Abstract
Since the introduction of endophenotype to neuropsychiatric disorders research by Gottesman and Shields in 1973, the study of endophenotypes has been expanded and incorporated into the strategic direction for schizophrenia research [1]. To be a qualified candidate for endophenotype status, the neurobiological marker should satisfy a set of stringent criteria, e.g. association with the illness, heritability, state independence, co-segregation with the illness, and quantification for reliable measurement [2].
Neurological soft signs also have features characteristic of useful endophenotypes [2,3] or intermediate phenotypes [4,5]. The term ‘soft signs’ refers to non-localizing neurological abnormalities that cannot be related to impairment of a specific brain region or are not believed to be part of a well-defined neurological syndrome [6,7]. It is opposite of the ‘hard signs’ that refer to specific impairment in identifiable brain regions or systems such as the extrapyramidal system [8]. However, the most recent imaging studies in healthy volunteers suggest that, although there is no specific region of the brain responsible for motor coordination signs such as fist-edge-palm action [9], there is a strong network brain activation between the right inferior and middle prefrontal regions [10]. A higher frequency of neurological soft signs has consistently been found in studies of individuals with schizophrenia [6,11].
One recent meta-analysis shows that fronto-temporal brain structural abnormalities are evident in non-psychotic individuals at high risk of developing schizophrenia including those people demonstrating schizotypal personality features [12]. In particular, substantial findings have also shown that while there are large effect sizes for prevalence between patients with schizophrenia and healthy controls [13], the non-psychotic first-degree relatives of the patients also demonstrated moderate to modest magnitudes of prevalence of soft signs compared to healthy controls [14]. This notion is most clearly understood within the neurodevelopmental framework [15]. Chen and Faraone [16] suggested further that genetic vulnerability to schizophrenia may manifest itself in schizophrenia-like personality disorders, including people with schizotypal personality features, in addition to full syndrome of schizophrenia. However, most of the studies that measured neurological soft signs in schizotypal personality disorders mainly came from non-psychotic probands who were associated with schizotypal traits (e.g. Gourion and colleagues [17–19]), very little is known about those people who also demonstrate schizotypal personality features but are not non-psychotic probands of the schizophrenia patients. These people are considered to be pseudo-schizotypals but share similar neurocognitive and behavioural difficulties in everyday life [20].
Chan and Chen [21], have recently demonstrated that high levels of neurological abnormalities, particularly neurological soft signs, characterize Chinese patients with schizophrenia. Based on these empirical findings of neurological soft signs in Chinese patients with schizophrenia, we would like to further explore the prevalence rate of neurological soft signs in people with schizotypal personality features. In particular, the purpose of this study was to focus on the prevalence of neurological soft signs and their relationship to schizotypal traits in individuals with psychometrically defined schizotypal features who did not have a known family history of schizophrenia.
Method
Participants
A total of 781 university undergraduates completed a full version of the Schizotypal Personality Questionnaire ([22]; Chinese version, [23]). SPQ was designed to measure schizotypal personality according to nine features of the DSM-III-R Schizotypal Personality Disorder (SPD). It is a 74-item questionnaire requiring simple ‘yes’ or ‘no’ ratings It captures specifically the nine traits of SPD, namely idea of reference, excessive social anxiety, odd beliefs or magical thinking, unusual perceptual experiences, odd or eccentric behaviour, no close friends, odd speech, constricted affect, and suspiciousness/paranoid ideation. Good psychometric properties of the original and Chinese versions have been described elsewhere (e.g. Raine et al. and Chen et al. [22–24]). Sixty-four participants (a total of 70 eligible students were approached, and 6 students refused to participate) with a raw score of the SPQ at the top tenth percentile of the current sample were considered to be psychometrically defined SPD-proneness cases and were recruited for participation [22].
