Abstract
The aim of the present study was to review and update historical controversies in relation to the limbic system in light of advances in the neuroanatmy and neurobiology of cortico-striatal–thalamic–cortical (CSTC) neural circuits. It is hypothesized that observed comorbidities between internalizing and externalizing childhood syndromes may be better understood in terms of excess communication between subcortical brain circuits. A neuroanatomical understanding of reciprocal and non-reciprocal connections between modular circuits provides a basis for understanding previously puzzling aspects of internalizing, externalizing comorbidity. Function depends on the degree of cooperative integration of goal orientation, Pavlovian stimulus response, and operant processing in cortical–subcortical circuits, and interventions may be targeted at any of these functions.
Keywords
Comorbidity between classes of child and adolescent psychiatry disorders has been frequently described [1]. Angold et al. reviewed the literature on the overlap between these conditions [1]. They pointed out that the way in which comorbidity is treated has changed over time. For example, the ICD-10 allows a mixed category of conduct and emotions, suggesting a failure of categorical diagnosis to appropriately describe separable syndromes on the one hand, and, on the other hand, an artefact of the failure to recognize the covariation between naturally occurring scalar syndromes. For example, between 50% and 80% of children with attention-deficit–hyperactivity disorder (ADHD) also met diagnostic criteria for other disruptive behaviour disorders, namely oppositional defiant disorder (ODD) [2] and conduct disorder (CD) [3], or for learning disorders and communication disorders [4]. There are also higher rates for internalizing problems such as anxiety [5].
Angold et al. conducted a meta-analysis of general population estimates of the strengths of associations between disorders [1]. The authors found that the association between CD and ODD was weaker than that between CD and depression. This argued against the existence of a unitary association between ‘internalizing’ and ‘externalizing’ disorders. While it has been argued that these comorbidity findings point to flaws in medical models of psychopathology, the investigators pointed out that the diagnostic literature has shown that associations between symptoms of different types occur at the extremes of psychopathology, and not only at the level of mild factor scores. Thus the issue for future research is to explore why symptoms group together in ways that they do, and why there is overlap between syndromes, whether defined by diagnostic criteria or factor scores.
Lahey utilized confirmatory factor analysis (CFA) of twin data in a large representative sample of 4069 6–17-year-old twins, in 28 urban, suburban and rural counties in Tennessee, to investigate a dimensional model of DSM-IV symptomatology [6]. They utilized adult caregiver and youth interviews on the Child and Adolescent Psychopathology scale. The symptom dimensions were organized hierarchically within higher-order externalizing’ and internalizing’ dimensions. Of present interest, the investigators found that for major depressive disorder (MDD)/generalized anxiety disorder (GAD) the higher-order CFA model that fitted best, loaded on both internalizing and externalizing dimensions, indicating an overlap between the traditional separation of these dimensions.
It is hypothesized that a neuroanatomical understanding of reciprocal and non-reciprocal connections between modular circuits could provide a basis for understanding previously puzzling aspects of comorbidity between internalizing and externalizing childhood syndromes, which have been regarded as orthogonal.
Limbic system controversies
Papez proposed that a circuit consisting of the hypothalamus, anterior thalamic nuclei, gyrus cinguli, and their connections constituted a mechanism that might elaborate the functions of central emotion, as well as participate in emotional expression [7]. This concept was further elaborated by MacLean, who added the amygdala to the ‘visceral brain’, and coined the term ‘limbic system’ [8,9]. The functions and boundaries of the limbic system, however, have remained controversial. Most of the neocortex was known to project to basal ganglia, but not to hypothalamus, leading to an important perceived distinction between the limbic system and basal ganglia (motor vs emotional or visceral brains).
Heimer et al. claimed that the Papez/MacLean concept of a limbic subcortical/hypothalamic emotional system [8,9] is incorrect, and has remained ill-defined and confused [10]. Heimer et al. believed that Papez and MacLean failed to realize that the major output of their limbic systems was to the basal forebrain, rather than the hypothalamus. Hypothalamic output is believed by them to yield deterministic and stereotypic outputs, be they neural, autonomic or endocrine. Heimer et al. described a greater portion of our behaviour as non-deterministic and governed by genetic predisposition, developmental learning, reward, and social norms [10].
