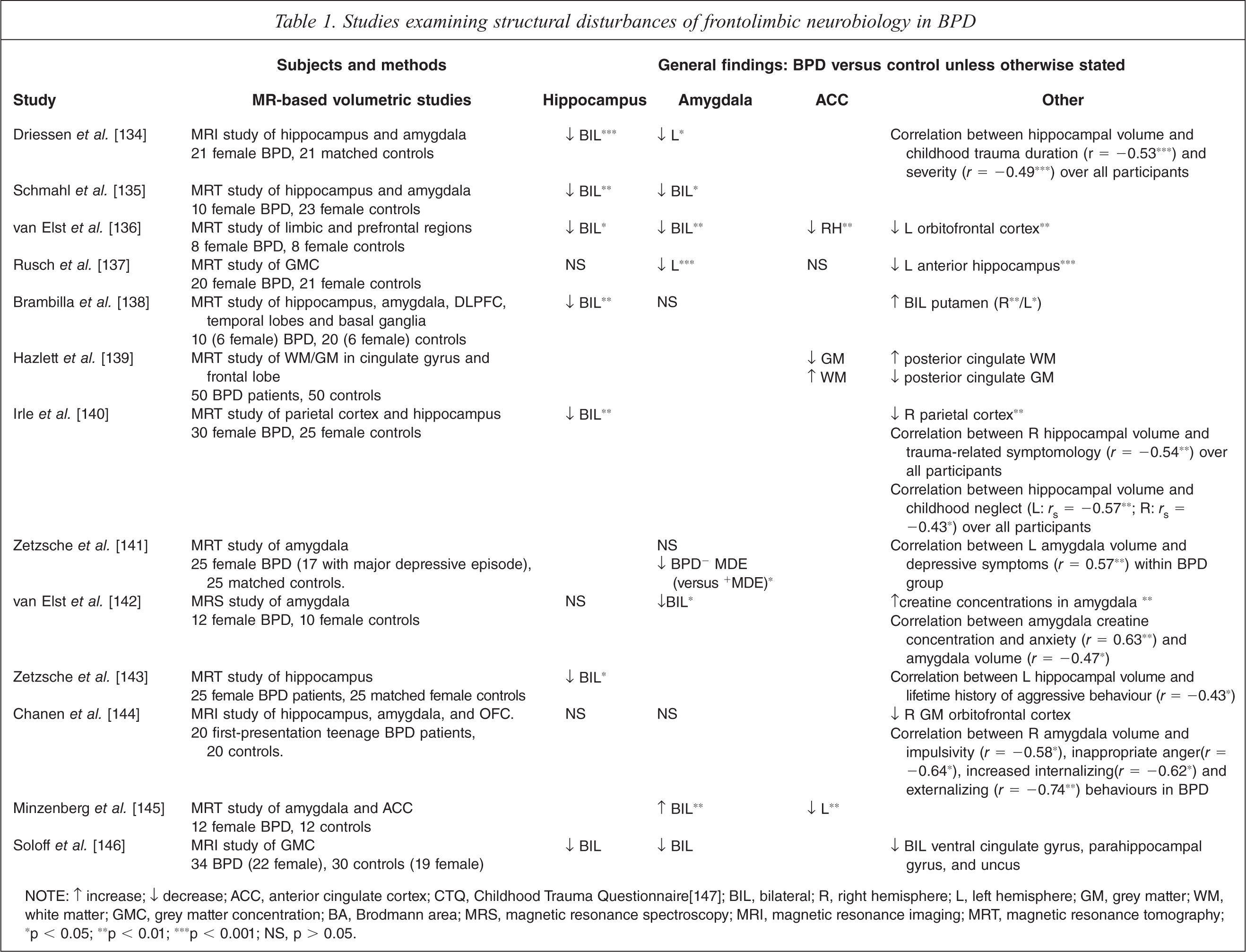
NOTE: ↑ increase; ↓ decrease; ACC, anterior cingulate cortex; CTQ, Childhood Trauma Questionnaire[147]; BIL, bilateral; R, right hemisphere; L, left hemisphere; GM, grey matter; WM, white matter; GMC, grey matter concentration; BA, Brodmann area; MRS, magnetic resonance spectroscopy; MRI, magnetic resonance imaging; MRT, magnetic resonance tomography; ∗p < 0.05; ∗∗p < 0.01; ∗∗∗p < 0.001; NS, p > 0.05.