Fifty-one participants (a total of 60 students were approached, 9 refused to participate) with a SPQ score below the cut-off were randomly selected as comparison controls. All the participants (both SPD-proneness and non-SPD-proneness cases) were screened by a trained research assistant to ascertain the absence of a history of psychiatric (other than presence of SPD in some cases) and neurological diseases. None had a history of psychosis in their first-degree relatives. IQ was estimated by short form of Chinese version of Wechsler Adult Intelligence Scale, including four subscales: information, arithmetic, similarity and digit span [25]. Handedness was assessed by the Annett Handedness Scale [26].
Measure: neurological soft signs
Neurological examination was performed by psychiatrists using the soft signs subscales of the Cambridge Neurological Inventory (CNI) [7]. In the original version of the CNI, three subscales addressed soft signs (categories of motor coordination, sensory integration, and disinhibition). The remaining four subscales elicit hard signs with specific localization of brain lesions (extrapyramidal signs, pyramidal signs, tardive dyskinesia, and catatonia) and were not included in the present study. The motor coordination subscale includes items such as finger thumb tapping, finger thumb opposition, diadochokinesia, fist-edge-palm, Oseretsky (score range 0–9); the sensory integration subscale includes extinction, finger agnosia, stereognosia, graphesthesia, left-right orientation (score range 0–8); the disinhibition scale includes saccade blink, saccade head, wink, and go no-go stimulus, and mirror movement of finger thumb opposition and diadochokinesia (score range 0–8). In the original scale, scoring was made according to standardized anchor points to indicate ‘normal’ response (scored as 0), ‘equivocal response’ (0.5), ‘abnormal’ response (1) or ‘grossly abnormal’ response (2). In the present study, item scores were dichotomized into either ‘absent’ (covering normal or equivocal) or ‘present’ (covering abnormal or grossly abnormal). Chan and Chen [21] have demonstrated that the CNI was sensitive to discriminate patients with schizophrenia from healthy controls in the context of Chinese setting, using the three subscales of neurological soft signs.
Procedures
This study received approval from the appropriate ethical committees as part of an extensive project examining the prevalence of schizotypy in a healthy population in China. Participants gave informed written consent and were assured of anonymity and confidentiality of the data to be collected. After identifying the SPD-proneness and non-SPD-proneness cases using the cut-off criteria, participants were approached and invited to take part in the second part of the study. Assessments were performed by two raters (R.C.K.C. and Y.W.) who were blind to the subgroup status of participants. Interrater reliability was established prior to the start of the study using five participants from the larger prevalence project. The intraclass correlation coefficients for the three subscales of neurological soft signs were 0.91, 0.82, and 0.9 for motor coordination, sensory integration, and disinhibition, respectively.
Data analyses
The prevalence of neurological soft signs (subscales and total soft signs) was compared between SPD-proneness and non-SPD-proneness participants using one-way ANOVA. Shapiro-Wilk tests showed the distribution of neurological soft sign scores were not normally distributed (all p < 0.001), thus we also used nonparametric Mann-Whitney test, results were similar to one-way ANOVA, we just present the results of ANOVA here. The correlations between schizotypal features and neurological soft signs were also analysed.
Results
Table 1 shows that no significant difference was found in age, education and IQ between the two groups (p > 0.05). Gender ratio was significantly different between groups (p = 0.005), however, gender difference was not significant on neurological soft signs (p > 0.05), thus gender was not considered in further analysis.
