Abstract
Brain asymmetry, involving structural and functional differences between the two hemispheres, is a major organizational principle of the human brain. The structural and functional connectivity within each hemisphere defines the hemispheric network or connectome. Elucidating left-right differences of the hemispheric network provides opportunities for brain asymmetry exploration. This review examines the asymmetry in the hemispheric white matter and functional network to assess health and brain disorders. In this article, the brain asymmetry in structural and functional connectivity including network topologies of healthy individuals, involving brain cognitive systems and the development trend, is highlighted. Moreover, the abnormal asymmetry of the hemispheric network related to cognition changes in brain disorders, such as Alzheimer’s disease, schizophrenia, autism spectrum disorder, attention deficit hyperactivity disorder, and bipolar disorder, is presented. This review suggests that the hemispheric network is highly conserved for measuring human brain asymmetries and has potential in the study of the cognitive system and brain disorders.
1 Brain asymmetry in a network perspective
Brain structural and functional differences between the left and right hemispheres are referred to as
The human brain is a
This review article aims to describe the brain asymmetry of structural and functional systems for healthy individuals and discuss atypical asymmetries in neurological and psychiatric disorders. In this review, the definition of a hemispheric structural and functional network is first proposed. Then, we provide a detailed review of brain asymmetry in structural and functional systems for healthy individuals. Notably, we focused on the key findings of the graph theory analysis of hemispheric networks and summarized the hemispheric network related to brain cognition lateralization for healthy individuals. Furthermore, we summarized the developmental processes of brain asymmetry in structural and functional systems for healthy individuals. Third, we discussed the abnormal brain asymmetry resulting in brain cognition changes in neurological and psychiatric disorders. Finally, we discuss some issues regarding future research on the hemispheric network.
2 Brain asymmetry in the structural and functional connectivity
Presently, the hemispheric structural network is mainly derived by reconstructing the WM fiber pathways within each hemisphere using
Complex network analysis of hemispheric structural and functional network
Using a complex network analysis, the hemispheric structural and functional network can be explored through the following six steps, as shown in Fig. 2: Magnetic resonance data acquisition: Diffusion tensor imaging data were used for the hemispheric structural network, and fMRI data were used for hemispheric functional network. Brain parcellation: Based on the brain parcellation atlas, these brain regions could be defined as the network nodes. Process data: A fiber-tracking algorithm is then performed to obtain the whole-brain WM tractography based on diffusion tensor imaging data. The time series from each region of interest is obtained by averaging the time series of each voxel within that region based on fMRI data. Generate hemispheric network: The connectivity could be defined as WM measurements, the connection probability between two regions of an individual diffusion tensor imaging dataset, the interregional correlations in the spectral coherence, or Granger causality measures between two magnetoencephalography sensors in groups of participants. The hemispheric network could be generated by compiling all pairwise associations between two nodes within the same hemisphere to produce a binary adjacency matrix or undirected graph. Characterize network topology: The topology parameters of the hemispheric network could be characterized at the global or nodal level. Calculate brain asymmetry: The brain asymmetry could be calculated using the formula: AS(X) = 100(X[R] – X[L])/(X[R] + X[L]), where X[R] and X[L] refer to the graph metric for the right and left hemispheric networks, respectively.
Network measures of graph theory
Small-world property: A network model with a combination of both random and regular properties, that is, high global efficiency (short path length) and high local efficiency (local clustering), respectively.
Hub/rich club: A hub is a highly central node that occupies an influential position within a network. A rich club is a set of hubs that are more densely interconnected to one another than expected by chance.
Network modularity: An important organizational principle of a network property that quantifies the extent to which a network is partitioned into individual subnetworks or modules. Many connections within modules and fewer connections between modules characterize a network with high modularity.
Shortest path length and efficiency: Path length is the minimum number of edges that must be traversed to go from one node to another. Efficiency is inversely related to path length, but is numerically easier to use to estimate topological distances between elements of disconnected graphs. These characteristics quantify the global efficiency (in terms of inverse path length) or the capacity for parallel information integration of a hemispheric network.
Nodal efficiency: This measures the information transmission ability of a node in the network. It is the inverse of the mean of the shortest path length between the node and all other nodes.
Nodal degree: The degree of a node is the number of connections that link it to the rest of the network. This is the most fundamental network measure and most other measures are ultimately linked to the node degree.
Clustering coefficient: The clustering coefficient quantifies the number of connections between the nearest neighbors of a node as a proportion of the maximum number of possible connections.
Betweenness centrality: This is defined as the number of the shortest paths between pairs of other nodes that pass through the node. Betweenness centrality is widely used to identify the most central nodes in a hemispheric network, which are associated with those nodes that act as bridges between the other nodes.
2.1 Asymmetry in structural connectivity
The structural asymmetry primarily assesses hemisphere differences in WM metrics. FA [29] and fiber number (FN) [30] are the most commonly used WM metrics. FA measures WM fiber organization and composition, and FN assesses tissue density of WM connectivity. For the FA metric, a larger number of WM studies revealed leftward asymmetry of the AF [31], cingulum bundles (CB) [32, 33], corticospinal tract (CT) [31, 32], inferior fronto-occipital fasciculus (IFOF) [33], and inferior longitudinal fasciculus (ILF) [33] for healthy adults. Additionally, Hidemasa et al. [31] observed rightward FA asymmetry in the frontal WM, UF, posterior corona radiata, and posterior corpus callosum in healthy young adults. For FN values, one structural study [34] using DTI found a significant leftward advantage in CT and direct connectivity between Broca’s and Wernicke territories and rightward asymmetry in the frontoparietal connections for healthy adults. Another DTI study [33] revealed a leftward asymmetry in the CB, ILF, and AF and a rightward asymmetry in the UF and OR for healthy adults. These studies indicate that brain WM is asymmetry.