According to Heimer et al., the neuroanatomical discovery of the ventral striatopallidal system, and revelations that the ventral striatopallidal system projects to the mediodorsal thalamus, marked the beginning of the notion of parallel cortico-subcortical re-entrant circuits, thought to relate to motor, prefrontal and limbic cortical areas [10]. Heimer et al. suggested that two central concepts, namely ‘ventral striatopallidum’ and ‘extended amygdala’ provide the basis of a neuropsychiatric approach to psychopathology. Thus the finding that the ‘ventral parts of the striatal complex, including the accumbens and striatal territories in the olfactory tubercle receive prominent allocortical (i.e. “limbic” cortical) inputs, and the dorsal parts of the striatal complex receive massive projections from the neocortex (isocortex)’ was thought to demonstrate a fundamental pattern of forebrain organization, namely that ‘the pattern of neural connections that characterizes basal ganglia is also expressed by “limbic” structures’ [10]. (see Figure 1)
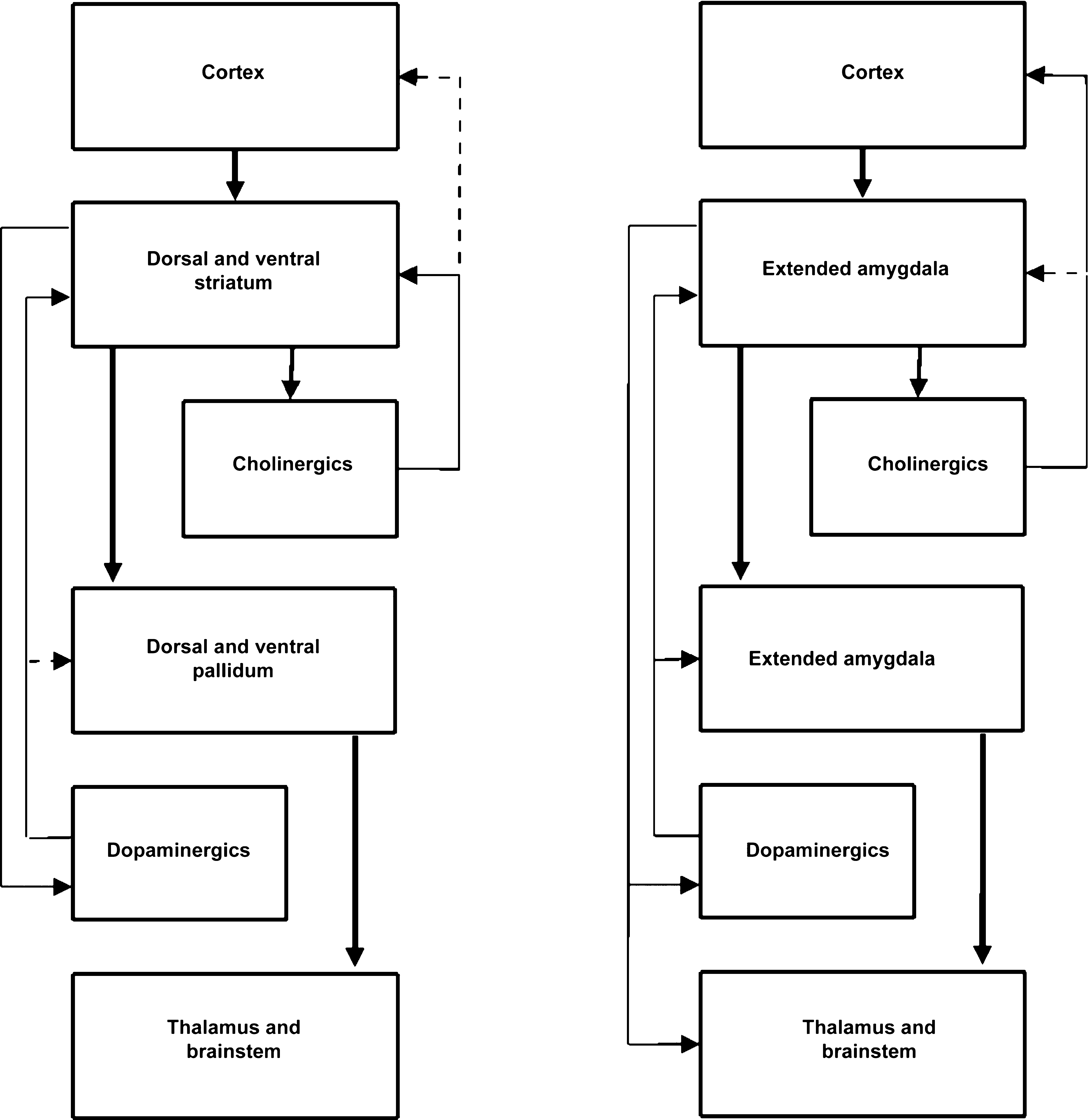
Striatopallidum and extended amygdala (adapted with permission from Heimer et al. 2008 [10])
LeDoux based his studies of fear conditioning involving the amygdala as comprising an important component of the system involved in the acquisition, storage and expression of fear memory, utilizing experimental approaches. He pointed out that limbic system concepts have survived because the anatomical concept and the emotional functions it was supposed to mediate were so vaguely defined as to be irrefutable [11].