Demographic and clinical data for participants with SPD-proneness and non-SPD-proneness
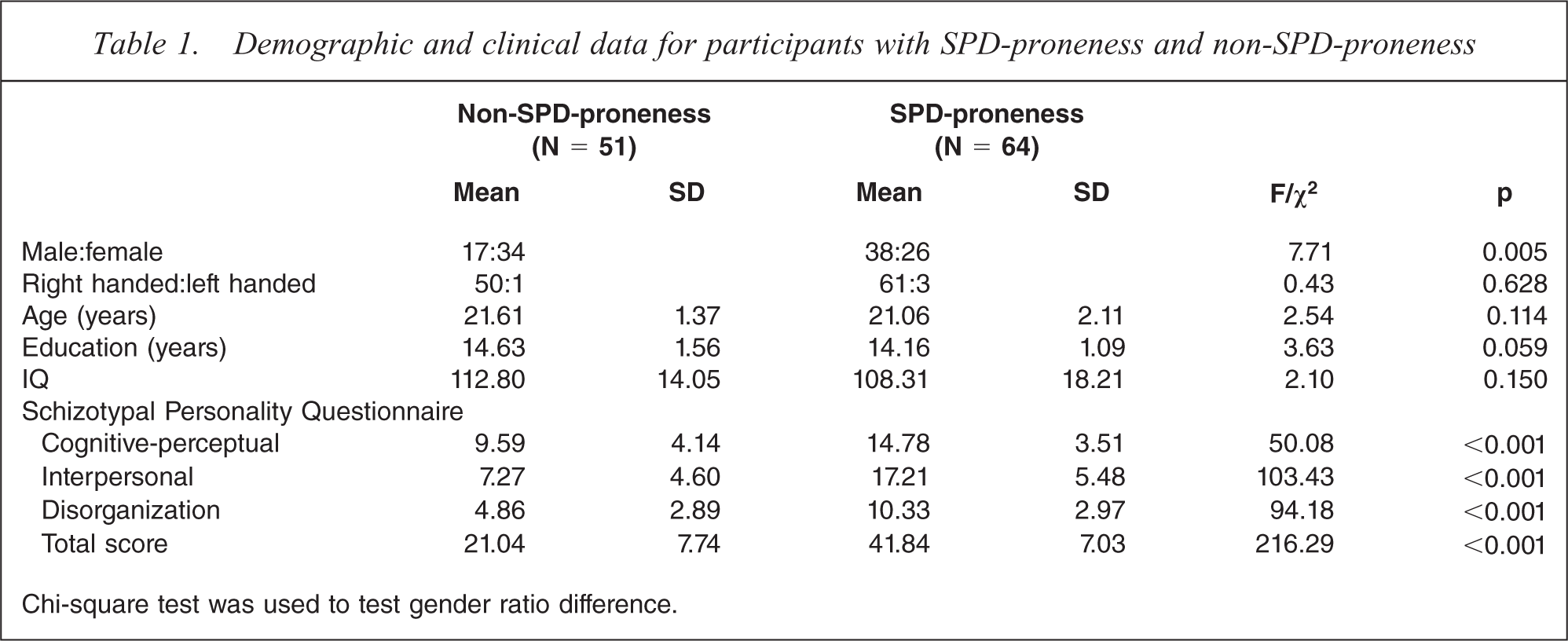
Chi-square test was used to test gender ratio difference.
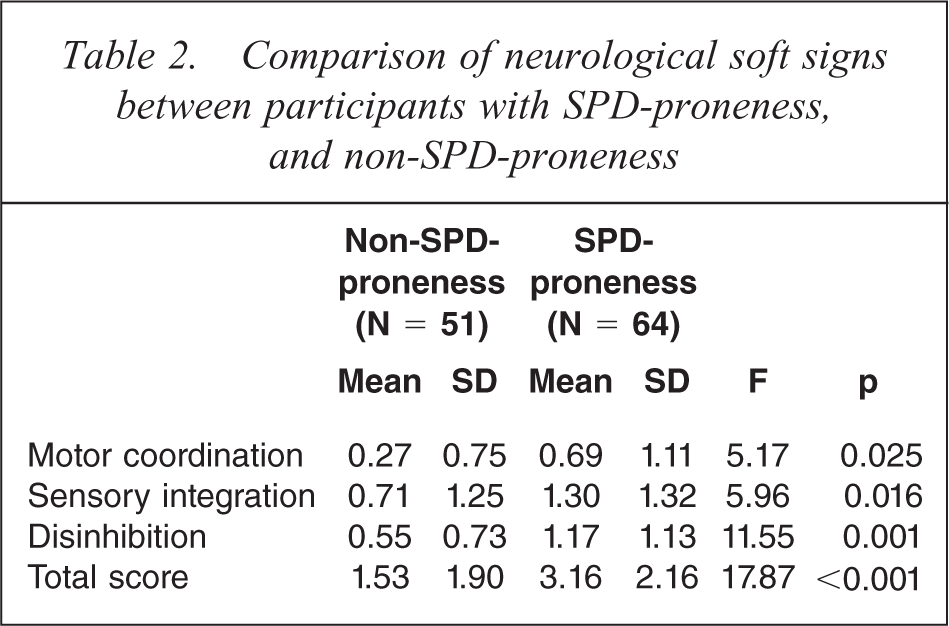
Subscales of neurological soft sign and total scores are summarized in Table 2. All subscales and total soft signs were significantly different between groups; with SPD-proneness participants showing more soft signs than non-SPD-proneness participants.