2.2 Asymmetry in functional connectivity
For resting-state functional network using fMRI and
For the task-state functional connectivity, Di et al. [28] found a rightward functional connectivity in the occipital and inferior/middle frontal regions and leftward functional connectivity in the fusiform gyrus (FFG) and inferior frontal and inferior/superior parietal regions within the task-positive connectivity. Moreover, within the task-negative connectivity, Di et al. [28] observed a leftward functional connectivity between the PCUN and medial prefrontal regions. During the auditory oddball task, Swanson et al. [39] revealed a left-greater-than-right advantage in the MFG and inferior frontal gyri (IFG) activity, as well as a right-greater-than-left advantage in the supramarginal gyrus (SMG) activity for healthy individuals. Additionally, the EEG study [41] revealed a rightward brain asymmetry in the frontoparietal network that conforms during an auditory location oddball paradigm. These findings provide stringent evidence of brain asymmetry in the task-state functional connectivity.
2.3 The relationship between structural and functional asymmetry in the brain
Research reveals a close relationship between brain structural asymmetry and brain functional asymmetry [42]. For example, language ability is associated with the GM and WM structures in the left temporal lobe [43]. The right temporal lobe is related to spatial cognition and musical ability [44–46]. Furthermore, the WM fiber bundles between different hemispheres can transmit information between different brain regions, thereby affecting the functional lateralization of the brain [47].
Therefore, a complex and diverse relationship exists between brain structural asymmetry and functional asymmetry, and to better understand the above interaction, further research is warranted [48].
3 Graph theory analysis of hemispheric networks
In this section, we review studies on brain asymmetry in graph measures including small-world, hub/rich club, modularity, and nodal topology of brain structural and functional networks in diverse experimental modalities for healthy individuals.
3.1 Small-world architectures of hemispheric networks
Small-world architecture can be assessed through the clustering coefficient and the shortest path length (larger global efficiency), which facilitates efficient information segregation and integration with low wiring and energy costs [24]. A large number of structural WM studies revealed significant brain asymmetry in small-world topology for healthy adults. For example, the global efficiency of the right hemispheric network is larger than that of the left hemispheric network, which was first reported by Iturria-Medina et al. [49]. Defining the mean FA as the weight of the hemispheric network edge, several deterministic tractography studies [11, 16, 26, 33, 50] found rightward asymmetry of global efficiency. Defining the proportion of fibers intersecting two regions as the edge weight, a study on a large sample of 457 young adults [51] found a right hemispheric advantage in the small-world topology of the hemispheric network. Another probabilistic tractography study [52] revealed a rightward asymmetry in network efficiencies of the hemispheric WM network for young adults. Defining the fraction of streamlines as the weight of hemispheric WM networks, evidence of right hemispheric dominance for network efficiency was demonstrated by a recent graph theory analysis [27, 53]. Notably, Li et al. [50] found that right-handed participants had significant asymmetries in small-world property, whereas left-handed participants had fewer asymmetries, indicating that handedness has a great influence on brain asymmetry. These structural results provide direct evidence of rightward asymmetry in a small-world topology for right-handed healthy adults, indicating right hemisphere advantage in efficient information segregation and integration.
For hemispheric functional networks of healthy adults, compared with females, one early fMRI study [54] demonstrated that males have a higher normalized clustering coefficient in the right hemispheric network but a lower clustering coefficient in the left hemispheric network, indicating that sex has a great effect on the brain asymmetry of the functional network. Another EEG study [55] revealed that local efficiency showed a rightward advantage in a range of intermediate to high-density values in beta and gamma bands for a binary hemispheric network. Recent multimodal work [27] revealed that hemispheric functional networks exhibited significant rightward advantages for network efficiency. These functional studies suggested that healthy adults showed more efficient information segregation and integration in the right hemisphere than in the left hemisphere. However, functional research [56] revealed contradictory findings, showing that the degree of leftward asymmetry in local network efficiency was remarkably increased with childhood to adulthood development. The developmental patterns of leftward asymmetries suggest that the protracted maturation of functional segregation in the left hemisphere could underlie language development from childhood to adulthood for human brain functional networks.
3.2 Rich club organization of hemispheric networks
The presence of a rich club organization of interconnected hubs is a key feature of hemispheric networks. Hubs are crucial for optimal information flow in brain networks, and this is reflected by the correlation between the hub centrality levels [57]. For hemispheric structural networks, the DTI research [58] has revealed rightward asymmetry in the nodal betweenness of hub regions, including the middle occipital gyrus (MOG) and superior occipital gyrus (SOG) regions, for healthy adults. Our prior work [59] revealed that the hub PCUN region showed significant rightward asymmetry in nodal efficiency for healthy individuals. Additionally, our recent work [25] revealed that healthy individuals exhibited rightward asymmetry of the nodal degree in the temporal pole hub regions: TPOmid, PCUN, and MOG. For hemispheric functional networks, an fMRI study [60] found that the leftward hub regions included the medial prefrontal cortex, posterior cingulate cortex, Broca’s area, and Wernicke area, whereas the rightward hub regions included the anterior insula (INS).