Several functional studies of BPD neurobiology have observed neurological hyper- and hypo-reactivity in response to emotionally negative or aversive stimuli, and dysregulation of serotonergic systems, which may have significant implications for sensitive and responsive parenting. As summarized in Table 2, fMRI research has broadly identified anomalous anterior cingulate cortex (ACC), orbitofrontal cortex, and hyper-reactive amygdale activity in BPD, particularly in response to affective stimuli. As part of the prefrontal cortex, the ACC is responsible for providing inhibitory input to the amygdala regarding fear response during periods of stress [126]. It has also been found to be critical in decision-making processes, particularly those related to reward-motivation and has been found to play a key role in the processing of, and response to emotional stimuli (primarily empathy [127]). Lesions to the PFC have been shown to result in emotional and social dysfunction, with individuals unable to accurately interpret emotional signals [128]. These results have been supported in animal models with lesions to the ACC associated with diminished socially affiliative behaviour, and lesions to the orbitofrontal cortex (OFC) with emotional dysregulation and increased impulsivity [129].
Studies examining functional disturbances of frontolimbic neurobiology in BPD
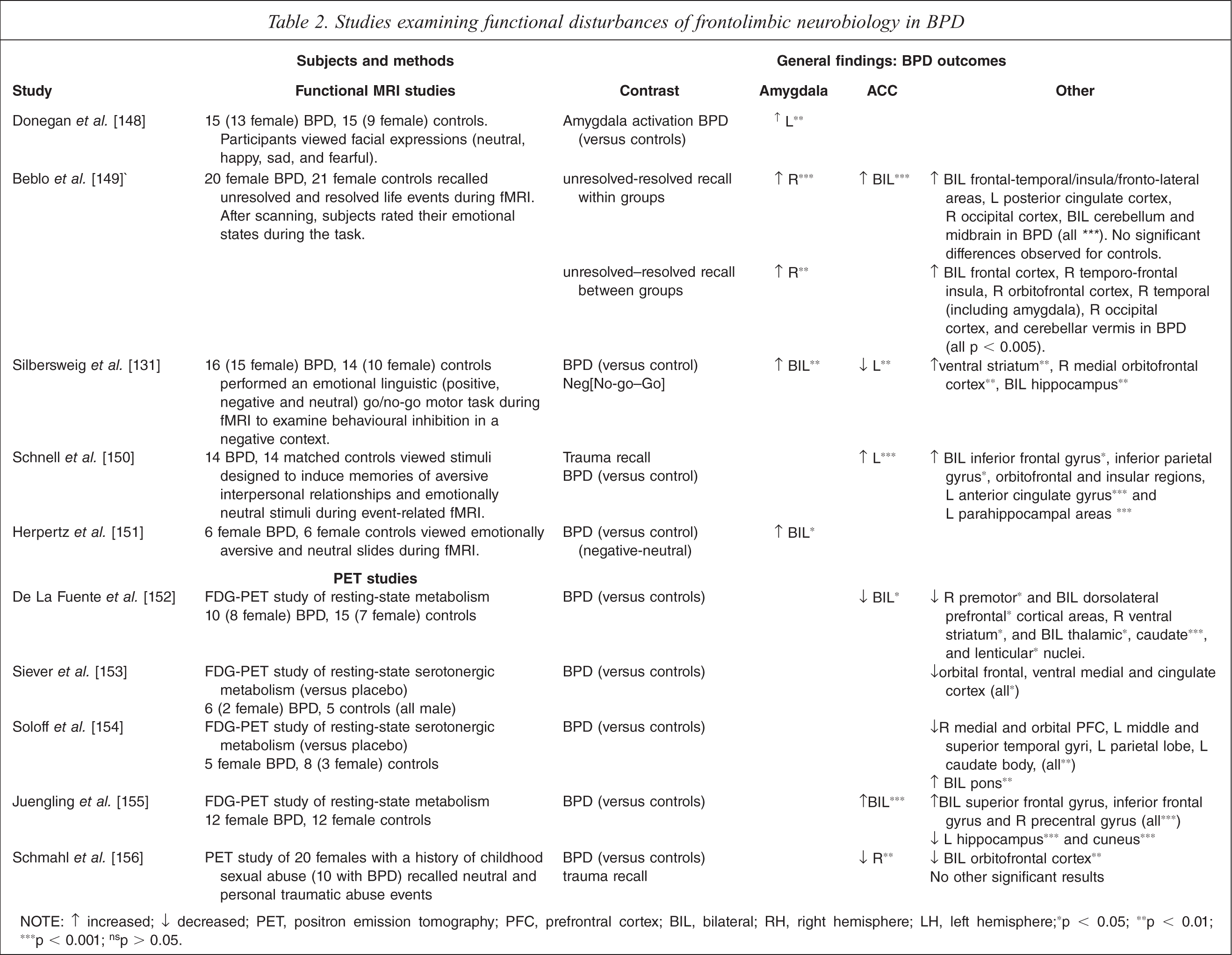
NOTE: ↑ increased; ↓ decreased; PET, positron emission tomography; PFC, prefrontral cortex; BIL, bilateral; RH, right hemisphere; LH, left hemisphere; ∗p < 0.05; ∗∗p < 0.01; ∗∗∗p < 0.001;ns p > 0.05.
Researchers using PET techniques have also observed hypo-stimulation of the frontolimbic system in women with BPD, both resting and when exposed to emotionally aversive stimuli (Table 2). Some researchers using these techniques have observed blunted serotonergic metabolism in the frontal and parietal lobe regions, though these findings are not consistent. An inability to regulate serotonin may predispose BPD patients to emotional disinhibition and impulsive aggression and contribute to parenting dysfunction within this population [130].
The inter-relationship between structural and functional impairment found in the highly interconnected orbitofrontal-limbic region may provide a crucial link in the breakdown of emotional processing. Silbersweig et al. [131] theorized that the increase in activation to aversive emotional stimuli indicate negative emotion may place a greater burden on decision-making processes, eliciting dysfunctional automatic responses. Behavioural consequences of limbic irritability may include the production of intense ‘flight or flight’ responses to emotionally challenging stimuli. As such, mothers with unresolved trauma histories may be at greater risk of repeating dysfunctional parent–infant