Cortico-striatal–thalamic–cortical loops
Stahl described cortico-striatal-thalamic-cortical (CSTC) loops as a very good example of how cortical engines in different topographical brain regions not only drive neuronal structures throughout the brain, but also receive feedback from them [12]. Each CSTC loop starts and ends in a pyramidal cortical cell, shaped like a triangular pyramid, and having extensively branched spiny apical dendrites and shorter basal dendrites. Pyramidal cells are located in four of the five vertical cortical laminae. The excitatory neurotransmitter output from most pyramidal cells is glutamate, while the input from the various brain regions is inhibitory GABAergic (gamma-aminobutyric acid). The inhibitory GABAergic interneurons are described as basket neurons, which input to the end of an apical dendrite in the pyramidal neuron. A second inhibitory neuron can inhibit the inhibition or disinhibit the pyramidal neuron. Thus the presence or absence of GABA tone on the pyramidal neuron can have a profound influence on the ability of that cortical pyramidal neuron to serve as the driver of a cortical engine that delivers the behavioural program of the brain. Stahl [12] also describes cortical pyramidal neurons as receiving many excitatory inputs, coming predominantly to synapse with the apical dendrites, and utilizing the excitatory neurotransmitter glutamate. Thus it is easy to see how the cortical pyramidal neuron can either be excited by these long-distance glutamate inputs, or inhibited by short-distance GABA inputs [12].
Similarly the same type of loop arising from the dorsal anterior cingulate gyrus (ACC) modulates selective attention and self-monitoring of performance, via the ventral region of the thalamus. A third CSTC loop arises from the ventral or subgenual part of the ACC, and projects to a part of the striatal complex, the accumbens, extending to the thalamus, and back to the subgenual ACC. This loop is thought to regulate emotions, including depression and fear. A fourth CSTC loop outputs from the orbital frontal cortex (OFC) to the ventral part of the caudate nucleus in the striatal complex, to the thalamus and back to the OFC, and is believed to regulate impulsivity and compulsivity. Finally, a fifth loop starts in the supplemental motor region of prefrontal motor cortex, and projects to the putamen in the lateral part of striatal complex, then to the thalamus, and back to the premotor cortex, and may modulate overactivity, psychomotor agitation and psychomotor retardation. These loops might also be deemed executive, attentional, emotional and motor modules [12].
According to Stahl, cortico-cortical circuits link neurons together into functional loops [12]. Thus while pyramidal cortical cells are tuned by neurotransmitters from below, cortical circuits may allow a third intermediary neuron to have positive or negative effects. Stahl has also described an important cortical circuit called the CSTC circuit, allowing information to be sent downstream, and yet get feedback on how information was processed. Thus the loop through the striatum may have a synapse through another part of the striatal complex, before it leaves to go to the thalamus, which relays back to the original region of prefrontal cortex (PFC). For example, the dorsolateral prefrontal cortex (DLPFC) projects to the rostral part of the caudate, within the striatal complex, then to the thalamus, and back to the DLPFC. Loops with this structure are thought to regulate executive functions, problem solving, and cognitive tasks, such as representing and maintaining goals, and allocating attentional resources to various tasks.
The concept of basal forebrain functional–anatomical systems or macrosystems is described by Zahm as providing a more comprehensive understanding of cerebral structure and function [13]. He pointed out that structural similarities shared by different macrosystems are reflected in a basic framework. The major macrosystems are: the dorso-striato-pallidal system (or the basal ganglia as classically described); the ventral striato-pallidal system (which includes the accumbens); and the extended amygdala (which includes the central nucleus of the amygdala). Structural similarities shared by the different macrosystems are reflected in a basic framework, essentially that of the basal ganglia, in which massive projections from the cortical mantle or cortical-like structures (which include the lateral amygdala) terminate densely in subcortical input structures, consisting predominantly of medium-sized densely spiny inhibitory GABAgergic neurons. Importantly, macrosystem outputs diverge into re-entrant pathways to the forebrain, including cortex, via synaptic relays in the thalamus, forebrain and brainstem, and descending pathways to somatic and autonomic motor effectors, via relays in the hypothalamus, mesopontine tegmentum and caudal brainstem.
Haber has demonstrated that within each region of cortico-basal connections, linking up regions with similar functions (maintaining parallel networks), there are, in addition, non-reciprocal connections associated with differing cortico-basal ganglia circuits [14]. Haber pointed out that the development and modification of goal-directed behaviours require continual processing of complex chains of events, reflected in the feed-forward and feedback organization of both striato-nigral connections and thalamo-cortical connections. Where parallel and integrative circuits are not coordinated, maladaptive syndromes may result.
Haber and McFarland have discussed direct and indirect output routes from the thalamus (the direct pathway from the striatum to the internal segment of the globus pallidus (GPi) and substantia nigra (SNr) pars reticulata to the thalamus), and indirect route (from the external segment of the globus pallidus (GPe) to the subthalamic nucleus to the GPi, and from the GPi to the thalamus) [15]. They noted that the complexity of the thalamo-cortico-thalamic circuit raises an issue related to the concept of parallel versus non-parallel processing of information through basal ganglia pathways. Because there are both reciprocal and non-reciprocal cortico-thalamic relay nuclei from basal ganglia output structures, the information that the relay nuclei convey to the cortex is not only affected by the parallel path through the basal ganglia structures, but is also modified by the feed-forward component of the non-reciprocal cortico-thalamic pathway, allowing influence from other functional regions of the cortex. Because these functional cortical regions are also, according to the authors, involved in their own basal ganglia loop, the transfer of information occurs between different functional basal ganglia loops in the final pathway to the cortex.