The rich club organization divided the edges of the network into three types of connections: rich-club connections between two hub nodes, feeder connections from one hub node to one nonhub node, or local connections between two nonhub nodes. Our prior research [59] noted significant rightward asymmetries in connectivity density and feeder and local connection strength for healthy adults. Moreover, the male group exhibited significantly increased rightward asymmetry in both the density and strength of local connections compared with the female group [59], suggesting that sex affects the hemispheric network hub nodes. Our other research employed connectivity density and connectivity strength and found a significant rightward asymmetry in the rich club and local connections for healthy adults [25].
3.3 Brain modularity of hemispheric networks
A network module, which is often the basis for specialized subunits of information processing, is a group of densely interconnected nodes. The human brain has a well-defined modular organization as reported by many structural [61, 62] and functional [63, 64] neuroimaging studies, with the presence of large-scale functional networks [62]. Very recently, a structural study [53] applied the k-means clustering method to each population-averaged hemispheric matrix/ network to identify the WM modules within each hemisphere. They found that the healthy control group exhibited significant brain asymmetry in both the efficiency and mean connectivity strength of hemispheric modules. Currently, few studies on hemispheric network asymmetry based on modules exist. Thus, the modular topology of hemispheric brain networks remains unclear.
3.4 Nodal topology of hemispheric networks
Based on structural findings of hemispheric networks, the asymmetry in graph measures of some brain regions in the AAL atlas is relatively certain. Figure 1(A) depicts that those regions with leftward asymmetry are mainly located in the frontal lobe and cingulate gyrus. The frontal lobe involving the superior frontal gyrus [medial] (SFGmed), inferior frontal gyrus [opercular part] (IFGoperc), inferior frontal gyrus [triangular part] (IFGtriang), and supplementary motor area (SMA) regions showed a significantly leftward asymmetry in nodal efficiency [11, 16, 26, 32, 50], betweenness centrality [33, 49], and nodal degree [25, 59]. The cingulate gyrus, including the ACG and median cingulate gyrus regions, has a leftward predilection in nodal efficiency [11, 33, 49, 50], betweenness centrality [33, 49], and nodal degree [25]. Furthermore, the other two regions of the IPL and Heschl gyrus (HES) also exhibited significant left-greater-than-right nodal efficiency [11, 16, 26, 32, 33, 50] and nodal degree [25, 27, 59] for right-handed healthy adults. Several regions with rightward asymmetry are mainly located in the parietooccipital area and limbic system as depicted in Fig. 1(B). The parietooccipital area related to the angular gyrus (ANG), SMG, superior parietal gyrus, SOG, and CUN showed a significant rightward asymmetry in nodal efficiency [11, 16, 26, 32, 52], betweenness [33, 49, 58], and nodal degree [25, 27, 59]. The limbic system, including the hippocampus (HIP) and parahippocampal gyrus (PHG) regions, displayed a significant rightward advantage in nodal efficiency and betweenness [11, 32, 33]. Additionally, the TPOmid region [16, 25, 27, 52] displayed a right-greater-than-left nodal efficiency and degree.
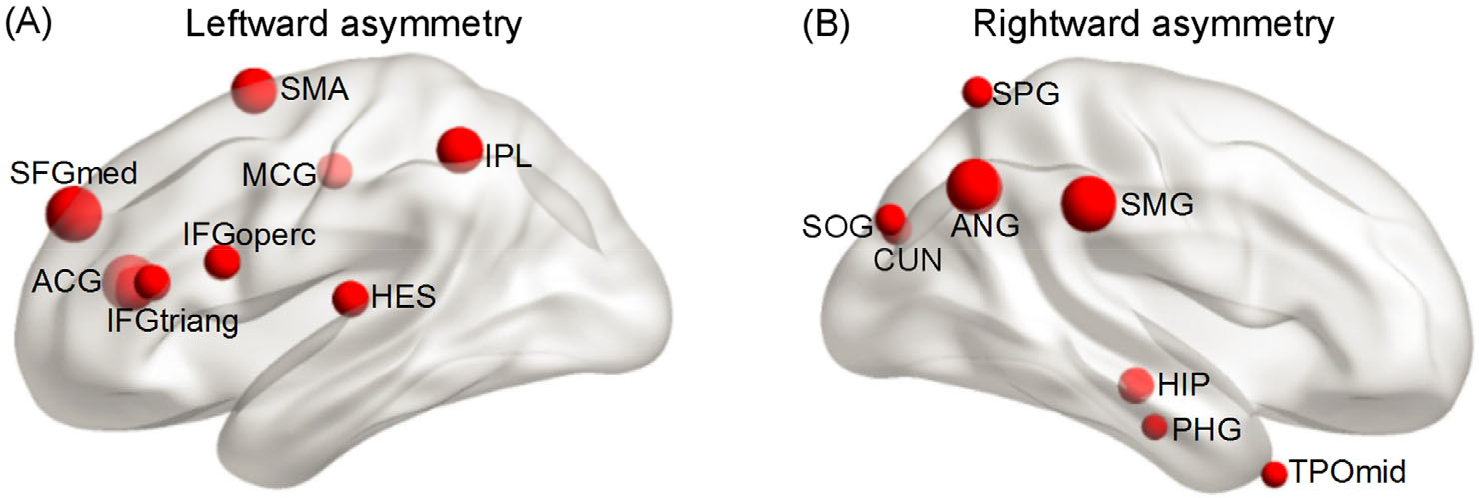
Brain asymmetry of graph measures in structural networks of healthy adults. Leftward asymmetry: supplementary motor area, inferior parietal lobule, median cingulate gyri, inferior frontal gyrus (opercular part), Heschl gyrus, superior frontal gyrus (medial part), anterior cingulate gyri, inferior frontal gyrus (triangular part). Rightward asymmetry: superior parietal gyrus, superior occipital gyrus, cuneus, angular gyrus, supramarginal gyrus, hippocampus, parahippocampal gyrus, and middle temporal gyrus.