Comparison of neurological soft signs between participants with SPD-proneness, and non-SPD-proneness
Pearson correlation was conducted to examine the relationships between neurological soft signs and schizotypal features. Significant positive correlations were found between all neurological soft signs and schizotypal features. These relationships remained significant between motor coordination, sensory integration subscales and total soft signs after Bonferroni correction, i.e. p value of < 0.01 (Table 3).
Correlations between neurological soft signs and schizotypal features in individuals with schizotypal personality and non-SPD-proneness
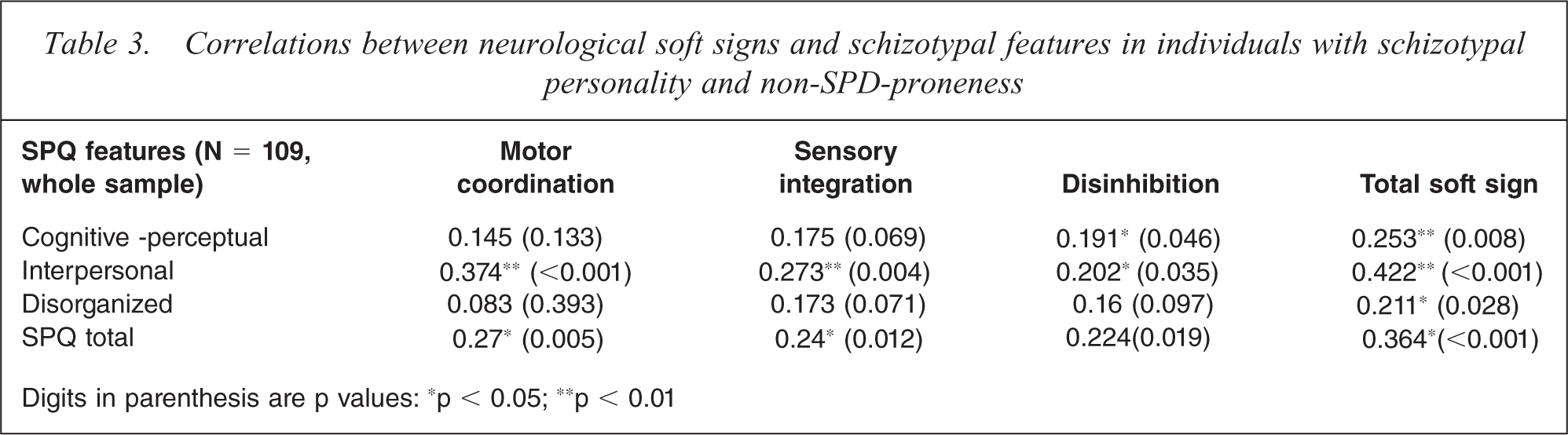
Digits in parenthesis are p values: ∗p < 0.05; ∗∗p < 0.01
Discussion
The main findings of the present study show that participants with psychometrically defined SPD features had a higher rate of neurological soft signs than non-SPD-proneness; SPQ factors were also found to be significantly correlated with motor coordination, sensory integration and total soft signs. The present findings of higher prevalence of neurological soft signs in participants with SPD features are consistent with a number of studies using different types of soft signs assessment [27–29]. Using the Neurological Evaluation Scale, Barkus et al. [27] demonstrated that participants with psychosis proneness exhibited a significantly higher prevalence of total neurological soft signs and a tendency towards higher motor coordination and sensory integration signs than healthy controls. Bollini et al. [28] further demonstrated that non-psychotic first-degree relatives of schizophrenia having SPD features also exhibited significantly higher rates of neurological soft signs compared to healthy controls. These findings are consistent with Barkus et al.'s [27] suggestion that the presence of neurological soft signs may be indicative of ‘gene-carrier’ status or a potential ‘endophenotype’ for schizophrenia.