Haber and McFarland described a tight anatomical and functional triad between the cortex, basal ganglia, and thalamus, with the same regions of the thalamus that convey basal ganglia output to the cortex, and projecting back to the striatum [15]. These thalamic nuclei therefore play a dual role in basal ganglia circuitry; both to relay basal ganglia output to frontal cortical regions, and provide direct feedback to the striatum. The authors speculated that thalamic input might sustain disinhibition of specific thalamo-cortical circuits, which would release responses and keep a behaviour focused on a task until its goal is achieved, or alternatively it may further reinforce or facilitate selection of basal ganglia circuits to enable desired and suppress unwanted behaviours. The control of adaptive motor behaviour thus results from a complex interplay of excitatory and inhibitory reciprocal and non-reciprocal cortico-thalamo-striatal circuits.
Dual cognitive process
According to Koziol and Budding, most behavioural processing can be categorized as stimulus-based, or higher-order control respectively. Stimulus-based control is thought to consist of habits, skills and procedures, which allows for a high speed of reaction [16]. When problem-solving and planning are required, however, higher-order processing is required, and the authors describe higher-order control as allowing autonomy by breaking a problem down into stimulus-based characteristics, and thus programming goal-directed activity. Koziol and Budding believed that the fronto-striatal system has evolved to operate both stimulus-based control, when this is advantageous, and higher-order control, when automatic processing is not sufficient, and that both systems interact with each other. Thus, while procedural learning and memory lie at the heart of automatic thought, a major evolutionary trend is the progressive involvement of the cortex in the processing of the thalamic sensory information, projected from cortex to basal ganglia to thalamus and back to cortex, via parallel segregated circuits. ‘As the neocortex dramatically increased in size and complexity throughout evolution, the basal ganglia are thought to have become more specialized, receiving specialized information from cortex, and sending information back to cortex via thalamic relays’ [16].
Koziol and Budding postulated that higher-level functions of the cortex are expressed through lower level systems, and these lower level systems are critical in modulating cortical functions. Cortical and subcortical functions operate in concert in deciding upon behaviours that are in the best interests of the organism, which ensure survival. This is because the basal ganglia and cerebellum play important roles in deciding what information is or is not used’ by the cortex. The authors described a stimulus-based processing system, composed of innate behaviour processes and behaviours that run on the basis of acquired associations, allowing for automatic processing when appropriate. The second system of higher-order control is thought to allow for novel problem-solving, thus providing flexibility in interacting with changing and unpredictable environments [16].
This raises the question of the relationship of comorbidity to modular subcortical vs cortical processes. Comorbidity between motor and attention syndromes may be partially explained by connections between parallel modular pathways. In contrast, comorbidity between internalizing and externalizing syndromes suggests connections with motivational circuits, challenging the traditional modular separation of these syndromes. Heimer et al. have discussed the degree of segregation of cortico-basal ganglia-thalamocortical circuits or loops [10]. They noted that although it may be tempting to overstate their closed and segregated nature, the work of Haber has suggested substantial feed-forward routing of impulse conduction from limbic lobe circuits, via executive circuits to cortical motor circuits [14]. According to Heimer, [10] similar neuroanatomical considerations [17–19] suggest a general flow of information from ventromedial to dorsolateral parts of the striatal complex consistent with the idea of a passage of information from motivational to motor parts of the cortico-basal ganglia-thalamocortical circuitry.
In an extensive review Balleine and O'Doherty have described two different learning processes; one encoding the relationship between action and consequences (operant conditioning), and the second involving the formation of stimulus–response (Pavlovian) associations [20]. They suggested that the basal ganglia are able to maintain these functions in parallel, but allow under some conditions one or other or both processes to exert cooperative control over choice and decision making. The authors describe progressive involvement of different neural networks, during different stages of learning, involving both feed-forward and negative feedback from striatum to Dopamine (DA) neurons on one hand, and potentiation of the DA signal through disinhibitory projections on the neighbouring cortico-basal ganglia neurons to regional interneurons on the other. While subject to ongoing neuroscientific investigation, the degree of cooperative integration of stimulus–response and operant processing may be of importance for comorbid symptomatology [20].
Cognitive/motor comorbidity
Comorbidity of Tourette's Disorder (TD) with hyperkinesis and obsessive-compulsive disorder (OCD) has often been described. Towbin et al. noted that leaving aside the chance that a diagnostic error has occurred, there are three possible relationships that may exist between two disorders when they are observed in one person [21]. The first is that they are entirely separate entities, running independent courses (true comorbidity). The second is when one disorder sets the stage for another, such as an opportunistic fungal infection, but both are aetiologically independent. The third case is when two disorders are aetiologically related, but are differentially expressed (such as peripheral neuropathy and retinal haemorrhages in diabetes).
Leckman and Cohen noted that complex tics can be extremely difficult to distinguish from compulsions, and that 30–60% of Tourette's patients are also diagnosed as having OCD [22], with some studies suggesting genetic relationships [22,23]. Leckman et al. claimed that approximately 50% of all clinically identified Tourette's syndrome patients also have ADHD [22]. While ascertainment bias may account for some of these relationships, Leckman attributed the underlying pathology of these conditions to parallel CSTC circuit pathology [24–26]. According to Leckman and Cohen, current consensus holds that CSTC circuitry had at least three components, those initiating from and projecting back to sensory-motor cortex; orbitofrontal cortex; or association cortices. Further components were also thought to include limbic system components [22].