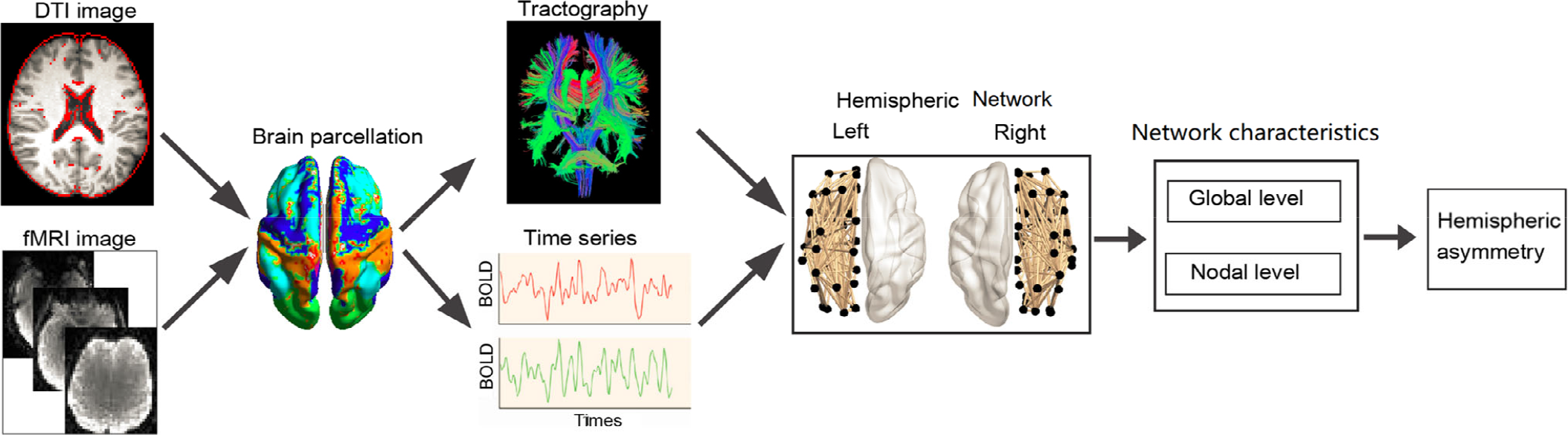
Complex network analysis of the hemispheric network.
The asymmetry of graph measures in some brain regions warrants further discussion. For example, the parietooccipital area comprising the PCUN, rolandic operculum, and lingual gyrus (LING) regions were found to have a leftward asymmetry in nodal betweenness [49] and nodal efficiency [32, 52, 65], but a rightward asymmetry in nodal efficiency [11, 16, 26, 27, 50, 59]. The temporal lobe involving the inferior temporal gyrus (ITG), STG, MTG, and FFG regions displayed leftward asymmetry in nodal efficiency in some studies [32, 50, 52] and rightward asymmetry in subsequent studies [11, 16, 26, 27, 59]. The precentral gyrus (PreCG) region showed a leftward asymmetry [32, 33, 49] and rightward asymmetry in some studies [16, 26, 50]. Some subcortical regions involving the amygdala (AMYG), putamen (PUT), and caudate nucleus (CAU) were found to exhibit leftward asymmetry in studies [32, 59] but rightward asymmetry in other researches [11, 16, 33, 50]. We speculate that the participants, data acquisition equipment, and network construction and analytical methods used in the research influence this inconsistent asymmetry of brain regions.
Compared to hemispheric structural studies, there is little functional work investigating brain asymmetry in graph metrics for healthy individuals. One early study [54] used rsfMRI to reveal a significant leftward asymmetry in nodal efficiency of the frontal lobe comprising the SFGmed, PreCG, and rectus gyrus regions and the occipital lobe including the MOG, calcarine cortex (CAL), and inferior occipital gyrus (IOG) regions and rightward asymmetry in nodal efficiency of the temporal lobe including the STG temporal pole: superior temporal gyrus (TPOsup), SMG, INS, and the ITG. This multimodel work [66] revealed that four regions comprising the orbitofrontal gyrus (inferior part), gyrus rectus, SFGdor, and SFGmed showed a leftward advantage, and the other six regions comprising the calcarine CAL, CUN, IPL, PCUN, SMG, and SOG mainly located in the parietooccipital area showed a rightward predilection of efficiency in elderly individuals. The results of another multimodel study [27] indicated a rightward predilection in the nodal degree of the frontal lobe, including the SFGdor, IFGtriang, MFG, ORBsup, and IFGoperc and the temporal lobe, including the MTG, TPOmid, STG; leftward asymmetry was observed in the CAU region. Based on
4 Hemispheric networks related to cognition lateralization
It is well-established that cognitive functions are lateralized, including the language, motor, attention, and memory systems. Many studies have shown that the lateralization of intrinsic cognition is highly associated with hemispheric network.