This interpretation is supported by two pieces of evidence from the current study. First, the significant relationships between neurological soft signs and clinical symptoms of schizophrenia extended to individuals with SPD features. According to Gottesman and Gould [2], the potential endophenotype should be independent of the state of the stability of a given trait across development, and independent of florid clinical symptoms. Chen and Faraone [16] further commented that genetic vulnerability to schizophrenia may manifest itself in schizophrenia-like personality disorders, e.g., schizotypal, rather than a full syndrome of schizophrenia.
Second, neurological soft signs, particularly the motor coordination and sensory integration signs, were significantly associated with several endophenotypic markers for schizophrenia, including sustained attention [4,15,16,30–32], visual working memory [30,33], verbal memory [34], and inhibitory control [35], more neurological soft signs were related to worse cognitive functions. These findings suggest that there may be common neural substrates between these cognitive markers and neurological soft signs. A recent neuroimaging study demonstrated that motor coordination soft signs were associated with activation of the prefrontal cortex usually involved in complex neurocognitive and executive functioning in healthy controls [9]. Taken together, although we did not recruit any non-psychotic first-degree relatives of schizophrenia in this study, the significant differences shown in the different subscales of soft signs and total neurological signs between non-SPD-proneness, SPD-proneness cases and schizophrenia suggest that the presence of neurological soft signs may be a potential ‘endophenotype’ marker for schizophrenia [3,35,36].
The validity of the results of this study may be limited by a number of factors. Schizotypal personality features were assessed using a self-report approach. We did not include any semi-structured interview to ascertain a clinical diagnosis of SPD. Moreover, the SPD-proneness cases we recruited were limited to those exhibiting behaviourally related features. We did not include any genetically related SPD cases, i.e. non-psychotic first-degree relatives of schizophrenia demonstrating SPD features. However, as we argued above that very little information was known about these psychometrically defined SPD cases or the pseudo-SPD cases as framed by Raine [20], the current findings can bridge such a gap in the literature. Although these individuals do not suffer from any clinical symptoms of schizophrenia, they are still facing a lot of similar neurocognitive and behavioural difficulties in daily life. These findings suggest pseudo-SPD individuals do show similar neurological abnormality to individuals with schizophrenia in terms of neurological soft signs. Finally, our present SPD sample was mainly from urban and public sector settings, hence may not be representative of broader populations. Future studies should consider a more rigorous participants recruitment and clinical interview or diagnosis of SPD cases from the community.
In conclusion, participants with SPD-proneness demonstrated significantly higher prevalence of neurological soft signs than those with non-SPD-proneness. SPQ subscales were significantly associated with ratings of motor coordination, sensory integration and total soft signs. These findings suggest that neurological soft signs are trait features of schizophrenia.
Footnotes
Acknowledgements
The authors would like to thank Jifang Cui, Yongyu Deng, Xiaoyan Li, and Xiaojing Gao for their help with data collection and data entry. This study was supported partially by the Project-Oriented Hundred Talents Programme (O7CX031003), the Knowledge Innovation Project of the Chinese Academy of Sciences (KSCX2-YW-R-131), a grant from the National Science Fund China (30770723), and the National Basic Research Programme (973 Programme No. 2007CB512302/5).