As described by Leckman and Cohen, cortical projections to the caudate and putamen (striatum) appear oriented in somatotopically organized domains. Information leaves the basal ganglia principally through the internal segment of the GPi and its brainstem counterpart (SNr) before ascending to the thalamus and cortex [22]. Striatal projections to the external segment of the GPe then project to the reticular thalamic nucleus and internal segment (GPi), exerting inhibitory influences modulated by GABA. Projections from striatum to pallidum are also GABAergic, and increased striatal activity (such as associated with movement) will phasically silence the tonically active GPi and SNr neurons, reducing GABAergic transmission to the thalamus, thereby activating thalamic target neurons. Leckman and Cohen described human studies supporting the importance of basal ganglia in Tourette's syndrome pathophysiology [22,27]. The CSTC, however, is also implicated in OCD [28].
Leckman and Cohen described the limbic system as consisting of the amygdala and hippocampus, the temporal lobe, the cingulum, the caudate nucleus and other basal ganglia structures in the subcortex, the hypothalamus and periaqueductal grey matter in the brainstem and connections with associative frontal cortex [22]. The involvement of the cingulate cortex in both Tourette's and OCD is described as inhibitory. Decreases in severity of OCD symptoms have been correlated with decreases in right anterior cingulate metabolic activity during successful medication therapy [28]. Finally, abnormalities in striatal function and related association cortex circuitry are thought to contribute to ADHD symptomatology [22,29]. Leckman and Cohen also suggested that limbic system circuitry, including the temporal lobe, may be involved in the sexual and aggressive content of many complex motor and vocal tics, while the sexual and aggressive content of many obsessions and compulsions suggests involvement of amygdala and related circuitry [22].
Towbin et al. examined the relationships between ADHD, OCD and Tourette's syndrome [21]. The authors noted that a categorical classification would view the three disorders as separate, whereas a concept of overlapping mechanisms, evolving from impairment at related anatomical sites or systems (such as the CSTC system) provides a better explanation of comorbidity.
Tics are thought to reflect a lack of balance (striasome vs matrix). According to Leckman, alterations in the structure of the basal ganglia, including loss of right–left symmetry, and cortico-thalamic inputs may be important in tic and hyperkinetic disorders [30]. Recent neurosurgical procedures have reinforced the functional importance of thalamic regions, which are part of the above cortical–subcortical loops. Additionally, ascending dopaminergic pathways are thought to have a role in the consolidation and performance of tics. Leckman noted that early results of in vivo neuroimaging studies have shown that voluntary tic suppression involves activation of regions of the PFC and caudate nucleus and bilateral deactivation of the putamen and GPi [30].
Although the aforementioned syndrome descriptions appear to reflect a modular, motor syndrome, the association of premonitory sensory urges, the presence of motor and phonic tics, as well as coprolalia, and associations with hyperkinesis and OCD suggest greater complexity. The division of tics into those with and without cognitive phenomena suggests that TD may be characterized with and without cognitive phenomena [30]. Haber described the mediation of cognitive and motor planning, and drive behaviours, in relation to these phenomena [14], by the parallel cortical–subcortical neural circuits originally described by Alexander et al. [31]. The Alexander et al. model, however, fails to address how information flows between circuits. Haber noted that recent anatomical evidence from primates demonstrates that neuron networks within the basal ganglia are in a position to move information across functional circuits. She described two networks: the striato-nigral, and the thalamo-cortical-thalamic network. Importantly, within each of these nets of connected structures, Haber described both reciprocal connections, linking regions associated with similar functions, and non-reciprocal connections linking regions that are associated with different cortico-basal ganglia connections. Thus each component of information (from limbic to motor outcome) sends feedback and feed-forward connections, allowing the transfer of information, which may help explain comorbid symptomatology in Tourette's disorder [14].
Thus Tourette's disorder, OCD, and ADHD could be conceptualized as manifesting respectively more or less cognition, as the involvement of orbitofrontal cortex in the case of OCD increases, with more or less affective reactivity as the involvement of limbic and temporal and cingulate lobes increases. This concept also applies to dorsal (cognitive) and ventral (affective) systems and symptomatology.
Motor/affective comorbidity
Heimer has described the ventral striatal (including accumbens), pallidal system and extended amygdala as the major components of the ‘new anatomy’ of the basal forebrain [32]. He described three re-entrant circuits: the anterior cingulate, the lateral orbitofrontal, and the medial orbitofrontal, which are closed in that they originate and terminate in the same region of the frontal lobe, but suggested that they can also interact. He suggested that the entire cerebral cortex including the hippocampus and major parts of the amygdala project to the basal ganglia and are involved in the regulation of specific emotional functions and adaptive behaviours ranging from fear–anxiety, and addictive–reward and appetitive behavior. The ventral striatum integrates various cortical and subcortical inputs to adapt motivational behavior in a similar way to that in which the motor loop through the dorsal parts of the basal ganglia is important for movement control.