interactional patterns, thus perpetuating the transgenerational transmission of trauma and abuse. Adequate or ‘good enough’ parenting relies upon the use of specific nurturing and care giving responses, particularly those related to affective attunement. The ability to empathize or infer and appropriately respond to the emotional experiences of others [132] may play a fundamental role in the development of secure attachment systems [133]. Impairment involving the PFC and ACC coupled with limbic irritability may be significant contributors to social and parenting dysfunction, and populations with BPD may provide the first steps towards a preliminary human model of abnormal parenting.
Conclusion
Evidence suggests that early parenting experiences may influence the development of skills necessary not only for normal infant development, but also the future capacity of that infant in their own parenting. The neurobiological development underpinning these abilities continues to be illuminated, with a notable overlap between those structures most affected by early parenting experiences and those utilized in normal parenting behaviours. However, disturbances in human parenting behaviour including neglect and abuse of children are poorly understood in terms of their neurobiological underpinnings. Figure 1 presents a summary model of the transmission of abnormal parenting behaviours through dysregulated emotional interactions which may affect both behavioural and neurological development, transgenerationally. Studies of BPD adults suggest a series of possible neurodevelopmental effects of early trauma with ongoing affective dysregulation and deficits in socioemotional processing as key features which may relate to difficulties in parenting. Early parenting in particular, involves processing of infant social and emotional communications as well as the need to regulate strong affect, and these capacities may be impaired in parents with BPD. Further, it may be hypothesized that parents who are experiencing parenting difficulties, particularly in maintaining a positive stance towards their child may be experiencing less reward in the interactions and have core issues in processing affective stimuli. Parents with BPD have fundamental difficulties in understanding and regulating their child's emotional and psychological experiences and distortions of early interactions may be related to high levels of stress and the effects of stress-related hormones on brain development as well as epigenetic effects on emerging brain functioning. This work suggests that it may be important to examine the specific patterns of neurobiological parenting difficulties in parents who have experienced significant early trauma as in BPD, and to examine neurodevelopmental sequelae in infants exposed to parenting disturbance.