4.1 Language system
One of the most robust findings of cognitive neuroscience is the hemispheric lateralization for the language system [32]. A large number of structural and functional network studies have provided evidence of brain asymmetry related to language function. From the viewpoint of structural findings, healthy neonates [68] showed left hemisphere dominance for nodal efficiency in the FFG region involved in language functions. A diffusion-weighted MRI study [69] observed a leftward asymmetry in the language pathway connectivity between the temporoparietal junction (TPJ) and IFG for right-handed healthy adults, indicating leftward asymmetry in language function. For FA and FN values, two previous studies [33, 70] revealed leftward asymmetries in the most important language pathways of AF. Moreover, several graph theory analyses of hemispheric structural networks found significant leftward asymmetries in nodal efficiency [16, 32, 50, 52] and betweenness [33] of many language-relevant regions, including the STG, HES, IFGtriang, MTG, and ITG regions, supporting left hemispheric dominance for the language system. These findings proved that brain language function exhibits leftward asymmetries based on anatomical connection patterns.
From the viewpoint of functional findings, an early fMRI study [4] revealed left-asymmetric activity in the frontal and temporal regions that form part of the language system. Another two rsfMRI studies [60, 71] elucidated that healthy adults showed left hemispheric predilection in functional connectivity related to the language system composed of the Broca’s and Wernicke area. To investigate language functional hemispheric connectivity in adults, Hurley et al. [72] used a seed-based approach. They found that the left language functional connectivity between the IFG and MTG regions was stronger than that in the right hemisphere. A recent fMRI study [73] proposed a left hemisphere advantage in functional connectivity between language areas involving the frontal regions, suggesting left hemisphere dominance in language function. During the word recognition task, the MEG study [74] revealed that in the early stages of word processing, the left inferior frontal activity modulates the ventral occipitotemporal cortex. During a word completion task, the EEG study [75] revealed that leftward activation patterns were observed in the following regions of the pars opercularis (pOp), pars triangularis, MFG, subcentral gyrus, postcentral gyrus (PoCG), and PrCG for the healthy individual. These functional studies provide direct evidence of hemispheric lateralization of the language system.
4.2 Motor system
Beyond language, one of the most common findings of cognitive neuroscience is the hemispheric lateralization of the motor system. Graph theoretical analysis of the hemispheric structural networks in neonates [68] and healthy adults [11, 16, 32, 50] revealed significant leftward asymmetries in the nodal efficiency of several important motor regions defined by the AAL atlas, such as the PreCG, SMA, PUT, and pallidum. In addition, the SMA region also showed a leftward advantage in betweenness centrality [33, 49] and nodal degree [25, 59]. Graph theory analysis [54] revealed a significant leftward asymmetry in the nodal efficiency of the PreCG motor region in a functional hemispheric study. Another rsfMRI study [76] demonstrated the existence of motor network asymmetries in the resting state of healthy male adults. A study on the human motor network [77] that utilized rsfMRI proved that handedness substantially influences hemispheric functional connectivity. A functional study [78] based on seed regions investigated the brain asymmetry of connectivity related to the primary motor cortex (M1) and SMA in healthy right-handed participants. The results showed that the left M1 was more connected with areas than the right M1 and that the right SMA-proper had more functional connections than its left counterpart. These structural and functional findings suggested leftward brain asymmetry in the motor system.
4.3 Attention system
In right-handed healthy individuals, it is generally agreed that the attention system is the rightward asymmetry. In terms of its functional organization, the attention subsystem, including ventral attention and visuospatial attention, has been linked to an extensive set of brain regions. From the perspective of the hemispheric network, structural WM research [69] observed a rightward asymmetry in ventral attention network connectivity between the TPJ and INS regions and ventral attention network connectivity with the INS and dorsolateral prefrontal cortex. Notably, structural research [69] also found that the degree of rightward asymmetry in connectivity between the TPJ and INS region was stronger in females than in males, demonstrating that sex affects the attention subsystem. Additionally, several graph theory analyses [32, 33, 50, 54, 59] using DTI revealed rightward asymmetry in the nodal topology of visuospatial attention-relevant regions including the SMG and ANG defined by the AAL atlas, consistent with rightward asymmetry in visuospatial attention for right-handed healthy adults.