Levy has discussed the role of the nucleus accumbens in gating anxiety levels. [33]. Mogenson et al. described the role of the nucleus accumbens in functioning as an interface between limbic and motor systems (‘from motivation to action’) [34]. Mogenson et al. described the nucleus accumbens as a key structure in linking motivation and action at the interface of the limbic system with motor mechanisms, receiving direct connections from amygdala, hippocampus and other limbic forebrain structures, as well as indirect connections via mesolimbic dopaminergic projections from the ventral tegmental region. The nucleus accumbens has direct motor connections to the GPi and indirect connections via the SNr and nigrostriatal dopaminergic system [35]. Heimer et al., however, commented that the concept of an “emotional motor system” like the limbic system oversimplifies basal forebrain systems [10]. While the functional areas are broadly thought to relate to motor, prefrontal and limbic cortical areas, the separation of functional domains is described as blurred, because the cortico-striatal projections from adjacent functional domains within the striatum overlap extensively with each other. ‘Limbic lobe projections (related to emotional-motivational functions) overlap with dorsal prefrontal projections (serving cognitive-executive functions). Dorsolateral prefrontal projections to striatum, likewise, overlap with projections from motor cortical areas [10].
Ventral circuits
The ventral striatum including the nucleus accumbens receives input from cortical regions other than the motor and sensory regions of the frontal and parietal lobes. It receives input mainly from regions that do not project to the dorsal striatum, notably the temporal (including the hippocampal formation), limbic, and orbitofrontal cortical regions as well as the basolateral amygdala. The ventral striatum in turn projects to the ventral pallidum. Information is then relayed via the dorsomedial and ventral anterior nuclei of the thalamus to the cingulate, orbitofrontal cortex and PFC and to the ventral tegmental region of the brainstem. The ventral circuitry parallels the dorsal circuitry and is thought to be involved with behavioural phenomena, reward and punishment and in integrating cognitive with emotional responses. Thus the ventral striatum, including the accumbens, is thought to be critical in integrating cognitive and emotional responses [10,12].
According to Grace [36,38,39], the amygdala is involved in emotional or affective properties of stimuli, enabling the subject to respond to events that are emotionally charged, and therefore of immediate survival value. In pathological states, the amygdala input may be overdriven to the extent that the maintenance of focus is overly disrupted by minor events. As described by Arnsten, high levels of dopamine and norepinephrine (NE) may have additive effects on information processing in PFC, reducing signals and increasing noise [37]. Arnsten noted that although PFC functions are often essential for successful organization of higher-order behaviour, there may be some conditions when it may be adaptive to shut down these complex, reflective operations and to allow more automatic or habitual responses, dependent on posterior cortical or subcortical structures to control behaviour.
The nucleus accumbens is described as the striatal region in which the limbic system has overlapping inputs. Accumbens neurons exist in a bistable state, with their membrane potential alternating between a hyperpolarized non-firing state and a depolarized plateau lasting several hundred milliseconds, during which spike activity is generated, allowing affective neurotransmission [36]. Goto and O'Donnell have reported timing-dependent limbic–motor synaptic integration in the nucleus accumbens [40]. In the current context the demonstration of differing effects of coincident and asynchronous PFC and limbic activity suggests a possible gating mechanism, whereby PFC activity can gate limbic input depending on the state of the neurons and the timing of inputs.
Fear models
Olsson and Phelps have pointed out that insight into the workings of social fear learning and neural mechanisms allows integration of biological principles of learning with cultural evolution [41]. They noted that research in the neurobiology of fear conditioning has focused on the amygdala in the medial temporal lobe as a key structure in the brain's circuitry. This is thought to involve sensory input and motor output systems, as well as regions that contribute to explicit and conscious aspects of learning and expression of fear. The basolateral complex in the primate amygdala has strong connections to visual cortex, in particular to the inferotemporal cortex region that responds to face identity and to facial expression. The authors also suggested that the hippocampus, another medial temporal lobe structure adjacent to the amygdala, is critical for coding contextual information about the fear learning situation, such as relationships between different features and the timing of events. Thus, while the amygdala is responsible for forming associations between somatosensory states and representation of individual stimuli, the hippocampus is important for encoding relations between the various cues that comprise the learning context.
Olsson and Phelps suggested two interacting mechanisms in social learning of fear [41]. While the amygdala is believed to process and store representations of conditioned–unconditioned contingencies, via connections with the medial PFC, observational fear learning is believed to be supported by shared affective representations in the anterior insula, as well as more explicit hippocampal representations about context and relevant social information, such as social status and familiarity [41]. They also suggested that for fears that are acquired through verbal communication, the conditioned stimulus–unconditioned stimulus (CS-US) association is represented in a distributed cortical network, which is left-lateralized, reflecting the verbal nature of the CS. The authors also noted that animal studies of fear conditioning have not emphasized the striatum, beyond its role in avoidance learning and active coping [42]. Although unidirectional projections have been emphasized, most of the regions involved have bidirectional connections with the amygdala. Thus, while fear learning is first expressed after social and non-social means of acquisition, further learning may result, which could change the nature of the representation further, facilitating an amygdala-dependent representation of the CS-threat association that was not present after initial verbal instruction [41,43].