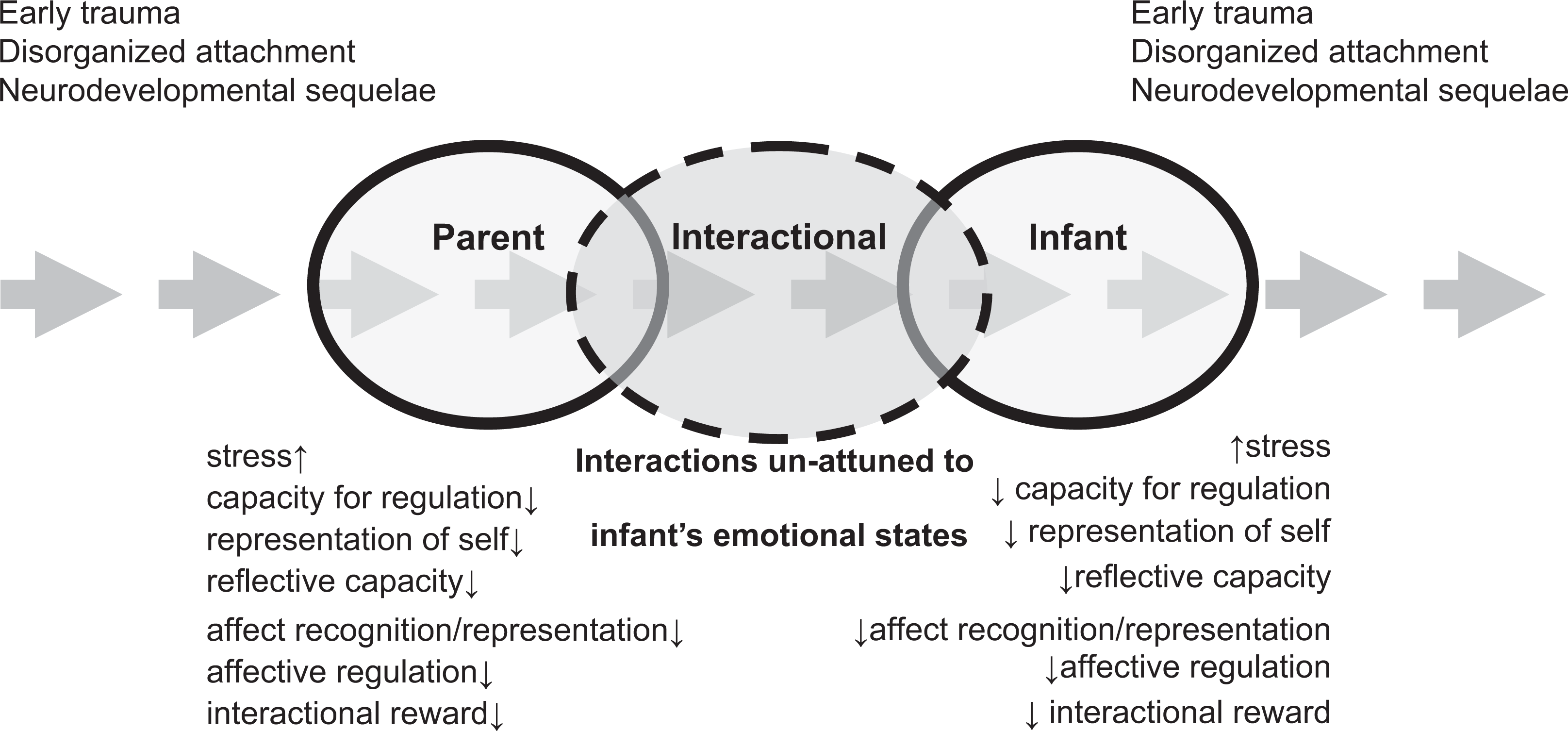
Transgenerational regulatory model of parent–infant interactional disturbance.
Footnotes
Acknowledgements