A rightward advantage in the connectivity of the attention subsystem was also elucidated by a functional research on the hemispheric network. For example, as a part of the distributed attention network, region of interest (ROI)-based functional connectivity analysis [37] showed a rightward advantage in connectivity strength of the cognitive ACG region associated with attention. Another fMRI study [4] demonstrated a rightward prediction in activity in the ANG and INS regions that have previously been associated with the attentional system. The complex network analysis of the hemispheric network [60] reported rightward functional asymmetry in the hub INS region in the attentional system. Based on graph theory analysis, a significant right hemispheric advantage in the nodal efficiency of the INS region was revealed by Tian et al. [54]. The EEG study [79], which utilized graph theory analysis of brain functional connectivity in the lower alpha band in the fatigued state, revealed a rightward asymmetric pattern of frontoparietal regions associated with sustained attention, supporting rightward attention function. From the perspective of the brain functional network, these findings demonstrated the rightward asymmetry of connections in the attention subsystem.
4.4 Memory system
From the perspective of the hemispheric network, studies have provided direct evidence of rightward asymmetry of the connectivity between brain regions in the memory subsystem. For instance, to reveal significant rightward asymmetries in the betweenness centrality of the HIP and AMYG regions in right-handed healthy adults, the hemispheric structural network [33] used graph theory. Other graph theory analyses found evident rightward asymmetry in the nodal efficiency of the memory system involving the PHG [11, 32], PCUN [16, 59], and CAU [11] defined by the AAL template in right-handed healthy adults. For the functional hemispheric network, Klostermann et al. [80] found that functional connectivity in the right CAU region correlated with working memory demands, supplying brain asymmetry in the memory system. These structural and functional studies provide evidence that brain asymmetry in the network is related to the right-lateralization of the memory system.
5 Brain asymmetry across the life span for healthy individuals
Together with previous work on brain asymmetry across different age groups [32, 49, 52, 66, 68], a trend in the topological asymmetry of hemispheric networks across the lifespan was observed. Because the developmental stage may continue from infancy into adulthood, there is a leftward predilection of network topology in neonatal and childhood brains. For instance, in neonatal brains, a leftward asymmetry of structural network efficiency was observed [68]. Functional research [81] based on voxel-wise connectivity showed an increase in left connectivity asymmetry in both language networks of the posterior STG and IFG from ages 3 to 5. Recent fMRI work [56] also reported leftward asymmetry in the network efficiency of hemispheric functional networks in a healthy childhood. These findings may underlie lateralized primary functional needs such as language and motor function development from neonatal stage to childhood. Different from leftward asymmetry in neonatal and childhood brains, rightward asymmetry in structural [11, 16, 27, 49, 59] and functional [27] network efficiency has been repeatedly proven in healthy adults. This rightward asymmetry in adults might be attributed to the significant development of cognitive processes in the right hemisphere, including visuospatial integration and attentional processing interactions, compared to the more specialized cognitive processes in the left hemisphere, such as language and motor processes [49].
In contrast to adults, rightward asymmetry disappears with advancing age. For example, several structural studies have shown the turnover of a right-larger-than-left asymmetry pattern in network efficiency [32, 66] and nodal degree involving the parietal and occipital lobes [82] in the elderly. Furthermore, Sun et al. [66] revealed symmetric network topology between the left and right hemispheric functional networks. These findings suggest that brain function degeneration in older adults is attributed to normal aging-related brain morphometric changes [83, 84].
6 Characterizing hemispheric networks in brain disorders
Since the work of pioneers, many neurological and psychiatric disorders manifest abnormal hemispheric connectivity. One main application of brain asymmetry in this context is to provide a new viewpoint to quantify differences in the hemispheric network between patient groups and appropriate comparison groups. Several studies have reported that the parameters of hemispheric networks derived from DTI, fMRI, EEG, and positron emission tomography (PET) data are altered in patients with AD, SZ, autism, ADHD, and BD [85].
6.1 Alzheimer’s disease
AD is a degenerative disorder. A structural study [8] revealed that fundamental anatomical connections were lost in the left hemisphere of patients with AD compared with healthy individuals, supporting lateralized deficits in network topologies of the left hemisphere. Another superficial WM study [86] demonstrated a decrease in the leftward asymmetry of hemispheric connectivity in the AD group compared with healthy adults, demonstrating left hemisphere disconnections. Using the graph theory, a comparable DTI study of the structural network [9] and a PET study of the functional network [10] confirmed a significant right-greater-than-left asymmetry in the network efficiency of the patient with AD owing to a less efficient communication of the left hemisphere compared to the communication capability in healthy individuals. These results showed that the structural and functional network of the left hemisphere degeneration is more rapid, and the damage is more severe in patients with AD. Language is mostly processed in the left hemisphere. Hence, these results imply that atypical topological asymmetries may relate to language function impairment in patients with AD [9].
Additionally, Cheng et al. [9] revealed that aberrations in hemispheric network efficiency asymmetry in patients with AD were correlated with memory performance. Moreover, they also found that an abnormal rightward asymmetry in patients with AD was primarily located in the PHG and CUN regions associated with episodic memory. These findings confirm that the abnormal brain asymmetry of WM is closely related to memory function in patients with AD. In summary, abnormal brain asymmetry in patients with AD extends our understanding of the neurophysiologic mechanisms of AD from a hemispheric network perspective.