Ramos and Arnsten have noted that NE has widespread projections throughout the brain, and is thus ideally positioned to orchestrate neural functions based on arousal state [44]. Thus NE can increase the signal/noise ratio in the processing of sensory stimuli, and thus enhance long-term memory consolidation in the amygdala and hippocampus, through actions at alpha-1 and beta adrenoreceptors. In addition, NE has been shown to play a powerful role in regulating the working memory and attention functions of the PFC. The modest levels of NE released under controlled conditions strengthen PFC functions, via actions at post-synaptic alpha-2 adrenoreceptors, while high levels released during stress impair PFC cortical functions via alpha-1 and possibly beta-1 receptors. Thus levels of NE can rapidly determine whether prefrontal cortical or cortical systems control behaviour and thought. Thus, while tonic/phasic NE mechanisms are able to rapidly switch behavior to fast-acting posterior cortical/amygdala control, slower acting dopaminergic tonic/phasic mechanisms are involved in prefrontal control of working memory and sequential behaviour.
Herba and Phillips discussed developmental studies of amygdala activation to fearful faces, and pointed out that with age, there is increased prefrontal and decreased subcortical activity [45]. Thus, early in development, children may have difficulty labelling neutral faces, which may be interpreted as ambiguous, giving rise to amygdala activation, whereas adults show greater amygdala activation in response to fearful facial expressions. Skuse has described an important evolutionary connection between emotions, feelings and ability to interact appropriately in social situations [46]. He noted that the amygdala responds specifically to eye contact in adults and is maximally activated by exaggerated wide-open eyes, associated with fearful expressions. Thus direct eye contact in primates can elicit an instinctive fear response of fight or flight. In humans, however, response to a stimulus that could be a threat is normally determined by a full evaluation of that stimulus by means of complex neocortical connections, allowing an appropriate social response. According to Skuse, a crucial component of the modulating circuitry in humans is the recruitment of language centres, and the conscious processing of a ‘feeling’ response.
Thus neural circuitry that evolved for the purpose of fear detection in others’ faces is now associated with the development of social skills. Interestingly, some of the ventral forebrain structures involved in these social functions resemble those originally described by Papez and McLean, such as amygdala, thalamus and medial frontal cortex, but project principally to the thalamus en route to cortex, and not to the hypothalamus.
Post-traumatic stress disorder models and comorbidity
The switching mechanisms described for post-traumatic stress disorder (PTSD) may also be involved in comorbidity, when very high arousal levels result in failure of homeostatic mechanisms at cortical levels, allowing direct transmission of fear-related stimuli from the amygdala to autonomic and motor centres [41]. The regulation of stress is carried out by a hormonal neural system consisting of the hypothalamic–pituitary–adrenal axis. Corticotropin-releasing factor is released from the hypothalamus, stimulating adrenocorticotropic hormone release from the pituitary, resulting in release of cortisone, which feeds back to the hypothalamus, and also to the hippocampus [41].
Noradrenaline (NA) is also released from the locus coerulus during stress. Animal studies have shown that the hippocampus, which is involved in verbal declarative memory, is very sensitive to the effects of stress. In addition the amygdala is involved in memory for the emotional valence of events, and is involved in the acquisition of fear responses. In addition, the medial PFC (including anterior cingulate, subcallosal gyrus and orbitofrontal cortex) modulate emotional responsiveness through inhibition of amygdala responsiveness. According to Bremner et al., ‘studies in patients with PTSD show alterations in brain regions implicated in animal studies, including amygdala, hippocampus and PFC, as well as in neurochemical stress response systems, including cortisol and norepinephrine’ [47].
Schulkin et al. have provided a framework that integrates animal studies on the regulation of chronic anxiety, fear and helplessness with human fear, anxiety and PTSD, all of which may result from anticipation of negative events (allostasis) [48]. They discussed the role of the central nucleus of amygdala in expectations of adversity giving rise to elevated glucocorticoid secretion. They argued that increases in corticotropin-releasing hormone caused by stress or glucocorticoids in the amygdala may affect allostatic load.
McFarlane et al. have noted that the emergence of PTSD with intrusive and distressing recollections is a progressive rather than immediate process, in which a traumatic event is a necessary but not sufficient explanation [49]. The authors have developed a model that incorporates neural network models, as well as modelling the manner in which complex associative networks are trained and modified (similar models can also be applied in OCD, schizophrenia and dissociative disorders and possibly autism). The disruption of cortical/subcortical connections is critical to the development of the model, which suggests that an individual's cognitive and affective adaptability becomes impaired by the dominance of internal memories. This occurs as a consequence of primary and top-down activation, which in turn interferes with the development of semantic networks to explain and integrate the trauma. The neural network model of parallel distributing processing attractor networks implies that memory and cognition reflect the cooperative interaction of multiple cortical regions rather than hierarchical control [49].