6.2 Schizophrenia
An abnormal hemispheric network has been proposed to be related to memory and attention function in SZ. Park et al. [87] highlighted the aberrant FA asymmetry in the episodic memory areas of UF for patients with SZ. Consistently, another DTI study [88] showed an increased leftward FA and a decreased MD asymmetry in the posterior limb of the internal capsule related to visuospatial memory and episodic memory [89]. A third DTI study [90] found FA asymmetry in the middle of the superior longitudinal fasciculus (SLF) involved in visuospatial attention for SZ patients. For the functional network, one EEG study [91] observed that SZ patients showed a generally attenuated asymmetry compared to controls who had rightward asymmetry at the local level (EEG power) and intraregional levels (1st and 2nd order S-estimator), as well as global asymmetry. An fMRI study using tract-based spatial statistics (TBSS) [92] found that SZ patients showed a significantly altered brain asymmetry of connectivity related to the cognition-related ACC regions. To reveal a markedly abnormal brain asymmetry in connections of the memory-related CAU region in SZ, Mueller et al. [93] used fMRI. Moreover, a decreased asymmetry in connectivity of the memory regions of the PCUN [94] and HIP/PHG [95] was observed in patients with SZ. Hence, abnormal brain asymmetry accounted for the deficits in memory function in SZ.
Furthermore, abnormal asymmetry of the hemispheric network in auditory or language networks has been investigated in patients with SZ. Swanson et al. [39] revealed differences in asymmetry of the IPL, MOG, and LING between healthy individuals and patients in an auditory oddball task. A DTI study [96] showed that the RD and MD values in interhemispheric callosal fibers were positively correlated with leftward language lateralization in patients with SZ, suggesting that the integrity loss in interhemispheric callosal fibers was associated with a disrupted leftward dominance for language. Consistent fMRI studies reported that patients with SZ showed altered functional connectivity asymmetry in the primary language cortex of the planum temporale [97], HES [98], and MTG/ITG regions [94]. These findings could serve as evidence that the aberrant brain asymmetry of the structural and functional network of the language network is a feature of SZ. Additionally, several studies [35, 71, 93] also found a significant decline in brain asymmetry of the functional network in patients with SZ, reflecting hemispheric functional connectivity disruption.
Based on the graph theory analysis, SZ patients showed abnormal asymmetry in graph metrics related to brain function. For example, a DTI study [99] revealed longer average characteristic path lengths of hemispheric WM in males with SZ, suggesting an impaired informational exchange that may be associated with the deficits in cognition and abnormal behavior observed in SZ [100]. Accordingly, Sun et al. [11] reported a significantly reduced rightward asymmetry of network efficiency in SZ patients, reflecting atypical lateralization for broader processes such as visuospatial integration and attentional processing [49]. They [11] also revealed aberrant asymmetry in nodal efficiency of language regions involving the IFGoperc and STG, memory-related HIP region, and the attention-related IPL region in SZ patients, demonstrating changes in intrinsic cognition asymmetry. An fMRI research [12] revealed that the TPOmid region involving socioemotional processing displayed a leftward advantage in patients and a rightward predilection in healthy individuals. Moreover, symptom severity increased with the decline in superior temporal efficiency asymmetry and with the increase in subcortical efficiency asymmetry [12]. The usefulness of hemispheric structural and functional network properties as potential biomarkers for the diagnosis and evaluation of disease severity and for understanding the underlying pathophysiologic mechanisms is reflected by these graph theory analyses of hemispheric networks.
6.3 Autism disorder
In autism, the neurobiological alterations in the hemispheric network have been linked to language, motor, and memory function. Regarding language-related brain function, DTI studies showed that patients with autism display atypical asymmetries in the AF language system for adolescents [13] and in the pOp for young children [14]. An fMRI study [101] exhibited a reduced leftward asymmetry in connections involving the language regions of the Wernicke area in patients with autism. These findings provide evidence of atypical language lateralization in patients with ASD. In addition, the rsfMRI study [102] showed an atypical rightward asymmetry in functional lateralization, not only in language but also in motor and visuospatial circuits of the ASD group. Consistently, one multimodel study [103] showed a remarkably reduced asymmetry in functional connectivity of the primary motor cortex region of the PreCG. Another functional study [104] reported that children with ASD showed a rightward asymmetry in the mean motor circuit connectivity, which was associated with poorer performance on all three Physical and Neurological Examination for Soft Signs measures. In addition, an fMRI study [105] found an abnormal hemispheric connectivity density asymmetry in the emotion-related posterior cingulate cortex, visual-related LING, memory-related PHG, and primary sensory areas of the PoCG in patients with ASD, reflecting aberrant hemispheric lateralization of intrinsic cognition.
One DTI study [106] revealed a decreased rightward asymmetry of global efficiency in the ASD group, demonstrating aberrant lateralization of visuospatial integration and attentional processing, from the perspective of hemispheric networks with graph theory analysis [49]. Moreover, several regions in the present study [106] exhibited tendencies toward atypical development of asymmetry in nodal efficiencies, mainly involving the language system of the posterior MTG, the motor system of the THA and PUT, as evidence of changes that occur in the lateralization of specific cognitive processes.