McFarlane et al. cited the work of Goldman-Rakic [50] on functional reciprocity of frontal and posterior association cortex, with complementary connection to up to 15 other cortical and subcortical regions [49]. The authors describe three processes characteristic of neural networks of interest in the study of PTSD. These are (i) interactive learning; (ii) pruning of neural interconnections, in which some connections die as a result of competition; and (iii) top-down activation, in which dominant or inflexible networks prime or bias activation toward stimuli relevant to certain memories. Thus a consequence of iterative learning is the facilitation of some pathways at the expense of destroying or inhibiting others by preferential adjustment of the synaptic strength between neurons. The authors suggest that the repeated replaying and contemplation of the traumatic memories modify associated fear networks [49].
McFarlane et al. also discussed the role of the NA system in PTSD. They noted that the central NA system is one of the few systems in which the afferents have widely dispersed projections [49]. Furthermore, the function of the NA system has been conceptualized as manifesting tonic/phasic activity. According to McFarlane et al., the phasic mode of activity promotes focused selective attention, whereas the tonic mode leads to attentional scanning, with associated effects on behavioural flexibility. Thus the interaction between the two states affects signal to noise ratio. The wide receptive domains of the NA system, originating from the locus coerulus domains of the NA system, carry non-specific information to many regions, thus integrating and modulating other transmitters and brain regions. The NA projections in the PFC are mediated by alpha-1 and alpha-2 receptors [49]. According to Friedman et al., the alpha-2 receptors improve prefrontal functions by inhibiting irrelevant and distracting sensory processing through the modification of prefrontal connections with the sensory association cortices, which in turn improves the cognitive processing of relevant information [51]. In contrast, as described by Arnsten et al., the alpha-1 receptors in the PFC inhibit cognitive performance in the PFC, possibly to allow rapid use of habitual subcortical routines in the face of danger [52].
In the present context, a hyper-catecholaminergic state in PTSD releases the inhibition typically maintained by the PFC, thereby releasing traumatic memories. As in the DA system, Aston-Jones et al. [53] suggested an inverted U relationship [54] between tonic locus coerulus activity and performance on discrimination tasks, where moderate tonic activity in association with phasic responses to targets leads to optimal performance. Thus in PTSD, the NA system is overactivated, resulting in suboptimal performance. This overactivation is thought to underlie symptoms such as flashbacks and intrusive thoughts [55].
According to McFarlane et al. the neural network model helps explain how the same neurotransmitter can produce both symptoms related to overactivity and to underactivity [49]. For example, the loss of the normal flexible modulation of parallel distributed processing by cortical networks may result in overactivity in phasic states and underresponsiveness to non-specific sensory stimuli, creating an environment of sensory understimulation and sensory deprivation.
McFarlane et al. suggested that in PTSD the loss of too many dendrites by pruning can result in the development of autonomous foci, whereby they can become activated in the absence of environmental primes, as well as leading to the inappropriate fusion of memory networks [49]. This may explain several phenomena ‘including the emergence of flashbacks and the development of dissociative reactions in response to traumatic triggers’. ‘Pruning can also explain how a particular traumatic event could serve as an activator of previously traumatic memories and create a domino effect in the processing of similar traumata’. ‘Finally, pruning has a secondary effect on general working memory structures and systems. For example, parasitic foci may have the ability to assert their influence over other concurrent inputs with the secondary effect of inducing systemic inactivity (with loss of flexibility)’ [49].
Phelps describes the amygdala and hippocampal complexes as two independent memory systems, which interact in subtle but important ways [56]. The amygdala is described as more or less specialized for the processing of emotion. According to Phelps, most research has focused on how the amygdala can influence hippocampal-dependent episodic memory for emotional stimuli. ‘It has been shown that there is an enhanced response in the amygdala to emotional stimuli (e.g. fearful faces) and this response is correlated with a similar response in the visual cortex’ [55,56].
The McFarlane et al. model of PTSD is particularly cogent for the present understanding of symptomatology [49]. The stress response system demonstrates the principle that when homeostasis of an iterative system is disrupted (in this case by an exaggerated amygdala response and diminished PFC control), a repetitive behavioural syndrome (which can go on for many years in terms of flashbacks and anxiety) results, possibly as a continuing attempt to restore homeostasis. The process of circuit formation in PTSD appears to be in an opposite direction to the process of normal development. In the latter there is a progression from modular to more domain-general or integrated information processing, while in PTSD there is a progression to more modular and fragmented processing.
Conclusion
The paradox of comorbid internalizing and externalizing behavioural phenotypes is better understood in terms of excess communication between subcortical circuits, possibly as a result of insufficient cortical inhibition, as well as excessive reliance on stimulus-based subcortical systems. A neuroanatomical understanding of reciprocal and non-reciprocal connections between modular circuits provides a basis for understanding previously puzzling aspects of comorbidity between internalizing and externalizing childhood syndromes, which have been regarded as orthogonal. Structure, however, does not determine function, which may depend on the degree of cooperative integration of goal orientation, stimulus response, and Pavlovian versus operant conditioning, involving both subcortical conditioning and cortical inhibitory processes. Interventions may be targeted at any of these functions.
Footnotes
Acknowledgements