6.4 Attention deficit hyperactivity disorder
Neuroimaging investigations have shown that ADHD is related to atypical asymmetry linked with brain functions. The WM research [107] revealed a reduced leftward FA asymmetry in the PUT–VLPFC and CAU–OFC tracts. These abnormal asymmetries were associated with inattention, hyperactivity, and Diagnostic and Statistical Manual of Mental Disorders criteria of ADHD. A DTI study [108] found that the mean absolute magnitude of hemisphere asymmetry in visual perception and reading of ILF and visuospatial attentional of SLF was increased in ADHD. Consistently, in patients with ADHD, functional research also found asymmetry of connections related to abnormal brain cognition. A leftward connectome pattern in controls but a rightward pattern in children with ADHD was confirmed in one fMRI study [15] on visual networks, demonstrating abnormal visual perception in children with ADHD. Another fMRI [109] study used a multivariate analysis of variance and found that genetic features have an impact on DMN FC asymmetry. Particularly, polymorphism rs8079626 affects information integration of the medial frontal gyrus and attention connectivity of the IPL asymmetry.
From the perspective of the hemispheric network, several studies found abnormal asymmetry of graph measures. For example, structural DTI research [16] showed a significantly reduced rightward brain asymmetry of global and local integration in patients with ADHD due to the significant increases in the network efficiency of the left hemisphere. Consistently, a functional EEG study [110] found atypical asymmetry of the clustering coefficient and characteristic path length of the delta EEG sub-band network of patients with ADHD because of significant differences in the left hemisphere. According to the notion presented by Iturria-Medina et al. [49], graph measure changes in the left hemisphere in patients with ADHD may represent an abnormal motor function. Moreover, our prior studies [16, 25] showed varying asymmetry patterns of nodal properties in motor regions of the PUT [111] in adults with ADHD, demonstrating altered motor function asymmetry in ADHD. These graph analyses of structural and functional studies provide proof of abnormal lateralization of the motor function.
6.5 Bipolar disorder
The first study [17] used TBSS and found that patients with BD had a significantly increased FA in the WM tracts of the left UF and significantly decreased FA in the right UF compared with healthy individuals, suggesting an increased leftward asymmetry in the UF. The UF is vital for mood regulation. Therefore, abnormal hemispheric FA asymmetry in the UF may contribute to mood dysregulation among patients with BD. Furthermore, hemispheric structural research [88] reported that patients with BD showed an increased leftward FA asymmetry in the WM subcortical tracts (the external and internal capsule) responsible for psychomotor function [112], as well as in the visuospatial attentional network of the IFOF [90] and in the information integration region of the corpus callosum [112].
Few studies exists using graph theory to quantify the topological properties of hemispheric networks in BD. A DTI study [26] observed a significant decline in the asymmetry of global efficiency, characteristic path length, and small-world property of hemispheric WM networks in patients with BD. According to cognitive theories, such absence in the rightward asymmetry in the global integration might underlie BD dysfunctions in attention and visuospatial abilities [113]. At the nodal level, our prior work also showed less rightward asymmetry mainly in the efficiency of regions located in the attention system of the SMG regions, visual processing of the SOG and LING regions in patients with BD [114], and an increased leftward asymmetry in the efficiency of the motor-related SMA region. These findings provide a new perspective on the brain asymmetry of brain networks and reveal abnormal brain functions in BD.
7 Concluding remarks and future perspectives
This paper elucidates the significant asymmetry of hemispheric network from five aspects: brain connectivity, graph theory, cognition system, life span, and disease. Brain structural research demonstrated that both FA and FN indices were asymmetrical. The functional connectivity of the right hemisphere is stronger than that of the left hemisphere. Handedness significantly influenced the asymmetry of graph theory index. For the cognition system, the language and motor systems are leftward asymmetries, while the attention and memory systems are rightward. A trend in the topological asymmetry of hemispheric networks across the lifespan was highlighted. Additionally, the use of hemispherical networks in brain diseases is a novel but a rapidly developing field. Several studies have examined structural and functional brain asymmetry in brain diseases and have found significant alterations in topological asymmetry from the perspective of hemispheric networks, demonstrating abnormal asymmetry in the cognitive system of diseases affecting the brain. In summary, research on brain asymmetry can help us better understand the nature of the brain and human behavior and also provide effective research ideas and methods to study brain diseases.
Limited literature exists elucidating the use of graph theory to investigate changes in the asymmetry of hemispheric networks in brain disorders. Moreover, it is noteworthy that current research on asymmetry in brain structure has mainly focused on the hemispheric level, highlighting the abnormality in corpus callosum connectivity as a result of brain asymmetry. Notably, the degree of brain asymmetry is not only closely related to brain disorder and age but is also affected by factors such as gender and handedness. Furthermore, the influence of sex and handedness on hemispheric asymmetries warrants further investigation. In addition, functional asymmetries are not always reflected or determined by structural asymmetries. Future studies are required to gain deeper insight into the relationship between structural and functional asymmetry.
Footnotes
Conflict of interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Natural Science Foundation of China (Grant Nos. 62176177, 61906130); the National Key R&D Program of China (Grant No. 2018AAA0102604); the Natural Science Foundation of Shanxi (Grant No. 20210302123112); and the Research Project Supported by Shanxi Scholarship Council of China (Grant No. 2021-039).
Authors’ contribution
Bin Wang, Dandan Li contributed to the conception of the work; Bin Wang, Dandan Li, Weichao An drafted the manuscript; Lan Yang, Wenjie Yan, Jie Xiang revised the manuscript. All the authors approved the final version.
